Advanced Catalytic Synthesis of Azosemide Intermediate for Commercial Scale-up
Advanced Catalytic Synthesis of Azosemide Intermediate for Commercial Scale-up
The pharmaceutical industry constantly seeks robust synthetic routes for critical loop diuretics, and the production of Azosemide relies heavily on the quality of its key precursor. Patent CN111689881A discloses a groundbreaking method for synthesizing 4-chloro-2-fluoro-5-sulfamoylbenzoic acid, a vital building block in the manufacture of this potent therapeutic agent. This technology addresses long-standing challenges in sulfonation chemistry by introducing a catalytic system that allows for milder reaction conditions and superior impurity control. For R&D directors and procurement specialists, understanding this shift from traditional harsh sulfonation to a catalyzed, staged-heating process is essential for securing a reliable pharmaceutical intermediate supplier. The strategic implementation of this patent not only enhances the chemical integrity of the supply chain but also offers a pathway to significant cost reduction in pharmaceutical intermediates manufacturing by drastically improving material throughput.
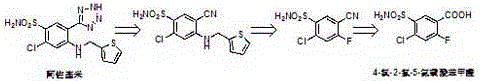
The significance of this intermediate cannot be overstated, as it serves as the direct precursor to Azosemide, a drug known for its长效 (long-acting) diuretic properties superior to furosemide in certain clinical applications. The structural fidelity of the sulfonamide group and the preservation of the halogen substituents during synthesis are paramount for biological activity. By leveraging the insights from this patent, manufacturers can transition from low-yield batch processes to a more efficient, scalable operation. This report analyzes the technical merits of this catalytic approach, providing a comprehensive view for stakeholders interested in the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-chloro-2-fluoro-5-sulfamoylbenzoic acid has been plagued by inefficiencies inherent in direct sulfonation using chlorosulfonic acid without catalytic assistance. Traditional protocols often require high temperatures that promote the formation of unwanted isomeric by-products, severely compromising the purity of the final crystalline product. Furthermore, the subsequent amination step in legacy processes frequently utilizes aqueous ammonia, which introduces a significant amount of water into the reaction matrix. This presence of water is detrimental because the intermediate sulfonyl chloride is highly susceptible to hydrolysis, leading to the degradation of the reactive species before it can be converted into the desired sulfonamide. Literature indicates that these conventional methods often struggle to achieve yields exceeding 51.1%, representing a massive loss of valuable starting materials and creating a bottleneck for reducing lead time for high-purity pharmaceutical intermediates.
The Novel Approach
The innovative methodology described in the patent fundamentally re-engineers the sulfonation landscape by incorporating specific inorganic salt catalysts, such as sodium chloride, potassium chloride, or lithium chloride, into the reaction mixture. This catalytic intervention allows the sulfonation of 4-chloro-2-fluorobenzoic acid to proceed at significantly lower temperatures, typically ranging between 80°C and 160°C, through a sophisticated staged heating protocol. By carefully controlling the thermal profile, the process effectively suppresses the generation of isomer impurities that typically arise under uncontrolled thermal stress. Moreover, the novel approach replaces hazardous and hydrolysis-prone aqueous ammonia with anhydrous alternatives like ammonia gas or solid ammonium salts. This strategic substitution eliminates the water-induced degradation pathway, ensuring that the sulfonyl chloride intermediate is preserved and efficiently converted. As illustrated in the reaction scheme below, this streamlined workflow results in a dramatic improvement in both yield and purity, establishing a new benchmark for high-purity OLED material and pharmaceutical intermediate production standards.

Mechanistic Insights into Catalytic Sulfonation and Hydrolysis Suppression
The core chemical advancement lies in the interaction between the chlorosulfonic acid and the added salt catalyst, which likely modifies the electrophilicity of the sulfur species or stabilizes the transition state during the electrophilic aromatic substitution. The use of catalysts such as lithium chloride or ammonium chloride facilitates the formation of the sulfonyl chloride moiety at the 5-position of the benzene ring with high regioselectivity. The staged heating mechanism is particularly ingenious; by initiating the reaction at a lower temperature (e.g., 80-110°C) and subsequently raising it (e.g., to 120-160°C), the system drives the reaction to completion while minimizing the kinetic energy available for side reactions that lead to structural isomers. This precise thermal management is critical for maintaining the integrity of the fluorine and chlorine substituents, which are essential for the downstream biological activity of the final diuretic drug.
Equally important is the mechanistic advantage gained during the amination phase. In traditional aqueous systems, the nucleophilic attack by ammonia competes with the nucleophilic attack by water molecules, leading to the formation of the corresponding sulfonic acid rather than the sulfonamide. By shifting to a solid-phase reaction or using dry ammonia gas in an organic solvent like isopropanol, the concentration of water is reduced to negligible levels. This anhydrous environment ensures that the nucleophilic substitution occurs exclusively at the sulfur center of the sulfonyl chloride, forming the stable sulfonamide bond. The subsequent acidification to a pH of less than 3 precipitates the product in its free acid form, allowing for easy isolation via filtration and recrystallization. This mechanism not only boosts the yield to impressive levels, such as the 90% reported in Example 3, but also ensures a liquid phase purity exceeding 99.6%, which is vital for meeting stringent regulatory requirements.
