Advanced Synthesis of o-Amino Benzotrifluoride: A Cost-Effective Route for Fluorine Chemistry Manufacturing
The global demand for fluorine-containing intermediates has surged in recent years, driven by their critical role in the development of high-performance agrochemicals and pharmaceuticals. Patent CN110668955A introduces a strategically optimized synthesis method for o-amino benzotrifluoride, a vital building block in the production of herbicides and specialty dyes. This innovative approach streamlines the manufacturing process into two distinct stages: a controlled nitration followed by a catalytic hydrogenation reduction. By utilizing benzotrifluoride as the primary raw material, the method leverages widely available feedstocks to ensure supply chain stability. The technical breakthrough lies not only in the chemical transformation but also in the operational parameters that prioritize safety and economic efficiency. For R&D directors and procurement managers alike, understanding this pathway offers a glimpse into the future of sustainable fine chemical manufacturing, where cost reduction and environmental compliance are achieved simultaneously through intelligent process design.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of o-amino benzotrifluoride has been plagued by significant technical and economic hurdles that hinder scalable manufacturing. Traditional routes often rely on the separation of ortho-isomers from complex mixtures generated during the nitration of benzotrifluoride, where the strong meta-directing effect of the trifluoromethyl group results in a predominance of the unwanted meta-isomer, leaving the ortho-content at merely around 10 percent. Alternative pathways, such as the ammonolysis of ortho-chlorotrifluoromethane, necessitate the use of expensive catalysts like cobalt or copper complexes and require harsh reaction conditions that accelerate equipment corrosion. Furthermore, routes involving aniline trifluoromethylation or the use of meta-bromotrifluorotoluene introduce additional steps, such as de-acetylation or debromination, which drastically increase the consumption of solvents and reagents. These multi-step processes not only inflate the overall production cost but also generate substantial amounts of hazardous waste, creating a heavy burden on environmental treatment facilities and complicating regulatory compliance for manufacturers.
The Novel Approach
In stark contrast to these cumbersome legacy methods, the novel synthesis route detailed in the patent simplifies the production landscape by focusing on direct transformation and efficient catalysis. This approach bypasses the need for exotic starting materials or complex protection-deprotection sequences, instead opting for a direct nitration of benzotrifluoride followed by a robust reduction step. The key differentiator is the implementation of mild reaction conditions, specifically maintaining the nitration phase at normal pressure and a low temperature range of 35-45°C. This moderation of thermal energy input significantly mitigates the corrosive impact of mixed acids on reactor vessels and piping systems, thereby extending equipment lifespan and reducing maintenance downtime. Moreover, the substitution of precious metal catalysts with economical nickel-based systems in the reduction phase represents a paradigm shift in cost management, allowing for high-yield production without the financial penalty associated with palladium or platinum recovery.
Mechanistic Insights into Mixed Acid Nitration and Nickel-Catalyzed Hydrogenation
The first stage of this synthesis involves an electrophilic aromatic substitution where benzotrifluoride reacts with a carefully prepared mixed acid system. The generation of the nitronium ion (NO2+) is facilitated by the interaction between nitric acid and sulfuric acid, with the patent specifying a mass ratio of approximately 1:1.7 to 1.8 to optimize the concentration of the active electrophile. Despite the deactivating nature of the trifluoromethyl group, the reaction proceeds effectively under controlled thermal conditions to yield a mixture of nitrobenzotrifluoride isomers. The mechanistic control here is crucial; by limiting the temperature to below 45°C, the process minimizes side reactions such as oxidation or polynitration, which could otherwise lead to difficult-to-remove impurities. The resulting organic layer contains the necessary nitro-intermediate, which is then isolated through phase separation and washing, ensuring that residual acids do not interfere with the subsequent reduction catalyst.
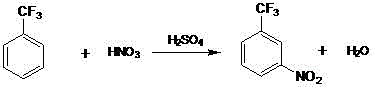
Following nitration, the process transitions to a catalytic hydrogenation reduction, a critical step where the nitro group is converted into the desired amino functionality. This transformation is mediated by a nickel catalyst suspended in methanol, operating under a hydrogen pressure of 2.5-4.0 MPa and temperatures between 100-115°C. The nickel surface facilitates the adsorption and dissociation of hydrogen molecules, which then transfer to the nitrogen-oxygen bonds of the nitro compound in a stepwise reduction mechanism. This heterogeneous catalysis is highly advantageous because nickel is far more abundant and less susceptible to poisoning by sulfur or other trace impurities compared to noble metals. The use of methanol as a solvent further enhances the reaction kinetics due to its ability to dissolve both the organic substrate and the gaseous hydrogen, creating a homogeneous reaction environment around the solid catalyst particles. Upon completion, the solvent is recovered via distillation, and the crude amine mixture is subjected to vacuum rectification to isolate the pure o-amino benzotrifluoride based on boiling point differences.
