Advanced Safety and Efficiency in Fmoc-Tyr(tBu)-OH Production for Global Peptide Supply Chains
The pharmaceutical and biotechnology sectors rely heavily on the consistent availability of high-quality protected amino acids for solid-phase peptide synthesis (SPPS). Among these critical building blocks, Fmoc-Tyr(tBu)-OH stands out as a fundamental component for incorporating tyrosine residues into complex peptide chains. However, the historical manufacturing processes for this intermediate have been plagued by significant safety hazards and operational inefficiencies. Patent CN112094204B introduces a transformative methodology that addresses these longstanding challenges by replacing hazardous gaseous reagents with safer liquid-phase alternatives and employing selective Lewis acid catalysis. This technical breakthrough not only enhances the safety profile of the manufacturing facility but also streamlines the production workflow, offering a compelling value proposition for reliable pharmaceutical intermediate suppliers aiming to optimize their supply chains. By shifting away from explosive isobutylene gas and utilizing a robust tert-butyl acetate system, this innovation sets a new standard for cost reduction in API manufacturing while ensuring the structural integrity of the sensitive Fmoc protecting group.
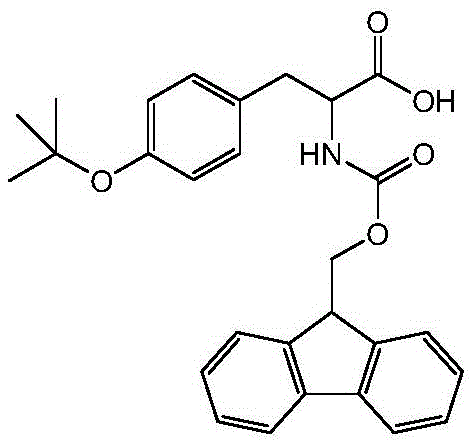
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of O-tert-butyl protected tyrosine derivatives has relied heavily on the use of isobutylene gas for the introduction of the tert-butyl group. As illustrated in prior art such as Patent CN103833593A and research by Li Xin et al., the conventional route typically involves a multi-step sequence starting from tyrosine, proceeding through Z-protection, followed by reaction with isobutylene gas to install the tert-butyl ether, and finally converting to the Fmoc derivative. This reliance on isobutylene presents severe logistical and safety bottlenecks for large-scale production. Isobutylene is a gas at normal temperature and pressure, requiring specialized high-pressure reactors and stringent leak detection systems to prevent the formation of explosive mixtures with air. Furthermore, the multi-step nature of the traditional Z-protection route—involving hydrogenolysis and subsequent Fmoc installation—drastically increases solvent consumption, waste generation, and overall processing time. These factors collectively contribute to higher production costs and increased supply chain vulnerability, making the conventional method suboptimal for modern, high-volume commercial scale-up of complex pharmaceutical intermediates.
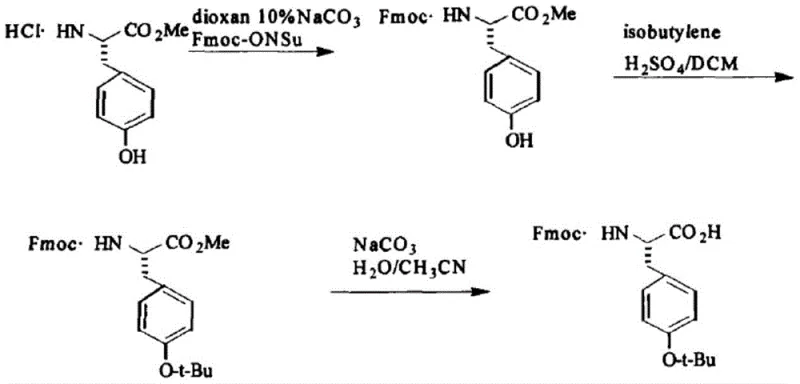
The Novel Approach
The methodology disclosed in CN112094204B fundamentally reengineers this synthetic pathway by eliminating the need for gaseous isobutylene entirely. Instead, the invention utilizes a liquid-phase reaction system comprising tert-butyl acetate, perchloric acid, and tert-butyl alcohol to effect the O-tert-butylation directly on the Fmoc-Tyr-OR precursor. This shift from gas to liquid reagents simplifies the reactor requirements, allowing the reaction to proceed under atmospheric pressure with standard agitation, thereby significantly lowering capital expenditure and operational risk. Moreover, the process integrates the Fmoc protection prior to the tert-butylation step, effectively shortening the synthetic sequence compared to the traditional Z-protection strategy. This streamlined approach not only improves the overall yield by minimizing unit operations but also enhances the controllability of the reaction parameters. The result is a more robust and scalable process that aligns perfectly with the industry's demand for high-purity OLED material and pharmaceutical precursors that can be manufactured with consistent quality and reduced environmental impact.
