Scalable Synthesis of High-Purity Iopromide Intermediates via Atmospheric Reduction
Introduction to Advanced Iopromide Intermediate Manufacturing
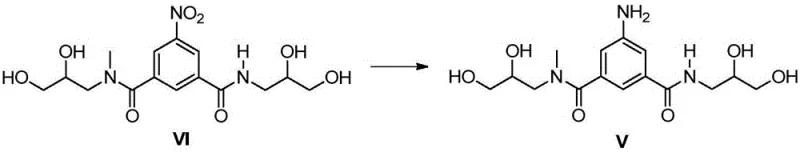
The global demand for non-ionic iodinated contrast agents continues to drive innovation in the synthesis of critical pharmaceutical intermediates. Patent CN111410615A introduces a transformative preparation method for the key iopromide intermediate, specifically 5-amino-N1,N3-bis(2,3-dihydroxypropyl)-N1-methyl-isophthalamide (Formula V). This technology addresses long-standing challenges in the industry by replacing hazardous high-pressure hydrogenation with a safer, atmospheric pressure reduction using Raney Nickel and hydrazine hydrate. For R&D directors and procurement specialists, this shift represents a significant opportunity to optimize production workflows while ensuring the supply of high-purity materials essential for angiography and CT imaging applications.
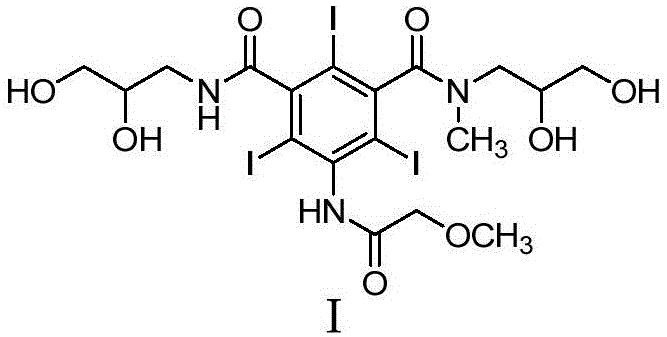
The strategic value of this patent lies in its ability to streamline the synthetic route from simple nitro-aromatic precursors to the complex triiodo final product. By focusing on the reduction step of the nitro group to an amine, the process eliminates the need for specialized pressure vessels, thereby lowering capital expenditure for manufacturing facilities. Furthermore, the use of water as a primary solvent aligns with modern green chemistry principles, reducing the environmental footprint associated with volatile organic compound (VOC) emissions. This report analyzes the technical merits and commercial implications of adopting this novel synthesis pathway for reliable pharmaceutical intermediates supplier operations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of iopromide and its precursors has been plagued by complex purification challenges and safety hazards. Prior art, such as U.S. Pat. No. 4,436,492, describes multiple routes that often suffer from the formation of difficult-to-remove diacylation byproducts, specifically the bismer represented by Formula 18. In these conventional pathways, the iodination steps frequently generate numerous side products, resulting in low purity intermediates that require extensive and costly chromatographic separation. Additionally, traditional reduction methods relying on catalytic hydrogenation necessitate the use of high-pressure reactors, which introduce significant operational risks and maintenance costs. Alternative metal reduction techniques using zinc powder have demonstrated poor efficiency, with reported purities ranging from only 48% to 73%, creating a bottleneck for downstream processing.
The Novel Approach
The innovative route disclosed in CN111410615A circumvents these issues through a meticulously designed sequence that prioritizes chemoselectivity and operational safety. The core breakthrough involves the reduction of the nitro-containing intermediate (Formula VI) using a Raney Nickel and hydrazine hydrate system. This method operates effectively at ambient temperature (20-25°C) and atmospheric pressure, completely removing the requirement for pressurized equipment. The reaction utilizes water or methanol as solvents, which are not only cost-effective but also facilitate easier product isolation compared to non-polar organic solvents. By avoiding the harsh conditions of previous methods, this approach minimizes degradation of the sensitive hydroxypropyl side chains, ensuring that the resulting amine intermediate (Formula V) is obtained with exceptional purity levels exceeding 96%.
