Advanced Synthesis of 4,4'-Diaminodiphenyl Ether for High-Performance Polymer Applications
The global demand for high-performance polyimide resins and advanced epoxy systems continues to drive the need for efficient, scalable production methods for key monomers like 4,4'-diaminodiphenyl ether (ODA). Patent CN109180501B introduces a transformative synthetic methodology that addresses critical bottlenecks in traditional manufacturing, specifically focusing on safety, cost-efficiency, and environmental sustainability. This technical disclosure outlines a novel two-step sequence involving an aqueous nitrosation reaction followed by a catalytic hydrogenation reduction. Unlike conventional routes that rely on harsh nitrating agents or high-temperature condensations, this approach leverages the unique reactivity of nitroso intermediates to streamline the supply chain for this vital fine chemical intermediate. For R&D directors and procurement strategists, understanding the mechanistic nuances of this patent is essential for evaluating potential technology transfers or sourcing partnerships that offer a competitive edge in the polymer and pharmaceutical sectors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 4,4'-diaminodiphenyl ether has been plagued by significant operational hazards and environmental liabilities inherent to established chemical pathways. The traditional p-dinitrobenzene method involves volatile and highly toxic raw materials that pose severe risks to worker safety and require expensive containment infrastructure. Alternatively, the direct nitration of diphenyl ether typically necessitates the use of concentrated sulfuric acid and nitric acid mixtures, which are extremely corrosive and generate vast quantities of acidic wastewater that are costly to treat and dispose of. Furthermore, condensation methods involving p-nitrochlorobenzene often demand rigorous equipment specifications to withstand high temperatures and pressures, while simultaneously producing complex byproduct profiles that complicate downstream purification. These legacy processes not only inflate the cost of goods sold through excessive utility and waste management consumption but also create supply chain vulnerabilities due to increasingly stringent regulatory scrutiny on hazardous chemical handling and emissions.
The Novel Approach
In stark contrast to these cumbersome legacy protocols, the methodology disclosed in CN109180501B presents a streamlined, greener alternative that fundamentally alters the reaction landscape for ODA production. By shifting the initial functionalization step to a nitrosation reaction conducted entirely within a water phase, the process eliminates the need for organic solvents during the critical bond-forming stage, thereby drastically reducing fire hazards and solvent recovery costs. The subsequent reduction of the dinitroso intermediate to the diamine is achieved under relatively mild hydrogenation conditions, capitalizing on the favorable stoichiometry of nitroso group reduction which requires only half the molar equivalent of hydrogen compared to nitro group reduction. This strategic shift not only lowers the operating pressure requirements for the hydrogenation reactor—enhancing overall plant safety—but also simplifies the isolation of the intermediate through straightforward crystallization and filtration, ensuring a robust and scalable pathway suitable for modern commercial manufacturing environments.
Mechanistic Insights into Aqueous Nitrosation and Catalytic Hydrogenation
The core innovation of this synthesis lies in the electrophilic aromatic substitution mechanism facilitated by the in situ generation of nitrosonium ions within an acidic aqueous medium. In the first stage, sodium nitrite reacts with hydrochloric acid to generate the active nitrosating species, which then attacks the electron-rich aromatic rings of diphenyl ether at the para positions. This reaction is highly selective and proceeds efficiently at low temperatures ranging from 0°C to 5°C, minimizing side reactions such as oxidation or polysubstitution that often plague higher-temperature processes. The resulting 4,4'-dinitroso diphenyl ether precipitates out of the aqueous solution as needle-shaped crystals, a physical property that is exploited for purification without the need for energy-intensive distillation or chromatography. This phase separation behavior is critical for maintaining high purity levels early in the synthesis, effectively acting as a built-in purification step that reduces the impurity load carried forward into the final reduction stage.
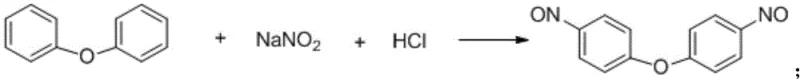
Following the isolation of the dinitroso intermediate, the process transitions to a catalytic hydrogenation phase where the nitroso groups are reduced to primary amines. This transformation is thermodynamically favorable and kinetically accessible using standard heterogeneous catalysts such as Raney nickel, palladium on carbon, or platinum on carbon suspended in an alcohol solvent like ethanol. The reduction mechanism involves the adsorption of hydrogen gas onto the catalyst surface, followed by the sequential addition of hydrogen atoms to the nitrogen-oxygen double bonds of the nitroso groups. Crucially, because the starting material contains nitroso (-NO) groups rather than nitro (-NO2) groups, the stoichiometric demand for hydrogen is significantly reduced, allowing the reaction to proceed effectively at lower pressures between 0.5 MPa and 4 MPa. This mechanistic efficiency translates directly into operational savings, as the lower pressure requirements reduce the capital expenditure on high-pressure vessels and lower the ongoing energy costs associated with hydrogen compression and recycling.
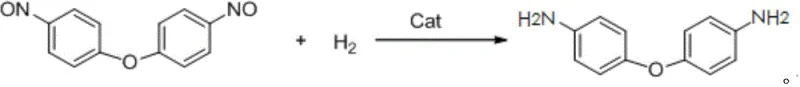
From an impurity control perspective, this two-step sequence offers distinct advantages over direct nitration routes which often suffer from the formation of ortho-isomers and polynitrated byproducts that are difficult to separate. The specificity of the nitrosation reaction under controlled acidic conditions favors the para-substitution pattern required for the target polymer-grade monomer. Furthermore, the crystallization of the dinitroso intermediate serves as a powerful purification gate; any unreacted diphenyl ether or soluble inorganic salts remain in the mother liquor and are removed prior to the reduction step. This ensures that the final hydrogenation reaction starts with a high-purity substrate, minimizing the formation of complex amine byproducts and simplifying the final work-up. The result is a product profile that meets the stringent purity specifications demanded by the electronics and aerospace industries, where trace impurities can critically degrade the thermal and mechanical properties of the resulting polyimide films.
