Advanced Crystallization Technology for High-Purity Biapenem Condensation Compound Manufacturing
Advanced Crystallization Technology for High-Purity Biapenem Condensation Compound Manufacturing
The global demand for next-generation carbapenem antibiotics continues to surge, driven by the critical need for effective treatments against resistant bacterial strains. At the forefront of this therapeutic class is Biapenem, a potent agent known for its stability against dehydropeptidase-I (DHP-I) and broad-spectrum activity. However, the commercial viability of such complex molecules relies heavily on the efficiency of their synthetic intermediates. Patent CN101747352B introduces a transformative preparation method for the biapenem condensation compound, specifically targeting the optimization of crystal quality and purity. This technical breakthrough addresses long-standing challenges in the downstream processing of carbapenem intermediates, offering a pathway to more robust and cost-effective manufacturing. The core innovation lies in the strategic manipulation of the crystallization environment to simultaneously drive the coupling reaction and purify the resulting solid. 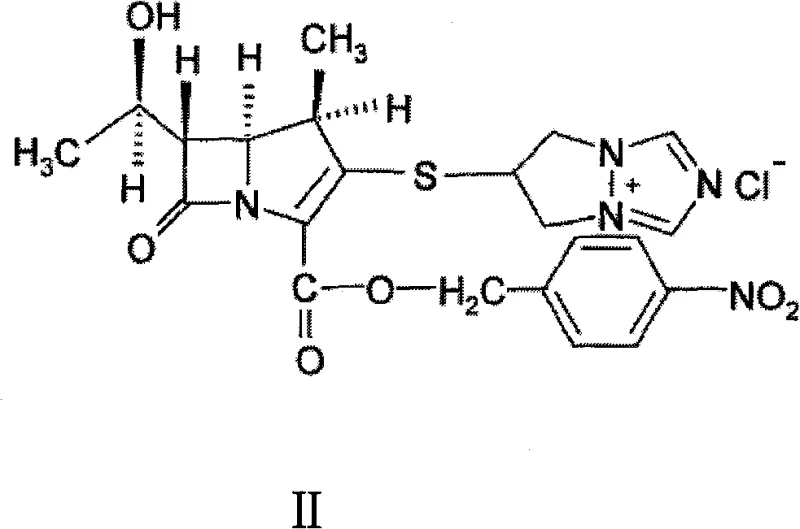
For procurement specialists and supply chain directors, the implications of this technology extend far beyond the laboratory bench. By enhancing the physical properties of the intermediate—specifically particle size and filterability—the process directly impacts production throughput and operational expenditure. As a reliable pharmaceutical intermediates supplier, understanding these nuanced process improvements is essential for securing a stable supply of high-quality active pharmaceutical ingredients (APIs). This report delves into the mechanistic underpinnings and commercial advantages of this novel crystallization technique, providing a comprehensive analysis for industry decision-makers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the biapenem condensation compound has been fraught with processing difficulties that hinder large-scale production. Conventional methodologies, such as those reported by Toshio Kumagai and later adapted by Liu Xiangkui, typically rely on complex solvent systems involving mixtures of acetonitrile, acetone, and dimethylformamide (DMF), or strictly anhydrous acetonitrile. A significant bottleneck in these traditional routes is the stringent requirement for starting material purity; the phosphate ester precursor (Compound III) and the thiol salt (Compound IV) must often exceed 98% purity to achieve acceptable yields and product quality. 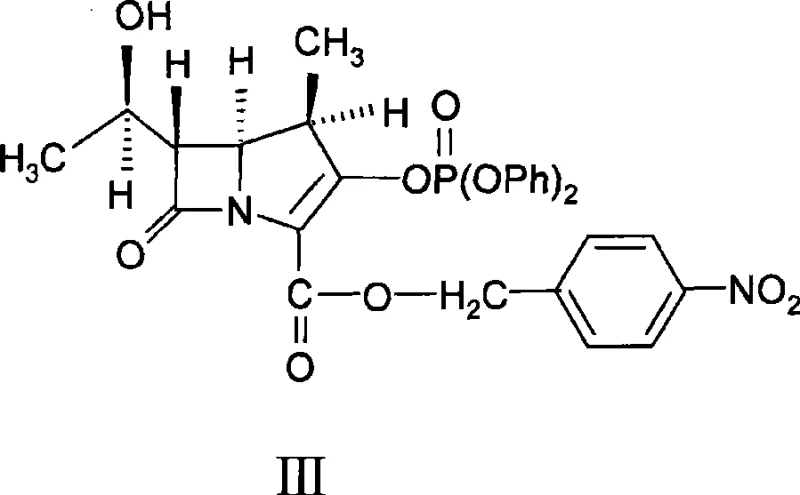
Furthermore, the crystals obtained from these legacy processes often exhibit poor morphological characteristics, such as fine particle size or needle-like habits, which create substantial resistance during filtration and washing steps. This leads to prolonged cycle times, increased solvent retention, and higher energy consumption during drying. The inability to effectively purge impurities during the crystallization step necessitates additional downstream purification stages, such as recrystallization or column chromatography, which further erodes profit margins and complicates the supply chain. These inefficiencies represent a critical vulnerability in the manufacturing of cost reduction in API manufacturing contexts.