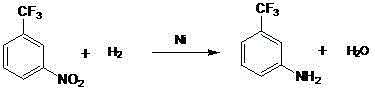
How to Synthesize o-Amino Benzotrifluoride Efficiently
Executing this synthesis requires precise adherence to the specified stoichiometric ratios and thermal profiles to maximize the yield of the ortho-isomer while ensuring operator safety. The process begins with the meticulous preparation of the mixed acid, followed by the dropwise addition of benzotrifluoride to manage the exotherm effectively. Once the nitro-intermediate is secured, the reduction phase demands rigorous exclusion of oxygen through nitrogen purging before introducing hydrogen gas to prevent explosive hazards. The final purification via vacuum rectification is the defining step for achieving the high purity required for downstream pharmaceutical or agrochemical applications. For a comprehensive breakdown of the specific operational parameters, reagent quantities, and safety protocols, please refer to the standardized synthesis guide provided below.
- Prepare mixed acid with a nitric to sulfuric acid ratio of 1: 1.7 to 1.8 and react with benzotrifluoride at 35-45°C under normal pressure to form nitrobenzotrifluoride.
- Perform catalytic hydrogenation on the nitrobenzotrifluoride mixture using methanol as a solvent and a nickel catalyst at 100-115°C and 2.5-4.0 MPa pressure.
- Recover methanol solvent via distillation and separate the final o-amino benzotrifluoride product from meta and para isomers through vacuum rectification.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers profound advantages that directly address the pain points of modern chemical procurement and supply chain management. The shift away from noble metal catalysts to nickel represents a structural change in the cost base of the molecule, eliminating the volatility associated with precious metal markets and reducing the capital tied up in catalyst inventory. Furthermore, the ability to recycle methanol solvent and recover sulfuric acid from the waste stream creates a closed-loop system that minimizes raw material consumption and waste disposal fees. For supply chain heads, the reliance on benzotrifluoride, a commodity chemical with a robust global supply network, ensures that production is not bottlenecked by the scarcity of specialized precursors. This stability allows for consistent lead times and reliable fulfillment of large-volume contracts, which is essential for maintaining the continuity of downstream drug or pesticide manufacturing lines.
- Cost Reduction in Manufacturing: The replacement of expensive palladium or platinum catalysts with economical nickel significantly lowers the variable cost per kilogram of the final product. This substitution eliminates the need for complex and costly precious metal recovery units, thereby simplifying the plant infrastructure and reducing operational expenditures. Additionally, the recovery and reuse of methanol solvent and the sale of byproduct sulfuric acid create ancillary revenue streams that further offset production costs, resulting in a highly competitive pricing structure for the final intermediate.
- Enhanced Supply Chain Reliability: By utilizing benzotrifluoride as the starting material, the process taps into a well-established supply chain with multiple global vendors, mitigating the risk of raw material shortages. The simplified two-step process reduces the number of unit operations required, which in turn decreases the probability of equipment failure or process delays that often plague multi-step synthetic routes. This operational simplicity translates to higher plant throughput and the ability to scale production rapidly in response to market demand fluctuations without compromising quality.
- Scalability and Environmental Compliance: The use of low-temperature nitration and standard hydrogenation equipment makes this process inherently scalable from pilot plants to multi-ton commercial reactors without significant engineering redesigns. The reduction in hazardous waste generation, particularly through the recycling of acids and solvents, aligns with increasingly stringent environmental regulations, reducing the liability and permitting hurdles associated with chemical manufacturing. This eco-friendly profile not only safeguards the company's social license to operate but also appeals to end-clients who prioritize green chemistry principles in their supplier selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of o-amino benzotrifluoride, derived directly from the patent specifications and industry best practices. These insights are intended to clarify the feasibility of adopting this route for large-scale production and to highlight the specific benefits over traditional methods. Understanding these nuances is critical for technical teams evaluating the integration of this intermediate into their existing supply chains.
Q: Why is a nickel catalyst preferred over palladium or platinum in this synthesis?
A: The use of a nickel catalyst significantly reduces production costs compared to noble metal catalysts like palladium or platinum, while maintaining high efficiency in the hydrogenation reduction step. Additionally, nickel catalysts are robust and suitable for large-scale industrial applications where cost control is critical.
Q: How does the new nitration process improve operational safety?
A: The patented method utilizes normal pressure and low temperature (35-45°C) conditions for the nitration reaction. This minimizes the corrosion of pipelines and equipment caused by mixed acids and reduces the risk of thermal runaway, making the process safer for operators and the environment compared to high-temperature alternatives.
Q: What are the primary challenges in separating the ortho-isomer from the reaction mixture?
A: Due to the meta-directing effect of the trifluoromethyl group, the nitration of benzotrifluoride predominantly yields the meta-isomer. The process addresses this by employing precise vacuum rectification techniques to separate the desired o-amino benzotrifluoride (boiling point 118 ± 2°C) from the meta and para isomers effectively.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable o-Amino Benzotrifluoride Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-quality fluorine intermediates in the development of next-generation agrochemicals and pharmaceuticals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory synthesis to industrial manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize advanced analytical techniques to verify the absence of critical impurities and isomers. Our facility is equipped to handle the specific safety requirements of nitration and hydrogenation processes, guaranteeing a secure and compliant supply of o-amino benzotrifluoride for your critical projects.
We invite you to collaborate with us to leverage these technological advancements for your specific application needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized synthesis method can enhance your product's competitiveness and supply chain resilience.