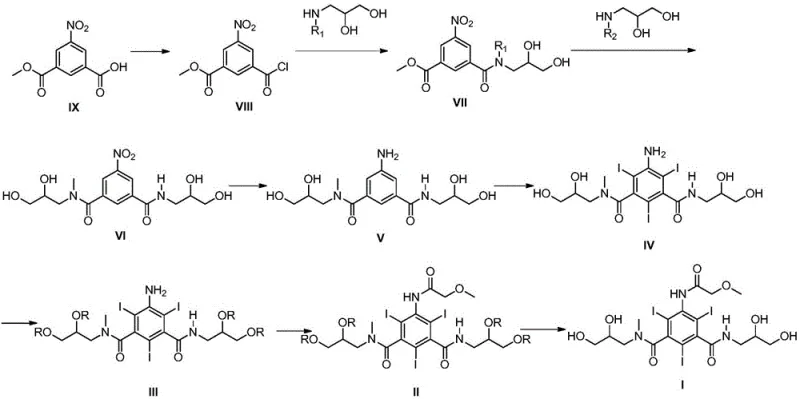
Mechanistic Insights into Raney Nickel Catalyzed Reduction
The heart of this improved synthesis lies in the mechanistic efficiency of the Raney Nickel/hydrazine hydrate reduction system. Unlike catalytic hydrogenation which requires gaseous hydrogen and high pressure to overcome activation energy barriers, this transfer hydrogenation method utilizes hydrazine as a hydrogen donor. The Raney Nickel catalyst provides a high surface area for the adsorption of both the nitro substrate and the hydrazine molecules. Upon contact, hydrazine decomposes on the catalyst surface to release active hydrogen species which subsequently reduce the nitro group (-NO2) to the primary amine (-NH2). This mechanism is highly selective, leaving the amide linkages and the hydroxyl groups on the side chains intact, which is crucial for maintaining the structural integrity of the molecule. The reaction proceeds smoothly in aqueous media, where the polar nature of the solvent helps stabilize the transition states and facilitates the dissolution of hydrazine hydrate.
From an impurity control perspective, this mechanism offers distinct advantages over zinc-mediated reductions. In zinc/acetic acid systems, the generation of zinc salts and the potential for over-reduction or incomplete reaction often lead to complex mixtures that are difficult to separate. In contrast, the Raney Nickel process generates nitrogen gas as the only byproduct, which simply evolves from the reaction mixture, leaving behind a clean solution of the desired amine. The catalyst can be easily removed by filtration and potentially recycled, further enhancing the economic viability of the process. The high purity of the resulting Formula V intermediate (96.4% vs 73% for zinc methods) significantly reduces the load on subsequent iodination and purification steps, ensuring a more robust and predictable manufacturing campaign for high-purity iopromide intermediate production.
How to Synthesize 5-Amino-N1,N3-bis(2,3-dihydroxypropyl)-N1-methyl-isophthalamide Efficiently
The synthesis of this critical intermediate begins with the preparation of the nitro precursor (Formula VI) through sequential acylation reactions, followed by the pivotal reduction step. The process is designed to be scalable, utilizing readily available reagents and standard laboratory equipment that can be easily adapted for industrial reactors. The key to success lies in the precise control of temperature during the addition of hydrazine hydrate and the maintenance of atmospheric conditions throughout the reduction phase. Detailed standard operating procedures regarding reagent ratios, stirring speeds, and workup protocols are essential for replicating the high yields and purity described in the patent documentation. For a comprehensive breakdown of the specific experimental conditions and stoichiometry required for each step, please refer to the standardized guide below.
- Prepare the nitro precursor (Formula VI) via sequential acylation of methyl 3-chloroformyl-5-nitrobenzoate with amino glycerol derivatives.