The Novel Approach
The methodology disclosed in patent CN101747352B represents a paradigm shift by integrating a lower alcohol co-solvent directly into the reaction-crystallization system. Instead of relying solely on acetonitrile or complex ternary mixtures, this innovative approach introduces solvents like ethanol, methanol, or isopropanol to the reaction matrix. This modification serves a dual purpose: it facilitates the nucleophilic substitution reaction between the phosphate ester and the thiol salt while simultaneously acting as an anti-solvent to control crystal nucleation and growth. The result is a process that is remarkably tolerant to minor variations in starting material quality, as the crystallization itself acts as a purification barrier.
By carefully tuning the ratio of acetonitrile to the lower alcohol, manufacturers can induce the formation of larger, more uniform crystals. This improvement in crystal habit is not merely aesthetic; it translates directly to enhanced processability. The larger particles settle faster and form more permeable cakes during filtration, significantly reducing the time required for solid-liquid separation. Moreover, the method effectively sequesters impurities within the mother liquor, ensuring that the isolated solid meets rigorous purity specifications without the need for extensive reprocessing. This streamlined workflow exemplifies the potential for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Alcohol-Mediated Crystallization
To fully appreciate the value of this technology, one must examine the physicochemical interactions occurring at the molecular level. The reaction involves the nucleophilic attack of the sulfur atom in the triazole-thiol salt (Compound IV) upon the activated carbon of the phosphate ester (Compound III). In traditional acetonitrile-only systems, the solvation shell around the reacting species can lead to rapid, uncontrolled nucleation, resulting in a multitude of small crystals that trap solvent and impurities. The introduction of a lower alcohol disrupts this dynamic by altering the dielectric constant and hydrogen-bonding network of the solvent medium. This change in solvation energy modulates the supersaturation profile, favoring crystal growth over nucleation.
From an impurity control perspective, the lower alcohol functions as a selective solubilizer. Many organic byproducts and unreacted starting materials possess different solubility profiles in alcohol-acetonitrile mixtures compared to pure acetonitrile. As the target biapenem condensation compound precipitates out of the solution, these impurities remain dissolved in the supernatant. This phenomenon, known as 'purification by crystallization,' is critical for achieving the high-purity biapenem intermediate standards required for regulatory approval. The patent data confirms that regardless of whether methanol, ethanol, or isopropanol is used, the resulting crystal lattice remains consistent, as evidenced by identical powder X-ray diffraction (PXRD) patterns, ensuring batch-to-batch reproducibility.
How to Synthesize Biapenem Condensation Compound Efficiently
Implementing this advanced crystallization protocol requires precise control over reaction parameters to maximize yield and crystal quality. The process begins with the dissolution of the key precursors in anhydrous acetonitrile, followed by the critical addition of the lower alcohol co-solvent. Temperature management is paramount; the reaction must be maintained at cryogenic conditions (0-5°C) to prevent degradation of the sensitive beta-lactam ring and to control the rate of precipitation. The following guide outlines the standardized operational procedure derived from the patent embodiments, designed to ensure optimal outcomes for process chemists.
