Advanced Liquid-Phase Synthesis of Fmoc-Ser(tBu)-OH for Commercial Scale-Up of Complex Pharmaceutical Intermediates
The pharmaceutical industry constantly seeks robust synthetic routes for amino acid derivatives that balance safety, purity, and scalability. Patent CN112094205A, published in December 2020, introduces a significant technological breakthrough in the preparation of Fmoc-Ser(tBu)-OH, a critical building block for solid-phase peptide synthesis. This invention addresses long-standing challenges in side-chain protection by shifting from hazardous gas-phase reactions to a controlled liquid-phase methodology. The core innovation lies in the strategic sequencing of protective group introduction, specifically prioritizing the Fmoc group installation before the tert-butyl ether formation. This approach not only mitigates the safety risks associated with traditional isobutylene usage but also dramatically enhances the chiral integrity of the final product. For R&D directors and procurement specialists, understanding this shift is vital, as it represents a move towards more sustainable and reliable supply chains for high-value peptide intermediates. The method utilizes readily available reagents such as tert-butyl acetate and perchloric acid, ensuring that the process remains economically viable while meeting stringent quality standards required for GMP manufacturing environments.
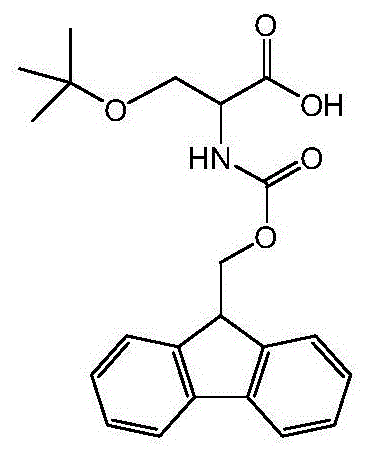
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of O-tert-butyl protected serine derivatives has relied heavily on the use of isobutylene gas for the introduction of the tert-butyl group. As illustrated in prior art literature, such as the work by Li Xin et al., this gas-phase alkylation presents severe operational hurdles for large-scale manufacturing. Isobutylene is a volatile gas at normal temperature and pressure, creating substantial safety hazards due to its tendency to form explosive mixtures with air upon leakage. Containing such gases requires specialized high-pressure reactors and rigorous leak detection systems, which drastically inflate capital expenditure and operational complexity. Furthermore, conventional synthetic routes often introduce the tert-butyl group before the Fmoc protection, necessitating a saponification step under basic conditions. This sequence exposes the alpha-carbon to strong bases, significantly increasing the risk of racemization and compromising the optical purity of the final amino acid. Such impurities are unacceptable in peptide drug manufacturing, where stereochemical integrity is paramount for biological activity and regulatory approval.
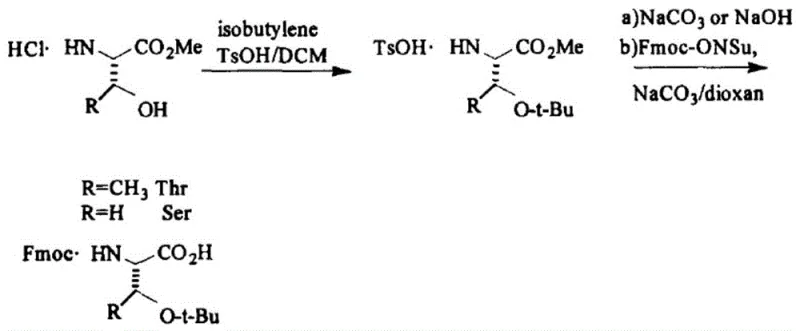
The Novel Approach
The methodology disclosed in CN112094205A fundamentally reengineers this workflow by adopting a liquid-phase tert-butylation strategy that eliminates the need for gaseous reagents. Instead of isobutylene, the process employs a mixture of tert-butyl acetate, perchloric acid, and tert-butanol to effect the protection of the serine hydroxyl group. This liquid-phase reaction occurs under mild conditions, typically between 15°C and 40°C, allowing for precise thermal control and eliminating the explosion risks inherent to gas handling. Crucially, the patent mandates the introduction of the Fmoc group prior to the tert-butyl protection. By securing the amine functionality first, the molecule is better shielded during subsequent processing steps. The final deprotection of the ester moiety is achieved using Lewis acids rather than traditional strong bases, a modification that preserves the chiral center. This novel sequence results in a product with exceptional chiral purity, often exceeding 99.9%, while simplifying the engineering requirements for the production facility, thereby offering a compelling value proposition for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Lewis Acid-Mediated Ester Hydrolysis
