Advanced Photocatalytic Synthesis of 2-Aminothiazole Pyrimidine Intermediates for Commercial Scale-Up
Introduction to Next-Generation CDK2 Inhibitor Synthesis
The pharmaceutical landscape for oncology treatments is continuously evolving, with Cyclin-Dependent Kinase 2 (CDK2) inhibitors representing a critical class of therapeutic agents. Patent CN111777604B introduces a groundbreaking synthetic methodology for producing 2-aminothiazole pyrimidine derivatives, which serve as potent CDK2 inhibitors. This technology addresses a longstanding bottleneck in medicinal chemistry: the efficient introduction of fluorine atoms into heterocyclic scaffolds. Traditionally, fluorination has been a hazardous and low-yielding step, often requiring extreme conditions that compromise safety and scalability. The disclosed innovation leverages visible-light photocatalysis to overcome these barriers, offering a pathway to high-purity intermediates essential for drug development. By shifting from cryogenic batch processes to ambient temperature photochemical reactions, this method not only enhances chemical efficiency but also aligns with modern green chemistry principles, making it a vital asset for forward-thinking pharmaceutical manufacturers seeking reliable sources for complex heterocyclic building blocks.
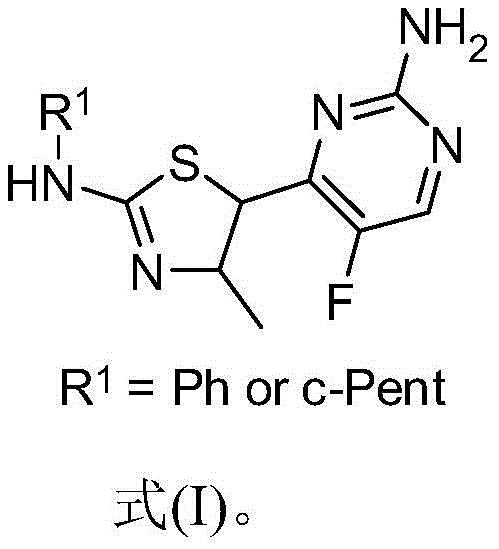
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fluorinated thiazole pyrimidines has been plagued by severe operational inefficiencies and poor atom economy. Conventional protocols typically mandate the use of aggressive fluorinating reagents under strictly controlled cryogenic conditions, often necessitating ice-salt baths maintained at -5°C for extended periods, such as three hours or more. These harsh thermal requirements impose a significant energy burden on manufacturing facilities and introduce complex engineering challenges for heat exchange and temperature control at scale. Furthermore, the chemical outcome of these traditional routes is frequently suboptimal, with reported yields hovering around a mere 35%. Such low conversion rates result in substantial material waste and generate complex impurity profiles that are difficult to manage. Post-reaction processing is equally burdensome, often requiring labor-intensive column chromatography to isolate the desired product, a technique that is notoriously difficult to translate from the laboratory bench to multi-kilogram production lines due to solvent consumption and throughput limitations.
The Novel Approach
In stark contrast, the methodology outlined in the patent utilizes a sophisticated photocatalytic strategy that fundamentally redefines the fluorination step. By employing a ruthenium-based photocatalyst, such as Ru(bpy)3(PF6)2, in conjunction with SelectFluor under visible light irradiation, the reaction proceeds efficiently at room temperature. This shift eliminates the need for energy-intensive cooling systems, allowing the fluorination to complete in just one hour with yields soaring to between 83% and 85%. The mildness of the reaction conditions preserves the integrity of the sensitive thiazole and pyrimidine rings, minimizing degradation and side-product formation. Additionally, the workup procedure is dramatically simplified; the high purity of the crude reaction mixture allows for isolation via simple recrystallization rather than chromatography. This streamlined approach not only accelerates the timeline from raw materials to finished intermediate but also drastically reduces the environmental footprint and operational costs associated with solvent disposal and energy consumption.
