Industrial Scale-Up of Tebipenem Intermediate via Optimized Crystallization and Solvent Engineering
Industrial Scale-Up of Tebipenem Intermediate via Optimized Crystallization and Solvent Engineering
The pharmaceutical landscape for carbapenem antibiotics continues to evolve, driven by the urgent need for potent agents against resistant pathogens. Central to this advancement is the efficient production of key intermediates, specifically 1-(4,5-dihydrothiazol-2-yl)-3-mercaptoazetidine hydrochloride, a critical building block for the oral carbapenem prodrug Tebipenem (L-084). Patent CN102532117B discloses a transformative preparation method that addresses longstanding purification bottlenecks inherent in earlier synthetic routes. By shifting from complex multi-step sequences to a streamlined hydrolysis and crystallization protocol, this technology enables the production of high-purity solids exceeding 98% purity with yields over 80%. For R&D directors and procurement specialists, this represents a significant leap forward in process reliability and cost-efficiency.
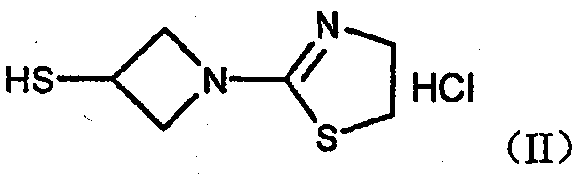
The structural integrity of the azetidine ring and the reactivity of the mercapto group pose unique challenges during synthesis. Traditional methods often struggle with the isolation of the free thiol species, which is prone to rapid oxidation into disulfide impurities. The patented approach mitigates these risks through precise temperature control and rapid processing times. Furthermore, the transition from oily residues to well-defined crystalline solids simplifies downstream processing, eliminating the need for extensive chromatographic purification. This technical breakthrough ensures a consistent supply of high-quality intermediates essential for the final assembly of beta-lactam antibiotics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art synthetic routes for this azetidine intermediate have been plagued by operational inefficiencies and purification difficulties. One established method, utilizing benzylamine as a starting material, involves a cumbersome multi-step sequence that results in relatively low overall yields. The complexity of protecting group manipulations and the requirement for expensive reagents render this pathway economically unviable for large-scale commercial production. Additionally, the monitoring of such lengthy reaction sequences introduces multiple points of failure, increasing the risk of batch variability and impurity accumulation.
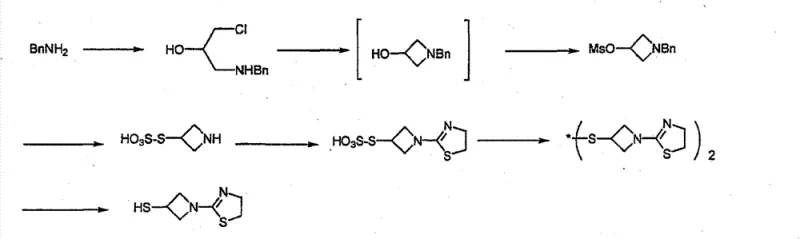
Another reported method employs benzhydrylamine, which offers improved reaction yields but introduces severe downstream processing challenges. While the chemical conversion steps are efficient, the isolation of the final product typically results in a thick, viscous oil rather than a solid. This physical state makes filtration and drying exceptionally difficult, often requiring the use of toxic solvents like acetonitrile for trituration. The reliance on acetonitrile not only raises environmental and safety concerns but also complicates solvent recovery systems. Moreover, the oily product tends to retain significant levels of disulfide impurities, compromising the quality required for pharmaceutical applications.