- Conduct the reduction reaction by adding Raney Nickel and hydrazine hydrate to the Formula VI solution in water or methanol at 20-25°C under atmospheric pressure.
- Filter the reaction mixture to remove the catalyst, adjust pH to 4-5, and proceed to iodination to form the triiodo intermediate (Formula IV).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers tangible benefits in terms of cost stability and operational reliability. The elimination of high-pressure hydrogenation equipment drastically reduces the capital investment required for setting up production lines, allowing for faster deployment of manufacturing capacity. Furthermore, the use of Raney Nickel and hydrazine hydrate, which are commodity chemicals with stable global supply chains, mitigates the risk of raw material shortages often associated with specialized noble metal catalysts like palladium or platinum. The ability to run the reaction in water also simplifies waste management and solvent recovery systems, leading to substantial cost savings in environmental compliance and utility consumption.
- Cost Reduction in Manufacturing: The transition to atmospheric pressure processing removes the need for expensive autoclaves and the rigorous safety inspections they require, directly lowering fixed asset costs. Additionally, the high purity of the intermediate reduces the consumption of silica gel and solvents needed for column chromatography purification, which is typically one of the most expensive steps in fine chemical manufacturing. The potential for recycling the Raney Nickel catalyst further contributes to a lower cost of goods sold (COGS) by minimizing catalyst turnover rates.
- Enhanced Supply Chain Reliability: By relying on robust, non-proprietary reagents such as zinc alternatives or hydrazine, the supply chain becomes less vulnerable to geopolitical fluctuations affecting rare earth or precious metal markets. The simplified reaction conditions also mean that production can be outsourced to a wider range of contract manufacturing organizations (CMOs) that may not possess high-pressure hydrogenation capabilities, thereby diversifying the supplier base and reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The aqueous nature of the reduction step aligns perfectly with increasingly stringent environmental regulations regarding VOC emissions. Scaling this reaction from pilot plant to commercial tonnage is straightforward since heat transfer and mixing are easier to manage in liquid-phase atmospheric reactions compared to gas-liquid high-pressure systems. This ease of scale-up ensures consistent quality across batches, which is critical for maintaining regulatory approval and market continuity for contrast agent manufacturers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the comparative data and experimental examples provided in the patent literature, offering clarity on why this method represents a superior choice for modern pharmaceutical manufacturing. Understanding these nuances helps stakeholders make informed decisions about process validation and vendor qualification.
Q: Why is the Raney Nickel/Hydrazine method preferred over catalytic hydrogenation for this intermediate?
A: The Raney Nickel/Hydrazine system operates at atmospheric pressure, eliminating the need for expensive high-pressure autoclaves required for traditional hydrogenation. This significantly reduces equipment costs and safety risks while maintaining high conversion rates and purity levels above 96%.
Q: How does this process avoid the diacylation byproducts mentioned in older patents?
A: By utilizing a stepwise acylation strategy starting from methyl 3-chloroformyl-5-nitrobenzoate and carefully controlling the stoichiometry of amino glycerol derivatives, the formation of the bis-acylated impurity (Formula 18) is effectively suppressed, simplifying downstream purification.
Q: What are the purity advantages of this specific reduction route?
A: Comparative data indicates that metal reduction methods like Zinc/Acetic Acid yield purities as low as 48-73%, whereas the disclosed Raney Nickel method consistently achieves purities around 96.4%, reducing the burden on subsequent chromatographic separation steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Iopromide Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the production of life-saving diagnostic agents. Our technical team has extensively analyzed the route disclosed in CN111410615A and possesses the expertise to implement this atmospheric reduction technology at scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of iopromide intermediate meets the exacting standards required for regulatory submission.
We invite you to collaborate with us to leverage these process improvements for your specific production goals. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your current manufacturing setup. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your transition to this advanced synthesis method is seamless, compliant, and economically advantageous.