Mechanistic Insights into Lewis Acid-Catalyzed Selective Hydrolysis
A critical innovation within this patent is the specific method employed for the final hydrolysis step to convert the ester intermediate Fmoc-Tyr(tBu)-OR into the free acid Fmoc-Tyr(tBu)-OH. In traditional peptide chemistry, ester hydrolysis is frequently performed using strong bases such as sodium hydroxide or sodium carbonate. However, the Fmoc (9-fluorenylmethoxycarbonyl) group is inherently base-labile; exposure to alkaline conditions can lead to premature deprotection via beta-elimination, resulting in significant impurity profiles and reduced yields. The patent elegantly solves this chemoselectivity problem by employing Lewis acids, such as anhydrous aluminum trichloride (AlCl3), aluminum tribromide (AlBr3), or lithium iodide (LiI), as the hydrolytic agents. These Lewis acids coordinate with the carbonyl oxygen of the ester group, activating it towards nucleophilic attack by trace water or solvent molecules, while leaving the carbamate linkage of the Fmoc group intact. This mechanistic specificity ensures that the delicate protecting group strategy remains undisturbed throughout the final deprotection stage.
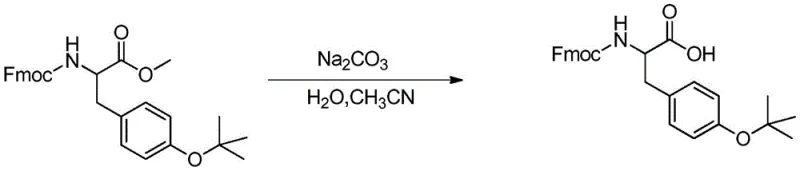
The superiority of the Lewis acid mechanism is further evidenced by comparative data within the patent. When traditional base-mediated hydrolysis using sodium carbonate was attempted (as shown in the comparative examples), the process yielded only 73.5% of the product with a purity of 96.8%. In stark contrast, the Lewis acid-mediated protocol consistently delivered yields exceeding 79% with purities surpassing 99.25%. This dramatic improvement in both yield and purity underscores the importance of matching the hydrolytic mechanism to the sensitivity of the substrate. By avoiding the harsh alkaline conditions depicted in comparative studies, the novel process minimizes the formation of dibenzofulvene and other Fmoc-degradation byproducts. This level of impurity control is vital for reducing lead time for high-purity pharmaceutical intermediates, as it reduces or eliminates the need for extensive recrystallization or chromatographic purification downstream, directly benefiting the procurement and manufacturing timelines of peptide drug developers.
How to Synthesize Fmoc-Tyr(tBu)-OH Efficiently
The implementation of this novel synthesis route requires precise control over reaction stoichiometry and conditions to maximize the benefits of the Lewis acid catalysis and the liquid-phase tert-butylation. The process begins with the preparation of the Fmoc-Tyr-OR precursor, followed by the critical acid-catalyzed etherification and the final selective hydrolysis. Operators must adhere to strict temperature controls, particularly during the exothermic addition of perchloric acid and the reflux conditions required for the Lewis acid step. The following guide outlines the standardized operational framework derived from the patent examples, ensuring reproducibility and safety at scale. For detailed laboratory protocols and specific equipment configurations, please refer to the technical documentation provided below.
- Esterify L-Tyrosine with alcohol and thionyl chloride to form Tyr-OR hydrochloride salt.
- Protect the amine group using Fmoc-OSu under alkaline conditions to yield Fmoc-Tyr-OR.
- Introduce the tert-butyl group on the phenolic hydroxyl using tert-butyl acetate and perchloric acid.