How to Synthesize 4-chloro-2-fluoro-5-sulfamoylbenzoic acid Efficiently
The operational execution of this synthesis requires careful attention to the stoichiometry of the catalyst and the thermal ramping rates to maximize the benefits of the patented method. The process begins with the suspension of the catalyst in chlorosulfonic acid, followed by the controlled addition of the benzoic acid substrate. Maintaining the specific temperature windows defined in the patent examples is crucial for suppressing isomer formation. Following the sulfonation, the quenching step must be performed rapidly and at low temperatures to prevent thermal degradation before the amination reagent is introduced. Whether utilizing gaseous ammonia or solid ammonium carbonate, the exclusion of moisture remains the golden rule for success. Detailed standardized synthesis steps see the guide below.
- Perform catalytic sulfonation of 4-chloro-2-fluorobenzoic acid with chlorosulfonic acid using a salt catalyst at staged temperatures (80-160°C).
- Conduct amination using ammonia gas or solid ammonium salts to minimize hydrolysis of the sulfonyl chloride intermediate.
- Execute acidification treatment to pH 2-3 followed by recrystallization to isolate the final high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic synthesis route translates into tangible operational efficiencies and risk mitigation strategies. The primary advantage is the substantial increase in process yield, which directly correlates to a reduction in the cost of goods sold (COGS). By moving from yields of approximately 35-51% in older methods to over 84-90% with this new technology, manufacturers can significantly reduce the volume of raw materials required per kilogram of finished product. This efficiency gain is not merely a laboratory statistic; it represents a drastic simplification of the supply chain logistics, as less waste needs to be managed and fewer batches are required to meet production targets. Consequently, this leads to significant cost savings in pharmaceutical intermediates manufacturing without compromising on quality.
- Cost Reduction in Manufacturing: The elimination of expensive purification steps required to remove isomeric impurities is a major driver of cost efficiency. Since the catalytic method inherently suppresses the formation of these by-products, the need for extensive chromatographic separation or multiple recrystallizations is minimized. Furthermore, the use of inexpensive, readily available inorganic salts as catalysts avoids the financial burden associated with precious metal catalysts often found in cross-coupling reactions. The ability to operate at lower temperatures also reduces energy consumption for heating and cooling cycles, contributing to a leaner and more economically viable production model that enhances overall profit margins.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures a more predictable and stable supply of the intermediate. Traditional methods prone to variable yields due to hydrolysis create uncertainty in production planning; however, the anhydrous amination technique described here offers consistent reproducibility. This reliability is critical for maintaining continuous API production lines and avoiding stockouts that could disrupt the availability of the final diuretic medication. By securing a source that utilizes this advanced technology, buyers can mitigate the risks associated with batch failures and ensure a steady flow of high-quality materials necessary for regulatory compliance.
- Scalability and Environmental Compliance: From an environmental perspective, the reduction in waste generation is a significant benefit. Higher yields mean less unreacted starting material and fewer by-products ending up in the waste stream, simplifying effluent treatment processes. The avoidance of large volumes of aqueous ammonia also reduces the load on wastewater treatment facilities. This aligns with modern green chemistry principles and helps manufacturers meet increasingly strict environmental regulations. The simplicity of the work-up procedure, involving straightforward filtration and recrystallization, makes the process highly amenable to scaling from pilot plant quantities to multi-ton commercial production without the need for specialized, hard-to-source equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this specific synthetic pathway. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this intermediate into their existing manufacturing portfolios. The answers provided are derived directly from the experimental data and claims within the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: How does the catalytic sulfonation method improve yield compared to conventional processes?
A: By introducing catalysts like sodium chloride or lithium chloride, the reaction temperature is lowered and staged heating suppresses isomer impurities, increasing yields from roughly 35% in older methods to over 90%.
Q: Why is solid-phase or gas-phase amination preferred over aqueous ammonia?
A: Aqueous ammonia introduces water which causes hydrolysis of the sensitive sulfonyl chloride group; using ammonia gas or solid salts eliminates this water source, significantly preserving the intermediate structure and improving overall purity.
Q: What represents the critical quality attribute for this intermediate in diuretic manufacturing?
A: High liquid phase purity (exceeding 99%) and controlled melting points are critical, as impurities in the 4-chloro-2-fluoro-5-sulfamoylbenzoic acid directly impact the efficacy and safety profile of the final Azosemide API.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-chloro-2-fluoro-5-sulfamoylbenzoic acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development and production of life-saving medications like Azosemide. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest international standards. Our capability to implement complex catalytic sulfonation and anhydrous amination techniques positions us as a strategic partner for global pharmaceutical companies seeking stability and excellence in their supply chain.
We invite you to collaborate with us to optimize your sourcing strategy for this essential diuretic intermediate. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our advanced manufacturing processes can lower your total procurement costs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us support your journey towards more efficient and reliable pharmaceutical production.