The chemical elegance of this patent lies in its manipulation of orthogonal protecting group stability. In traditional synthesis, removing a methyl or ethyl ester from a serine derivative usually requires sodium hydroxide or potassium hydroxide. However, these strong nucleophilic bases can attack the chiral alpha-proton, leading to epimerization and the formation of D-isomers. The patented process circumvents this by utilizing Lewis acids, such as aluminum trichloride (AlCl3) or lithium iodide (LiI), to cleave the ester bond. Lewis acids function by coordinating with the carbonyl oxygen of the ester, increasing the electrophilicity of the carbonyl carbon and facilitating cleavage by weak nucleophiles or through thermal elimination, without generating the high concentration of hydroxide ions that drive racemization. This mechanistic choice is critical for maintaining the high enantiomeric excess observed in the experimental data. Additionally, the presence of the bulky Fmoc group provides steric hindrance that further protects the chiral center from unwanted side reactions during the acidic tert-butylation step.
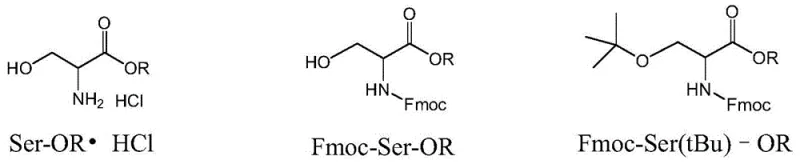
Furthermore, the inclusion of tert-butanol in the reaction mixture serves a dual purpose: it acts as a solvent and a stabilizer. During the acid-catalyzed formation of the tert-butyl ether, the equilibrium can sometimes favor the reverse reaction (deprotection) if water is present or if the acid concentration is too high. The excess tert-butanol drives the equilibrium forward according to Le Chatelier's principle, ensuring high conversion rates. The patent specifies a molar ratio optimization where the Lewis acid is used in a 5:1 to 8:1 ratio relative to the substrate, ensuring complete hydrolysis without degrading the acid-sensitive tert-butyl ether or the base-sensitive Fmoc group. This delicate balance of reactivity allows for the isolation of Fmoc-Ser(tBu)-OH with purity levels reaching 99.9% and chiral purity up to 100% in optimized examples, demonstrating the robustness of the mechanism against impurity formation.
How to Synthesize Fmoc-Ser(tBu)-OH Efficiently
The synthesis protocol outlined in the patent provides a clear, three-stage pathway that is amenable to standard chemical processing equipment. The process begins with the protection of the amine group on a serine ester hydrochloride salt using Fmoc-OSu in a biphasic or mixed solvent system of acetone and water, buffered to a pH of 8-9. Following the isolation of the Fmoc-Ser-OR intermediate, the critical tert-butylation step is performed by dissolving the solid in tert-butyl acetate and adding perchloric acid and tert-butanol. This mixture is stirred at ambient temperatures until TLC analysis confirms complete consumption of the starting material. The final step involves refluxing the protected ester in ethyl acetate with a stoichiometric amount of anhydrous Lewis acid, followed by an aqueous workup to isolate the free acid. Detailed standardized operating procedures for scaling this route from laboratory to pilot plant are essential for maximizing yield and safety.
- React Ser-OR·HCl with Fmoc-OSu in alkaline conditions to form Fmoc-Ser-OR solid.
- Perform tert-butylation using tert-butyl acetate, perchloric acid, and tert-butanol at 15-40°C to yield Fmoc-Ser(tBu)-OR.
