Advanced Synthesis of Sacubitril Intermediates: Enhancing Purity and Scalability for Global Supply Chains
Advanced Synthesis of Sacubitril Intermediates: Enhancing Purity and Scalability for Global Supply Chains
The pharmaceutical landscape for cardiovascular treatments continues to evolve, with Sacubitril (AHU377) remaining a cornerstone molecule in the management of heart failure. As demand for high-quality active pharmaceutical ingredients (APIs) and their precursors grows, the efficiency of synthetic routes becomes paramount. Patent CN114409558A introduces a groundbreaking methodology for synthesizing a specific Sacubitril intermediate impurity, which serves not only as a critical reference standard for quality control but also as a versatile precursor for the main intermediate. This innovation addresses long-standing challenges in yield and operational safety, offering a robust pathway for manufacturers aiming to optimize their production lines. By leveraging a mild, aqueous-based alkaline hydrolysis technique, this technology promises to redefine the benchmarks for purity and process reliability in the synthesis of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
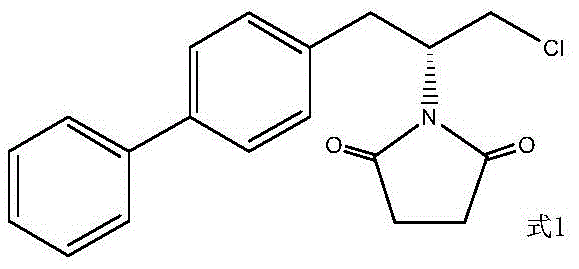
Traditional synthetic routes for Sacubitril intermediates, as illustrated in the comprehensive reaction scheme below, often involve a cumbersome eight-step sequence starting from S-epichlorohydrin. This legacy approach relies heavily on Grignard reactions, Mitsunobu couplings, and subsequent hydrolysis steps that are fraught with inefficiencies. Specifically, the hydrolysis of chlorinated hydrocarbons and acyl-limb bonds is typically conducted under acidic conditions in a one-pot fashion. While conceptually straightforward, this acid-catalyzed hydrolysis frequently suffers from relatively low yields and requires harsh reaction conditions that can compromise the integrity of sensitive functional groups. Furthermore, the generation of impurities during the alkaline hydrolysis phase of these conventional routes has historically been viewed as a detrimental side reaction, complicating purification and reducing overall material throughput.
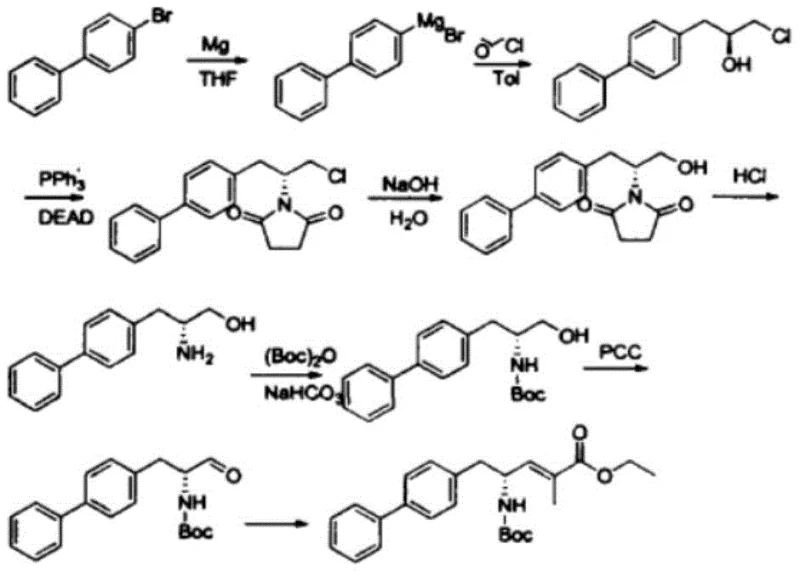
The Novel Approach
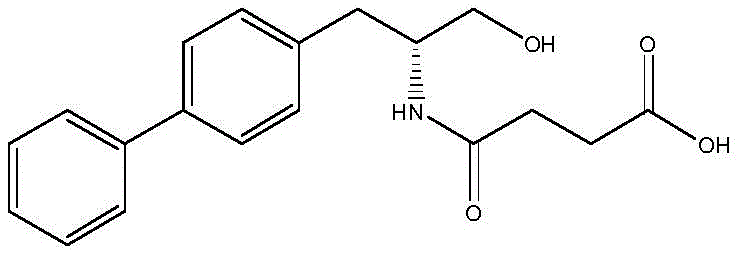
In stark contrast to the convoluted traditional pathways, the novel method disclosed in the patent capitalizes on the very impurities previously deemed problematic. By intentionally isolating and optimizing the formation of the intermediate generated during the alkaline hydrolysis of the Mitsunobu product, this approach transforms a liability into a valuable asset. The new process eliminates the need for aggressive acid hydrolysis in the initial stages, instead utilizing a controlled alkaline environment to simultaneously hydrolyze the chlorinated moiety to a hydroxyl group and open the lactam ring to form a carboxylic acid. This strategic shift not only simplifies the operational workflow but also drastically improves the reaction yield, achieving conversion rates exceeding 95%. The result is a streamlined, safer, and more environmentally benign process that aligns perfectly with modern green chemistry principles while delivering superior product quality.