- Dissolve the phosphate ester precursor (Compound III) and the thiol salt (Compound IV) in anhydrous acetonitrile within a reactor vessel.
- Introduce a lower alcohol solvent, such as ethanol or methanol, into the reaction mixture to modify the solubility profile and crystal growth kinetics.
- Cool the mixture to approximately 0°C, add diisopropylethylamine (DIPEA) as the base, and maintain the temperature between 0-5°C for 2 to 5 hours to complete the coupling and crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and operational continuity, the adoption of this crystallization technology offers compelling strategic benefits. The primary advantage lies in the drastic simplification of the isolation process. By generating crystals with superior filtration characteristics, manufacturers can significantly reduce the downtime associated with centrifugation or filtering operations. This increase in equipment turnover rate allows for higher production volumes within the same timeframe, effectively expanding capacity without capital investment in new hardware. Furthermore, the reduction in solvent retention minimizes the energy load required for drying, contributing to a lower carbon footprint and reduced utility costs.
- Cost Reduction in Manufacturing: The elimination of extensive downstream purification steps results in substantial cost savings. Because the crystallization process itself removes impurities, the need for resource-intensive recrystallization or chromatographic purification is minimized or entirely removed. Additionally, the tolerance for slightly lower purity starting materials reduces the procurement costs of raw materials, as expensive high-grade precursors are no longer a strict prerequisite for success. This holistic reduction in processing complexity translates directly to a more competitive cost structure for the final intermediate.
- Enhanced Supply Chain Reliability: Robustness is the cornerstone of a resilient supply chain. This method's ability to consistently produce high-quality crystals across a range of lower alcohol solvents provides flexibility in raw material sourcing. If ethanol supply is constrained, the process can seamlessly switch to methanol or isopropanol without compromising product quality or crystal form. This adaptability mitigates the risk of production stoppages due to solvent shortages, ensuring a steady flow of materials to downstream API synthesis units and reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces unforeseen challenges, particularly regarding heat transfer and mixing efficiency. However, the improved physical properties of the crystals generated by this method facilitate easier handling at the multi-kilogram and tonne scales. The larger particle size reduces the risk of filter clogging and dust generation, enhancing operator safety. Moreover, the use of common, lower-toxicity alcohols like ethanol aligns with green chemistry principles, simplifying waste stream management and ensuring compliance with increasingly stringent environmental regulations regarding solvent emissions and disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and validation of this crystallization technology. These insights are derived directly from the experimental data and claims presented in the patent literature, aimed at clarifying the operational nuances for technical teams evaluating this route for adoption.
Q: How does the addition of lower alcohol improve the purity of the biapenem intermediate?
A: The inclusion of a lower alcohol solvent, such as ethanol, acts as a crystallization modifier that selectively precipitates the target compound while keeping specific organic impurities in solution, effectively purifying the product during the formation of the crystal lattice.
Q: What are the physical advantages of the crystals produced by this method?
A: This novel process yields crystals with significantly larger particle sizes compared to conventional methods, which drastically improves solid-liquid separation rates and reduces drying times, making it highly suitable for industrial manufacturing.
Q: Is this method compatible with various lower alcohol solvents?
A: Yes, the patent data indicates that the process is robust across a range of lower alcohols including methanol, ethanol, n-propyl alcohol, isopropanol, and tert-butyl alcohol, all yielding high-purity products with consistent crystal forms.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Biapenem Condensation Compound Supplier
The transition from laboratory innovation to industrial reality requires a partner with deep technical expertise and proven manufacturing capabilities. NINGBO INNO PHARMCHEM stands at the intersection of scientific rigor and commercial execution, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with state-of-the-art cryogenic reactors and precision filtration systems capable of executing the delicate low-temperature crystallization protocols required for carbapenem intermediates. We adhere to stringent purity specifications and operate rigorous QC labs to ensure that every batch of biapenem condensation compound meets the exacting standards necessary for global pharmaceutical registration.
We invite procurement leaders and R&D directors to collaborate with us to leverage this advanced crystallization technology for your supply chain. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, ensuring that your project benefits from the highest levels of quality, efficiency, and supply security available in the market.
