Scalable Production of High-Purity m-Hydroxyphenylacetic Acid via Novel Protective Group Strategy
Scalable Production of High-Purity m-Hydroxyphenylacetic Acid via Novel Protective Group Strategy
The pharmaceutical industry continuously demands intermediates with exceptional purity profiles to ensure the safety and efficacy of final drug substances. A recent technological breakthrough documented in patent CN116283541A introduces a robust and scalable preparation method for high-purity m-hydroxyphenylacetic acid, a critical building block in medicinal chemistry. This innovative approach addresses long-standing challenges regarding impurity control and process safety that have plagued conventional synthesis routes. By implementing a strategic phenolic protection group combined with a unique ester-based purification protocol, the method achieves an HPLC purity exceeding 99% with single impurities controlled below 0.1%. This technical advancement represents a significant leap forward for manufacturers seeking a reliable m-hydroxyphenylacetic acid supplier capable of meeting stringent regulatory standards.
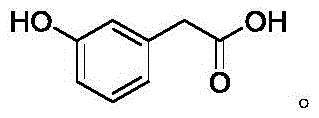
The structural integrity of m-hydroxyphenylacetic acid is paramount for its downstream applications, yet achieving high purity has historically been problematic due to the reactivity of the phenolic moiety. The patented process begins with the inexpensive and readily available starting material, m-hydroxyacetophenone, effectively bypassing the need for costly halogenated precursors used in older methodologies. The core innovation lies in the temporary masking of the reactive phenolic hydroxyl group, which prevents unwanted side reactions during the subsequent carbon-chain extension steps. This strategic modification not only simplifies the reaction profile but also facilitates a purification pathway that was previously unattainable through direct crystallization of the free acid. For procurement managers and supply chain directors, this translates to a more predictable production timeline and reduced risk of batch rejection due to out-of-specification impurity levels.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of m-hydroxyphenylacetic acid has relied on pathways fraught with significant operational hazards and economic inefficiencies. One prominent prior art method utilizes 3-iodophenylacetic acid as a starting material, which is prohibitively expensive and limits the economic viability of large-scale production. Furthermore, this route requires specialized catalysts like monomethoxycyclohexanepentaol, which are difficult to source commercially and challenging to synthesize in-house, creating a fragile supply chain bottleneck. Another common approach involves the use of m-hydroxybenzaldehyde reacted with sodium cyanide under cryogenic conditions of -78°C. This method poses severe safety risks due to the handling of highly toxic cyanide salts and incurs substantial energy costs to maintain deep-freeze temperatures, making it unsuitable for modern green chemistry initiatives. Additionally, direct Willgerodt-Kindler reactions on unprotected m-hydroxyacetophenone have been verified to yield products with poor purity profiles, where single impurities cannot be controlled below the critical 0.1% threshold required for high-grade pharmaceutical intermediates.
The Novel Approach
In stark contrast to these legacy methods, the novel process outlined in CN116283541A employs a sophisticated five-step sequence that prioritizes both safety and purity. The journey begins with the nucleophilic substitution of the phenolic hydroxyl group using chloromethyl methyl ether to form a stable MOM-ether protected intermediate. This protected ketone then undergoes a Willgerodt-Kindler reaction with sulfur powder and morpholine to generate a thione derivative, effectively extending the carbon chain without degrading the aromatic ring. Following alkaline hydrolysis to reveal the carboxylic acid functionality, the process diverges from tradition by converting the crude acid into a methyl ester. This esterification step is the key enabler for the subsequent rectification purification, allowing for the removal of trace impurities based on boiling point differences rather than solubility. Finally, the high-purity ester is hydrolyzed back to the target acid, ensuring a final product that meets the rigorous demands of the medical intermediate industry.