Mechanistic Insights into Alkaline Hydrolysis and Ring Opening
The core of this technological advancement lies in the precise manipulation of reaction conditions to favor specific bond cleavages. The mechanism involves a dual-transformation where the starting material, a chlorinated lactam derivative obtained from the Mitsunobu reaction, undergoes nucleophilic attack by hydroxide ions. This attack first targets the terminal chlorinated carbon, displacing the chloride ion to form a primary alcohol. Concurrently, the basic conditions facilitate the hydrolysis of the cyclic amide (lactam) bond. This ring-opening reaction converts the cyclic imide structure into a linear amino acid derivative containing a free carboxylic acid group. The synergy between these two hydrolytic events is temperature-dependent, with optimal conversion observed between 80°C and 110°C. Maintaining this thermal window ensures complete reaction kinetics without degrading the sensitive biphenyl backbone or the newly formed chiral centers.
Controlling the impurity profile is critical for any pharmaceutical intermediate synthesis, and this method excels by design. The use of water as the primary solvent creates a homogeneous reaction medium that minimizes localized hot spots and concentration gradients, which are common sources of byproduct formation. By strictly regulating the mass ratio of the intermediate to water (1:10 to 1:20), the process ensures adequate solubility and fluidity, preventing the viscous mixtures that often lead to incomplete reactions or polymerization. Furthermore, the subsequent acidification step is carefully managed to precipitate the target product selectively. Adjusting the pH to less than 7 protonates the carboxylate anion, reducing its solubility and inducing crystallization. This precise pH control, combined with cooling to 0-20°C, effectively excludes soluble impurities from the crystal lattice, resulting in a final product with purity levels surpassing 96% directly from the reactor.
How to Synthesize Sacubitril Intermediate Impurity Efficiently
The implementation of this synthesis route is designed for ease of adoption in both laboratory and pilot plant settings. The protocol begins with the dissolution of the high-purity Mitsunobu intermediate in water, followed by the addition of a readily available inorganic base such as sodium carbonate or sodium hydroxide. The mixture is then heated to reflux temperatures to drive the hydrolysis to completion. Once the reaction is verified, the system is cooled, and the pH is adjusted using mineral acids to isolate the product. This straightforward sequence eliminates the need for exotic reagents or complex extraction procedures, making it an ideal candidate for rapid technology transfer. For detailed operational parameters and specific stoichiometric ratios, please refer to the standardized synthesis guide below.
- Dissolve the Mitsunobu reaction intermediate in water with a mass-volume ratio of 1: 10 to 1:20 under stirring.
- Add an alkaline reagent (e.g., sodium carbonate or sodium hydroxide) at a mass ratio of 0.5-1.5: 1 and heat to 80-110°C until reaction completion.
- Adjust pH to less than 7 using an acid reagent, cool to 0-20°C for crystallization, and filter to obtain the target product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis method offers tangible strategic benefits that extend beyond mere technical feasibility. The shift towards an aqueous-based system fundamentally alters the cost structure of manufacturing this critical intermediate. By replacing organic solvents with water, the process significantly reduces raw material costs and eliminates the substantial expenses associated with solvent recovery and disposal. Moreover, the high reaction yield (>95%) means that less starting material is required to produce the same amount of product, directly lowering the cost of goods sold (COGS). This efficiency gain is compounded by the high purity of the crude product, which often negates the need for resource-intensive recrystallization or chromatographic purification steps, further driving down operational expenditures.
- Cost Reduction in Manufacturing: The elimination of expensive organic solvents and the reduction in purification steps create a leaner manufacturing process. The use of commodity chemicals like sodium carbonate and hydrochloric acid ensures that reagent costs remain low and stable, shielding the supply chain from volatile market prices associated with specialized catalysts or reagents. Additionally, the high atom economy of the hydrolysis reaction minimizes waste generation, leading to substantial savings in waste treatment and compliance costs.
- Enhanced Supply Chain Reliability: The robustness of this aqueous process enhances supply continuity by reducing the risk of batch failures. The mild reaction conditions (80-110°C) are easier to control on a large scale compared to cryogenic or high-pressure alternatives, ensuring consistent output quality. Furthermore, the reliance on widely available raw materials mitigates the risk of supply disruptions, allowing for more flexible sourcing strategies and stronger negotiation leverage with upstream vendors.
- Scalability and Environmental Compliance: From a sustainability perspective, this method aligns with increasingly stringent environmental regulations. The absence of halogenated solvents and heavy metal catalysts simplifies the environmental permitting process and reduces the facility's ecological footprint. The scalability is inherent in the design; the simple work-up involving filtration and drying is easily adapted from kilogram to ton-scale production, facilitating rapid commercial scale-up of complex pharmaceutical intermediates without the need for major capital investment in specialized equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of what partners can expect when integrating this route into their manufacturing portfolio. Understanding these nuances is essential for R&D teams planning process validation and for procurement officers evaluating supplier capabilities.
Q: What are the advantages of using alkaline hydrolysis over acid hydrolysis for this intermediate?
A: Alkaline hydrolysis offers significantly higher yields (>95%) compared to traditional acid hydrolysis methods which often suffer from lower conversion rates. Additionally, the alkaline process operates under milder conditions (80-110°C) in an aqueous system, reducing safety risks and environmental impact.
Q: Can this synthesis method be scaled for commercial production?
A: Yes, the method utilizes water as the primary solvent and common inorganic bases, making it highly scalable and cost-effective. The simple work-up procedure involving pH adjustment and crystallization facilitates easy isolation of the product on a large scale.
Q: What is the purity level achievable with this patented method?
A: The patented process consistently achieves a target product purity of over 96% without the need for further purification steps, meeting stringent requirements for research and reference standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sacubitril Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global pharmaceutical supply chain. Our team of expert chemists has extensively analyzed the potential of the synthesis method described in CN114409558A and is fully prepared to deploy this technology for our clients. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition seamlessly from development to full-scale manufacturing. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Sacubitril intermediate meets the highest industry standards for consistency and performance.
We invite you to collaborate with us to leverage this advanced synthesis route for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized processes can enhance your supply chain efficiency and reduce your overall manufacturing costs.