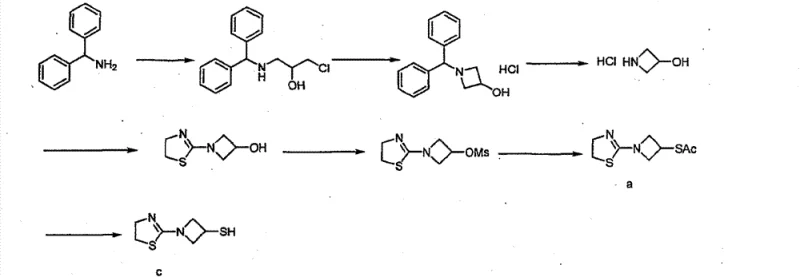
The Novel Approach
The methodology described in CN102532117B fundamentally reengineers the isolation step to overcome these physical and chemical barriers. Instead of relying on toxic acetonitrile or accepting oily residues, the inventors identified a specific binary solvent system comprising isopropanol and tetrahydrofuran (IPA/THF). This mixture creates a solubility profile that forces the rapid precipitation of the hydrochloride salt as a microcrystalline powder. The transition from oil to solid is not merely a cosmetic improvement; it drastically enhances the removal of mother liquor impurities, including the problematic disulfide dimers. By integrating this crystallization step with a single-solvent reaction phase (methanol), the process achieves a harmonious balance between reaction kinetics and product isolation.
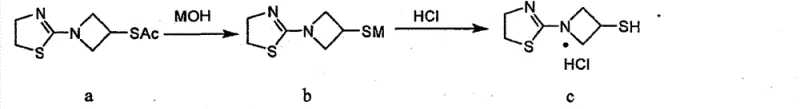
Mechanistic Insights into Alkaline Hydrolysis and Salt Formation
The core chemical transformation in this process is the selective hydrolysis of the thioacetate protecting group in Compound a to reveal the free thiol in Compound b. This reaction is mediated by an alkali metal hydroxide (such as KOH or NaOH) in methanol. The mechanism involves a nucleophilic attack by the hydroxide ion on the carbonyl carbon of the thioester, followed by the elimination of the acetate group. Crucially, this step must be performed at low temperatures, typically between -10°C and 0°C, to suppress side reactions. The azetidine ring is sensitive to harsh basic conditions, and prolonged exposure or elevated temperatures can lead to ring opening or polymerization. The patent specifies a remarkably short reaction window of merely 1 to 5 minutes, which is sufficient for complete conversion while minimizing degradation.
Following hydrolysis, the immediate addition of hydrochloric acid serves a dual purpose: it neutralizes the excess base and protonates the thiolate anion to form the stable hydrochloride salt (Compound c). This acidification step is critical for preventing the oxidation of the thiol to a disulfide. In basic media, thiolate anions are highly susceptible to aerobic oxidation. By rapidly lowering the pH, the concentration of the reactive thiolate species is reduced, effectively 'locking' the molecule in its reduced thiol form. The subsequent concentration and addition of the IPA/THF mixture leverage the differential solubility of the hydrochloride salt versus the neutral impurities. The specific volume ratio of 1:0.5 to 1:3 (IPA:THF) is optimized to maximize yield while ensuring the exclusion of organic byproducts, resulting in a product with purity levels consistently above 98%.
How to Synthesize 1-(4,5-Dihydrothiazol-2-yl)-3-mercaptoazetidine Hydrochloride Efficiently
Implementing this synthesis requires strict adherence to temperature profiles and solvent ratios to replicate the high purity observed in the patent examples. The process begins with the dissolution of the thioacetate precursor in methanol, followed by the controlled addition of the base. Operators must ensure that the exotherm is managed effectively to maintain the sub-zero temperature range. Once the hydrolysis is complete, the acidification should be performed swiftly to minimize the lifetime of the free thiolate. The final crystallization step is the most critical parameter for product quality; the ratio of isopropanol to tetrahydrofuran must be carefully adjusted based on the concentration of the residue to induce optimal nucleation. Detailed standardized operating procedures for this specific pathway are outlined below.
- Perform alkaline hydrolysis of the thioacetate precursor using methanol and a base like KOH or NaOH at low temperatures (-10 to 0°C) for a short duration to prevent oxidation.