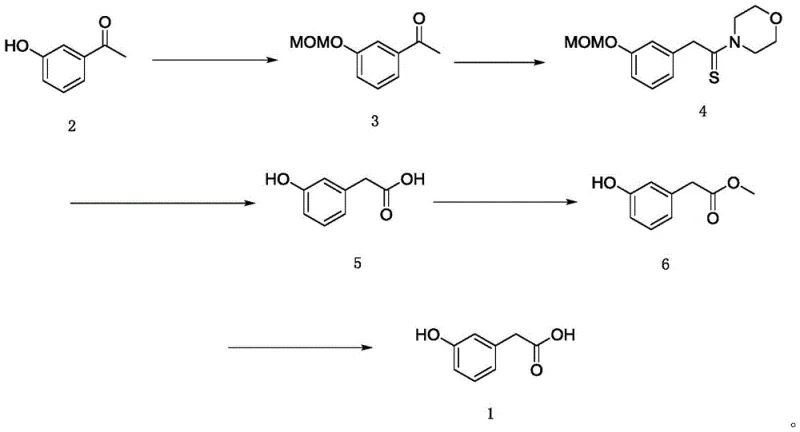
Mechanistic Insights into the Protective Group Strategy and Purification
The success of this synthesis hinges on the precise manipulation of functional group reactivity through the use of the methoxymethyl (MOM) protecting group. In the initial step, the phenolic oxygen acts as a nucleophile, attacking the chloromethyl methyl ether in the presence of a strong base such as potassium tert-butoxide or sodium hydride. This transformation converts the acidic and reactive phenol into a chemically inert ether, which is stable under the vigorous conditions of the subsequent Willgerodt-Kindler reaction. Without this protection, the free phenol would likely participate in side reactions with sulfur or morpholine, or undergo oxidation, leading to a complex mixture of byproducts that are structurally similar to the target molecule and notoriously difficult to separate. The stability of the MOM group ensures that the reaction proceeds selectively at the acetyl group, preserving the integrity of the aromatic substitution pattern throughout the chain extension process.
Perhaps the most critical mechanistic insight for R&D directors is the purification strategy employed in the latter stages of the synthesis. Traditional methods often attempt to purify the free acid directly via crystallization, but the polarity and hydrogen bonding capability of the carboxylic acid and phenol groups often trap impurities within the crystal lattice. By converting the crude acid into methyl m-hydroxyphenylacetate, the molecule becomes more volatile and less polar, enabling purification via fractional distillation or rectification. This physical separation method is far more effective at removing non-volatile tars and closely related organic impurities than recrystallization. Once the ester is distilled to high purity (HPLC > 99.4%), the final hydrolysis step simply cleaves the ester bond and removes the MOM group (often concurrently or in a dedicated step depending on conditions), yielding the final high-purity acid. This "purify-as-ester" tactic is a powerful tool for cost reduction in pharmaceutical intermediate manufacturing, as it minimizes yield loss associated with multiple recrystallizations.
How to Synthesize m-Hydroxyphenylacetic Acid Efficiently
The execution of this synthesis requires careful attention to reaction parameters, particularly temperature control and stoichiometry during the protection and thione formation steps. The process is designed to be telescoped where possible, with the intermediate MOM-protected ketone often used directly in the next step without isolation to maximize throughput. Detailed standard operating procedures for each transformation, including specific solvent choices like DMF or dioxane and precise molar ratios of reagents, are essential for replicating the high purity results reported in the patent. Operators must ensure complete conversion at each stage, particularly the hydrolysis of the thione, to prevent carryover of sulfur-containing impurities into the final product. For a comprehensive guide on the specific reaction conditions, workup procedures, and quality control checkpoints, please refer to the standardized synthesis protocol below.
- Protect the phenolic hydroxyl group of m-hydroxyacetophenone using chloromethyl methyl ether and a base to form the MOM-ether intermediate.
- Perform a Willgerodt-Kindler reaction with sulfur and morpholine to convert the ketone into a thioamide derivative.