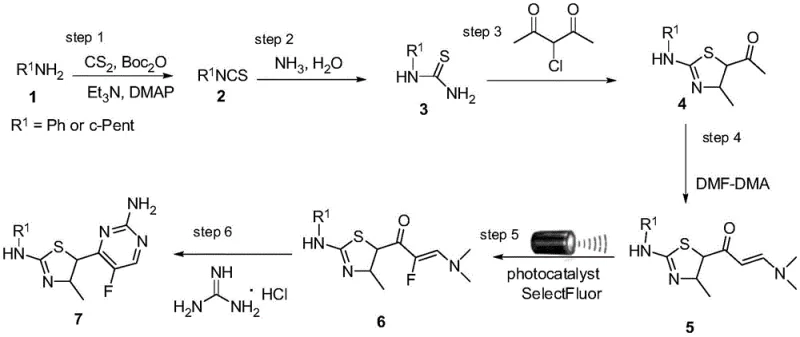
Mechanistic Insights into Photocatalytic Fluorination
The core innovation of this synthetic route lies in the mechanistic elegance of the photocatalytic fluorination step. The process initiates when the photocatalyst absorbs visible light energy, promoting an electron to a higher energy state and creating a potent oxidizing species. This excited state catalyst interacts with the enaminone substrate and the fluorinating reagent, SelectFluor (1-chloromethyl-4-fluoro-1,4-diazoniabicyclo[2.2.2]octane bis(tetrafluoroborate)), facilitating a single-electron transfer (SET) process. This mechanism generates a radical intermediate that selectively installs the fluorine atom at the alpha-position of the carbonyl group. Unlike electrophilic fluorination which can be indiscriminate and harsh, this radical pathway offers superior regioselectivity and functional group tolerance. The use of organic solvents like acetonitrile or DMA further stabilizes these intermediates, ensuring a smooth transformation. This mechanistic understanding is crucial for R&D directors, as it highlights the robustness of the chemistry against varying substrate electronic properties, ensuring consistent quality across different batches of the CDK2 inhibitor precursors.
From an impurity control perspective, the photocatalytic mechanism offers distinct advantages over thermal alternatives. Traditional thermal fluorination often leads to over-fluorination or decomposition of the heterocyclic core due to the high energy input required to overcome activation barriers. In the photocatalytic regime, the energy is delivered precisely via photon absorption, activating only the specific catalytic cycle required for mono-fluorination. This precision minimizes the formation of poly-fluorinated byproducts or ring-opened degradation products that are common in thermal processes. Furthermore, the rapid reaction kinetics—completing in approximately 60 minutes—reduce the residence time of the product in the reactive medium, thereby limiting opportunities for secondary reactions. The result is a cleaner reaction profile where the major impurity is often just unreacted starting material, which can be easily removed during the subsequent recrystallization or carried through to the final cyclization step without impacting the quality of the final API intermediate.
How to Synthesize 2-Aminothiazole Pyrimidine Efficiently
The synthesis of these valuable CDK2 inhibitor intermediates follows a logical six-step sequence that transforms simple amines into complex fluorinated heterocycles. The process begins with the conversion of primary amines, such as aniline or cyclopentylamine, into isothiocyanates using carbon disulfide and di-tert-butyl dicarbonate. These isothiocyanates are then treated with ammonia to generate thiourea derivatives, which serve as the nucleophilic partners for the subsequent Hantzsch thiazole synthesis. Reaction with 3-chloro-2,4-pentanedione constructs the thiazole core, which is then functionalized into an enaminone using DMF-DMA. The pivotal fifth step involves the aforementioned photocatalytic fluorination, installing the critical fluorine atom. Finally, the pyrimidine ring is closed via condensation with guanidine hydrochloride under basic conditions. This modular approach allows for the easy variation of the R1 group, enabling the rapid generation of diverse analog libraries for structure-activity relationship (SAR) studies while maintaining a robust and scalable backbone for commercial production.
- Convert primary amines to isothiocyanates using CS2 and Boc2O, then react with ammonia to form thiourea derivatives.
- Perform Hantzsch thiazole synthesis with 3-chloro-2,4-pentanedione, followed by enaminone formation using DMF-DMA.
- Execute photocatalytic fluorination at room temperature using SelectFluor and a ruthenium catalyst, then cyclize with guanidine hydrochloride.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this photocatalytic synthesis route translates directly into tangible operational efficiencies and risk mitigation. The most immediate impact is the elimination of cryogenic infrastructure requirements. Traditional fluorination processes demand specialized reactors capable of maintaining sub-zero temperatures, which represent a significant capital expenditure and a potential single point of failure in continuous manufacturing setups. By operating at ambient temperature, this new method allows for the utilization of standard glass-lined or stainless steel reactors, vastly expanding the pool of eligible contract manufacturing organizations (CMOs) and reducing dependency on specialized facilities. This flexibility enhances supply chain resilience, ensuring that production schedules are not disrupted by equipment availability or maintenance issues associated with complex cooling systems.