- Perform selective hydrolysis of the ester group using a Lewis acid (e.g., AlCl3) in ethyl acetate to obtain the final acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to the synthesis method described in CN112094204B offers tangible strategic advantages beyond mere chemical elegance. The elimination of isobutylene gas removes a significant bottleneck in raw material logistics, as sourcing and storing compressed explosive gases require specialized infrastructure and regulatory compliance that many facilities lack. By switching to liquid reagents like tert-butyl acetate and tert-butyl alcohol, the supply chain becomes more resilient and flexible, allowing for bulk purchasing and simpler storage solutions. Furthermore, the shortened synthetic route reduces the total volume of solvents and reagents consumed per kilogram of product, which translates directly into lower waste disposal costs and a reduced environmental footprint. These operational efficiencies enable suppliers to offer more competitive pricing structures while maintaining healthy margins, addressing the constant pressure for cost reduction in pharmaceutical intermediate manufacturing.
- Cost Reduction in Manufacturing: The novel process eliminates the need for high-pressure reactors and complex gas handling systems associated with isobutylene usage, resulting in substantial capital expenditure savings. Additionally, the higher selectivity of the Lewis acid hydrolysis minimizes product loss due to Fmoc deprotection, thereby improving the overall mass balance and reducing the cost of goods sold. The simplified workflow also reduces labor hours and utility consumption, contributing to a leaner and more cost-effective production model that can withstand market fluctuations.
- Enhanced Supply Chain Reliability: By relying on stable, liquid-phase reagents that are readily available in the global chemical market, the risk of supply disruption due to hazardous material transport restrictions is significantly mitigated. The robustness of the reaction conditions allows for consistent batch-to-batch quality, ensuring that downstream peptide synthesizers receive materials that meet stringent specifications without delay. This reliability is crucial for maintaining continuous production schedules in the fast-paced biopharmaceutical sector, where delays in intermediate delivery can halt entire drug development programs.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from kilogram to multi-ton production without the safety constraints imposed by explosive gases. The use of Lewis acids and the ability to isolate products via simple pH adjustment and filtration streamline the workup procedure, reducing the generation of aqueous waste streams. This alignment with green chemistry principles not only simplifies regulatory compliance but also enhances the corporate sustainability profile of the manufacturing entity, a key consideration for modern ESG-focused procurement strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Fmoc-Tyr(tBu)-OH using this advanced methodology. These insights are derived directly from the experimental data and technical specifications outlined in the patent literature, providing a transparent view of the process capabilities. Understanding these details helps stakeholders make informed decisions regarding vendor selection and process integration.
Q: Why is the elimination of isobutylene gas critical for industrial production?
A: Isobutylene is a volatile, explosive gas at room temperature that requires specialized high-pressure equipment and rigorous safety protocols. Replacing it with liquid reagents like tert-butyl acetate significantly reduces operational hazards and capital expenditure.
Q: What is the advantage of using Lewis acid for hydrolysis over traditional base hydrolysis?
A: Traditional base hydrolysis (e.g., using NaOH or Na2CO3) lacks selectivity and can cleave the Fmoc protecting group, leading to lower yields and purity. Lewis acids selectively cleave the ester bond while preserving the Fmoc group, achieving purities above 99%.
Q: What purity levels can be achieved with this novel synthesis route?
A: The patented method consistently achieves product purities exceeding 99.25%, with some embodiments reaching 99.6%, significantly reducing the burden on downstream purification processes for peptide manufacturers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fmoc-Tyr(tBu)-OH Supplier
At NINGBO INNO PHARMCHEM, we recognize that the quality of your final peptide therapeutic is only as good as the building blocks used to create it. Our technical team has extensively analyzed the advancements presented in CN112094204B and integrated similar high-efficiency, safety-first protocols into our own manufacturing platforms. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on the stringent purity specifications required for GMP-grade peptide synthesis. Our rigorous QC labs employ state-of-the-art analytical techniques to verify the absence of critical impurities, guaranteeing that every batch of Fmoc-Tyr(tBu)-OH delivers the performance your R&D teams expect.
We invite you to collaborate with us to optimize your supply chain for peptide intermediates. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are prepared to provide specific COA data from recent batches and conduct comprehensive route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your long-term production goals securely and efficiently.