- Hydrolyze the thioamide to crude acid, esterify to methyl ester, purify via rectification, and finally hydrolyze to the target high-purity acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers substantial advantages that directly impact the bottom line and supply chain resilience for global buyers. The shift away from exotic starting materials like 3-iodophenylacetic acid to commodity chemicals like m-hydroxyacetophenone drastically reduces the raw material cost base. Furthermore, the elimination of cryogenic requirements and toxic cyanide reagents lowers the barrier to entry for manufacturing partners, expanding the pool of qualified suppliers and enhancing supply security. The robustness of the process allows for significant scale-up potential, moving from kilogram-level laboratory batches to multi-ton annual commercial production without the need for specialized low-temperature infrastructure. This scalability ensures that procurement managers can secure long-term contracts with confidence, knowing that the supply of this critical intermediate will remain continuous and stable even during periods of high market demand.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven primarily by the substitution of expensive reagents with cheap, bulk-available commodities. By avoiding the use of precious metal catalysts or difficult-to-source organometallic reagents, the overall cost of goods sold is significantly lowered. Additionally, the ability to purify via rectification of the ester intermediate reduces the consumption of large volumes of crystallization solvents and minimizes product loss typically associated with mother liquor disposal. The mild reaction temperatures (60°C to 100°C) also result in lower energy consumption compared to processes requiring deep freezing or high-pressure hydrogenation, contributing to a leaner and more cost-effective manufacturing operation.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of raw materials that are widely produced and stocked by major chemical distributors globally. Unlike routes dependent on single-source catalysts or hazardous materials that face strict transportation regulations, the reagents in this process (sulfur, morpholine, chloromethyl methyl ether) are standard industrial chemicals. This ubiquity reduces the risk of supply disruptions caused by geopolitical issues or regulatory crackdowns on specific precursor chemicals. Moreover, the simplified safety profile of the plant operations means that fewer specialized permits are required, allowing for faster ramp-up of production capacity at CDMO facilities to meet urgent customer needs.
- Scalability and Environmental Compliance: The environmental footprint of this synthesis is markedly smaller than that of cyanide-based alternatives, aligning with the increasing pressure on pharmaceutical companies to adopt greener supply chains. The absence of heavy metals and highly toxic reagents simplifies waste stream treatment and reduces the cost of environmental compliance and disposal. The process is inherently safer, reducing the risk of catastrophic accidents and ensuring business continuity. From a scalability standpoint, the unit operations involved—stirred tank reactions, distillation, and filtration—are standard across the fine chemical industry, meaning that technology transfer to large-scale reactors is straightforward and low-risk, facilitating rapid commercialization.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this high-purity intermediate. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, offering clarity on the process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this material into their own synthesis pipelines.
Q: Why is the ester rectification step critical in this synthesis?
A: Direct crystallization of the crude acid often fails to remove structurally similar impurities below 0.1%. Converting the acid to a volatile methyl ester allows for high-efficiency rectification, removing trace impurities before final hydrolysis ensures >99% HPLC purity.
Q: How does this method improve safety compared to traditional routes?
A: Unlike prior art methods requiring highly toxic sodium cyanide or cryogenic conditions (-78°C), this process utilizes mild temperatures (60-100°C) and avoids剧毒 reagents, significantly reducing operational risk and waste treatment costs.
Q: What is the role of the MOM protecting group?
A: The methoxymethyl (MOM) group protects the phenolic hydroxyl during the harsh Willgerodt-Kindler reaction conditions. Without this protection, side reactions occur on the phenol ring, leading to complex impurity profiles that are difficult to separate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable m-Hydroxyphenylacetic acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving medications. Our team of expert chemists has extensively analyzed the pathway described in CN116283541A and possesses the technical capability to implement this advanced synthesis route immediately. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of m-hydroxyphenylacetic acid we deliver meets the highest international standards for pharmaceutical use.
We invite you to collaborate with us to optimize your supply chain for this essential building block. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our implementation of this novel route can reduce your overall project costs. Please contact us today to request specific COA data from our recent pilot batches and to discuss route feasibility assessments for your upcoming projects. Let us be your partner in driving efficiency and quality in your pharmaceutical manufacturing operations.