- Hydrolyze the ester using a Lewis acid (e.g., AlCl3) in ethyl acetate at 70-85°C to obtain the final Fmoc-Ser(tBu)-OH product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from gas-phase to liquid-phase chemistry offers tangible benefits beyond mere technical feasibility. The elimination of isobutylene removes a major bottleneck in facility scheduling, as reactions no longer require dedicated time slots in high-pressure gas manifolds or specialized containment zones. This flexibility allows for increased batch frequency and better utilization of existing reactor volume. Moreover, the safety profile improvement directly correlates to reduced insurance premiums and lower compliance costs associated with handling hazardous gases. The use of common organic solvents like ethyl acetate and acetone simplifies waste stream management, as these solvents are easily recovered and recycled through standard distillation columns, contributing to substantial cost savings in raw material consumption. The high chiral purity achieved reduces the burden on downstream purification processes, meaning less chromatography media and fewer recrystallization cycles are needed to meet specification, further driving down the cost of goods sold.
- Cost Reduction in Manufacturing: The replacement of expensive and logistically complex gas handling systems with liquid reagents significantly lowers the barrier to entry for production. By avoiding the need for specialized high-pressure equipment and the associated maintenance, manufacturers can achieve a more lean operational model. The high yields reported in the patent examples indicate efficient atom economy, minimizing the waste of valuable starting materials like Fmoc-OSu. Additionally, the ability to perform the reaction at near-ambient temperatures for the protection step reduces energy consumption compared to processes requiring cryogenic cooling or extreme heating, leading to a lower overall carbon footprint and utility costs.
- Enhanced Supply Chain Reliability: Relying on liquid reagents such as tert-butyl acetate and perchloric acid ensures a more stable supply chain compared to compressed gases, which are subject to stricter transportation regulations and potential shortages. The raw materials for this process are commodity chemicals available from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the method against racemization means that batch-to-batch variability is minimized, ensuring consistent quality for downstream peptide synthesizers. This reliability is crucial for maintaining continuous production schedules in the fast-paced pharmaceutical sector, where delays in intermediate delivery can halt entire drug development pipelines.
- Scalability and Environmental Compliance: The process is inherently scalable because it avoids the mass transfer limitations often encountered when bubbling gases into liquid reaction mixtures. Liquid-liquid reactions mix more homogeneously, allowing for straightforward scale-up from liters to cubic meters without significant re-engineering of the agitation or dosing systems. From an environmental perspective, the avoidance of volatile organic compound (VOC) emissions associated with gas venting improves the facility's environmental standing. The use of Lewis acids, while requiring careful quenching, generates inorganic salts that are easier to treat in wastewater facilities compared to complex organic byproducts, aligning with modern green chemistry principles and regulatory expectations for sustainable manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in CN112094205A, offering clarity on why this method represents a superior choice for modern peptide chemistry. Understanding these nuances helps stakeholders make informed decisions about technology adoption and vendor qualification.
Q: Why is the Fmoc-first strategy superior for chiral purity?
A: Introducing the Fmoc group before the tert-butyl protection and subsequent hydrolysis minimizes the risk of racemization. Traditional methods that saponify before Fmoc protection expose the chiral center to harsh basic conditions, whereas this patent utilizes selective Lewis acid hydrolysis which preserves stereochemistry.
Q: What are the safety advantages of this liquid-phase method?
A: This method eliminates the need for isobutylene gas, which is flammable and forms explosive mixtures with air. By using liquid reagents like tert-butyl acetate and tert-butanol, the process significantly reduces explosion hazards and simplifies containment requirements for industrial reactors.
Q: Can this process be scaled for industrial production?
A: Yes, the patent explicitly states the method is suitable for modern industrial production. The use of common solvents like ethyl acetate and acetone, along with manageable temperature ranges (15-85°C), ensures the process is controllable and scalable without requiring specialized high-pressure gas equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fmoc-Ser(tBu)-OH Supplier
At NINGBO INNO PHARMCHEM, we recognize that the quality of peptide therapeutics begins with the purity of the amino acid building blocks. Our technical team has extensively analyzed the methodology described in CN112094205A and possesses the expertise to implement this advanced liquid-phase synthesis at an industrial scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee >99.9% ee. We understand the critical nature of Fmoc-Ser(tBu)-OH in the synthesis of complex biologics and are committed to delivering a product that meets the highest international pharmacopoeia standards.
We invite pharmaceutical companies and contract research organizations to collaborate with us on optimizing their supply chain for peptide intermediates. By leveraging our technical capabilities, you can secure a stable source of high-quality materials while benefiting from our process efficiencies. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing excellence can support your drug development goals and accelerate your time to market.