How to Synthesize 4,4'-Diaminodiphenyl Ether Efficiently
To implement this synthesis effectively in a pilot or commercial setting, operators must adhere to precise temperature controls and stoichiometric ratios as defined in the patent embodiments. The process begins with the careful preparation of the aqueous nitrite solution and its slow addition to the diphenyl ether slurry under vigorous stirring to maintain the exothermic reaction within the narrow 0-5°C window. Once the intermediate is isolated and dried, it is transferred to a hydrogenation vessel where the choice of catalyst and solvent plays a pivotal role in achieving the reported yields of over 90%. Detailed standard operating procedures regarding mixing speeds, gas flow rates, and safety interlocks are essential for replicating the success of this method.
- Perform nitrosation by reacting diphenyl ether with sodium nitrite and hydrochloric acid in a water phase at 0-5°C to form 4,4'-dinitroso diphenyl ether.
- Filter and dry the resulting crystals under vacuum to isolate the intermediate dinitroso compound.
- Conduct catalytic hydrogenation in an autoclave using alcohol solvent and Raney nickel catalyst at 0.5-4MPa and 70-120°C to yield the final diamine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this nitrosation-based synthesis route represents a strategic opportunity to optimize the cost structure and reliability of 4,4'-diaminodiphenyl ether sourcing. The shift away from corrosive mixed-acid nitration eliminates the logistical burdens and costs associated with handling, storing, and disposing of large volumes of concentrated sulfuric acid, which is a significant hidden cost in traditional manufacturing. Additionally, the use of water as the primary reaction medium for the first step removes the dependency on volatile organic solvents, mitigating fire risks and reducing the need for complex solvent recovery systems. These operational simplifications translate into a more resilient supply chain that is less susceptible to regulatory disruptions and raw material price volatility, ensuring a steady flow of high-quality intermediates for downstream polymer production.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the superior atom economy of the reduction step and the elimination of expensive solvent systems. Since the reduction of nitroso groups consumes only half the amount of hydrogen required for nitro groups, the operational costs related to hydrogen gas procurement and compression are substantially decreased. Furthermore, the avoidance of organic solvents in the nitrosation step and the use of common alcohols like ethanol in the reduction step significantly lower raw material expenses. The simplified purification via crystallization also reduces energy consumption compared to distillation-heavy processes, leading to a leaner, more cost-effective production model that enhances margin potential for bulk buyers.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including diphenyl ether, sodium nitrite, and hydrochloric acid, are commodity chemicals with robust, global supply chains that are less prone to disruption than specialized nitrating agents. The moderate reaction conditions, particularly the lower pressure requirements for hydrogenation, allow for production in a wider range of facilities without the need for specialized high-pressure infrastructure. This flexibility increases the pool of qualified manufacturers capable of producing this intermediate, thereby reducing the risk of single-source dependency and shortening lead times for customers seeking reliable long-term supply agreements for their polymer or pharmaceutical projects.
- Scalability and Environmental Compliance: As environmental regulations tighten globally, the ability to demonstrate a reduced environmental footprint is a critical competitive advantage. This process generates significantly less acidic wastewater and hazardous waste compared to traditional nitration methods, simplifying compliance with local environmental protection laws. The water-based nature of the first step and the use of recyclable alcohol solvents in the second step align with green chemistry principles, making the scale-up from pilot to commercial tonnage smoother and less likely to face permitting delays. This sustainability profile not only future-proofs the supply chain against regulatory changes but also appeals to end-users in the electronics and automotive sectors who are increasingly prioritizing eco-friendly material sourcing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this specific synthetic route for 4,4'-diaminodiphenyl ether. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the technology's viability. Understanding these details is crucial for technical teams assessing the feasibility of adopting this method for their own production lines or for procurement specialists validating supplier capabilities.
Q: What are the primary advantages of the nitrosation route over traditional nitration for ODA synthesis?
A: The nitrosation route described in patent CN109180501B operates in a water phase, eliminating the need for large volumes of corrosive concentrated sulfuric acid used in traditional nitration. Furthermore, the stoichiometric requirement for hydrogen in the reduction step is significantly lower (1:1 molar ratio for nitroso vs 1:2 for nitro groups), leading to reduced reaction pressure and enhanced safety profiles.
Q: What catalysts are suitable for the reduction step in this specific synthesis method?
A: The patent specifies the use of heterogeneous catalysts such as Raney nickel, palladium on carbon (Pd/C), or platinum on carbon (Pt/C). In the preferred embodiment, Raney nickel is utilized effectively in an ethanol solvent system to achieve high conversion rates under moderate hydrogen pressure.
Q: How does this method impact environmental compliance and waste generation?
A: By utilizing a water-phase nitrosation reaction, the process avoids the generation of massive amounts of acidic wastewater associated with mixed-acid nitration. The simplified work-up procedure involving filtration and vacuum drying minimizes solvent usage and reduces the overall three-waste burden, aligning with stricter modern environmental regulations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,4'-Diaminodiphenyl Ether Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity 4,4'-diaminodiphenyl ether plays in the manufacture of advanced polyimides and specialty polymers. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of order volume. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch meets the exacting standards required for high-performance applications in the aerospace, electronics, and pharmaceutical industries.
We invite you to collaborate with us to leverage this innovative synthesis technology for your specific project needs. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this efficient route can optimize your overall budget. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you secure a sustainable, cost-effective supply of this essential chemical intermediate.