- Neutralize the reaction mixture immediately with a methanol solution of hydrochloric acid to form the hydrochloride salt in situ.
- Concentrate the solution and induce crystallization using an isopropanol and tetrahydrofuran (IPA/THF) mixed solvent system at low temperatures to isolate the pure solid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers substantial strategic benefits beyond simple yield improvements. The shift away from complex multi-step syntheses reduces the number of unit operations required, directly correlating to lower labor costs and reduced equipment occupancy time. Furthermore, the simplification of the solvent system allows for more efficient recycling protocols. Methanol, being the primary reaction solvent, is widely available and inexpensive, and its recovery infrastructure is standard in most pharmaceutical manufacturing facilities. This contrasts sharply with processes requiring specialized or hazardous solvents that demand costly waste treatment or incineration.
- Cost Reduction in Manufacturing: The elimination of expensive starting materials like benzhydrylamine and the reduction of reaction steps significantly lowers the raw material cost per kilogram. Additionally, the short reaction time of only a few minutes reduces energy consumption for heating and cooling, contributing to a leaner manufacturing footprint. By avoiding the formation of oily products that require extensive reprocessing or chromatography, the facility saves on consumables such as silica gel and elution solvents. These cumulative efficiencies result in a markedly lower cost of goods sold (COGS) for the final intermediate.
- Enhanced Supply Chain Reliability: The robustness of the crystallization process ensures consistent batch-to-batch quality, reducing the risk of production delays caused by out-of-specification results. The ability to produce a free-flowing solid rather than a viscous oil improves handling characteristics, facilitating automated packaging and transport. Moreover, the use of common, non-restricted solvents like methanol, isopropanol, and THF mitigates supply chain risks associated with regulated or scarce chemicals. This reliability is crucial for maintaining continuous production schedules for downstream API manufacturing.
- Scalability and Environmental Compliance: From an environmental perspective, replacing acetonitrile with an IPA/THF mixture reduces the toxicity profile of the waste stream, simplifying effluent treatment compliance. The high purity achieved through crystallization minimizes the generation of hazardous waste associated with purification steps. The process is inherently scalable, as the heat transfer requirements for the short reaction time are manageable even in large reactors. This scalability ensures that the supply chain can respond flexibly to market demand fluctuations without requiring disproportionate capital investment in new equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and validation of this synthesis route. Understanding these nuances is vital for technology transfer teams evaluating the feasibility of adopting this process within existing GMP facilities. The answers are derived directly from the experimental data and comparative examples provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: Why is the IPA/THF solvent system critical for this synthesis?
A: Conventional single solvents like methanol or acetonitrile fail to precipitate the product as a solid, resulting in difficult-to-handle oils. The specific polarity balance of the IPA/THF mixture forces rapid crystallization, significantly improving purity and filtration efficiency.
Q: How does this method control disulfide impurities?
A: The process utilizes a very short reaction time (minutes) with alkali at low temperatures. This minimizes the exposure of the free thiol group to oxidative conditions, thereby drastically reducing the formation of disulfide dimers compared to longer prior art processes.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the method uses methanol as the primary reaction solvent, which is inexpensive and easily recycled. The elimination of toxic acetonitrile and the conversion of the product from an oil to a filterable solid make it highly amenable to industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-(4,5-Dihydrothiazol-2-yl)-3-mercaptoazetidine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of next-generation antibiotics relies on the availability of high-quality intermediates produced via robust and scalable pathways. Our technical team has extensively analyzed the process described in CN102532117B and possesses the expertise to implement this optimized crystallization technology at an industrial scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 1-(4,5-dihydrothiazol-2-yl)-3-mercaptoazetidine hydrochloride meets the exacting standards required for carbapenem synthesis.
We invite potential partners to engage with our technical procurement team to discuss how this advanced manufacturing route can enhance your supply chain resilience. By leveraging our capabilities, you can secure a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our commitment to process innovation translates into tangible value for your organization.