- Cost Reduction in Manufacturing: The economic implications of this technology are profound, driven primarily by the dramatic increase in chemical yield and the simplification of downstream processing. Increasing the yield of the fluorination step from roughly 35% to over 85% effectively more than doubles the output per unit of raw material input, significantly lowering the cost of goods sold (COGS). Furthermore, the replacement of column chromatography with recrystallization removes a major cost center; chromatography is solvent-intensive, time-consuming, and difficult to scale, whereas recrystallization is a standard, low-cost unit operation. The removal of expensive transition metal scavenging steps, often required for other catalytic methods, further contributes to cost optimization, making the final intermediate more price-competitive in the global market.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions directly correlates with improved supply continuity. Cryogenic reactions are sensitive to fluctuations in cooling capacity and can suffer from runaway exotherms if temperature control is lost, leading to batch failures and supply delays. The room-temperature photocatalytic process is inherently safer and more stable, reducing the risk of batch rejection due to thermal excursions. Additionally, the shorter reaction time—one hour compared to several hours plus cooling time—increases the throughput of existing manufacturing assets. This increased velocity allows suppliers to respond more rapidly to fluctuating demand from pharmaceutical clients, reducing lead times and ensuring a steady flow of high-quality intermediates necessary for clinical trial material and commercial API production.
- Scalability and Environmental Compliance: As regulatory pressure mounts regarding solvent usage and waste generation, this process offers a clear path to compliance. The ability to purify products via recrystallization significantly reduces the volume of organic waste generated compared to silica gel chromatography. Moreover, the use of visible light as a traceless reagent aligns with green chemistry metrics, improving the overall E-factor of the synthesis. The scalability of photocatalysis has matured significantly, with flow chemistry solutions now available to handle large volumes efficiently. This ensures that the transition from gram-scale laboratory synthesis to ton-scale commercial production is seamless, avoiding the typical pitfalls of scale-up where heat and mass transfer limitations often degrade performance in traditional batch processes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the practical benefits for potential partners and licensees. Understanding these nuances is essential for evaluating the feasibility of integrating this route into existing supply chains.
Q: What is the primary advantage of the photocatalytic fluorination method described in patent CN111777604B?
A: The primary advantage is the drastic improvement in reaction conditions and yield. Unlike traditional methods requiring cryogenic temperatures (-5°C) and yielding only ~35%, this photocatalytic approach operates at room temperature and achieves yields of 83-85%, significantly simplifying post-treatment.
Q: How does this synthesis route impact the purity profile of the final CDK2 inhibitor intermediate?
A: The route enhances purity by replacing complex column chromatography purification with simple recrystallization steps. The mild reaction conditions minimize side reactions and decomposition, resulting in a cleaner crude product that is easier to purify to stringent pharmaceutical specifications.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is highly scalable. The elimination of energy-intensive cryogenic cooling and the use of robust photocatalytic conditions make it ideal for kilogram-to-ton scale production, offering substantial operational cost reductions and improved supply chain reliability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Aminothiazole Pyrimidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the photocatalytic route described in CN111777604B. As a premier CDMO partner, we possess the technical expertise to translate such innovative laboratory protocols into robust, GMP-compliant manufacturing processes. Our facility is equipped with state-of-the-art photocatalytic reactors and extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We understand that the successful commercialization of CDK2 inhibitors depends not just on the chemistry, but on the consistency and purity of the supply. Our rigorous QC labs and stringent purity specifications ensure that every batch of 2-aminothiazole pyrimidine intermediate meets the exacting standards required for oncology drug development, providing our clients with the confidence to advance their pipelines without supply chain interruptions.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this cutting-edge technology for their specific projects. Whether you require custom synthesis of novel analogs or large-scale production of the standard intermediates, our team is ready to assist. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and detailed route feasibility assessments to demonstrate how our implementation of this photocatalytic process can optimize your budget and accelerate your time to market.
