Advanced One-Pot Synthesis of 4-Tolyloxy Benzylamine for Scalable Agrochemical Manufacturing
Advanced One-Pot Synthesis of 4-Tolyloxy Benzylamine for Scalable Agrochemical Manufacturing
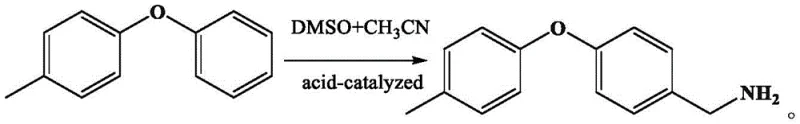
The global demand for high-efficiency pyrazole amide insecticides, such as Tolfenpyrad, continues to drive innovation in the synthesis of key intermediates. A significant technological breakthrough has been documented in patent CN115028542A, which details a novel preparation method for 4-tolyloxy benzylamine. This compound serves as a critical building block in the agrochemical sector, specifically for the production of next-generation acaricides and insecticides. The disclosed technology represents a paradigm shift from traditional, hazardous reduction methods to a safer, acid-catalyzed benzylic amination process. By utilizing a one-pot reaction system involving 4-phenoxytoluene, dimethyl sulfoxide (DMSO), and acetonitrile, this method addresses long-standing challenges regarding process safety, environmental impact, and cost-efficiency. For R&D directors and supply chain managers alike, understanding the nuances of this synthetic route is essential for securing a reliable agrochemical intermediate supplier capable of delivering high-purity materials without the baggage of high-risk manufacturing protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-tolyloxy benzylamine has relied heavily on the reduction of 4-tolyloxy benzonitrile. This conventional pathway typically necessitates the use of high-pressure hydrogenation or strong reducing agents such as sodium borohydride. From a process safety perspective, these methods introduce substantial risks; hydrogenation requires specialized high-pressure reactors and rigorous safety controls to prevent catastrophic failures, while sodium borohydride poses explosion hazards if not handled with extreme precision. Furthermore, these traditional routes often suffer from poor controllability regarding productivity per unit time and generate significant amounts of hazardous waste, leading to elevated discharge costs. The reliance on transition metal catalysts in hydrogenation also introduces the complex downstream burden of removing trace heavy metals to meet stringent pharmaceutical and agrochemical purity specifications, thereby complicating the purification workflow and inflating operational expenditures.
The Novel Approach
In stark contrast, the innovative method described in the patent utilizes a direct benzylic amination of 4-phenoxytoluene in a homogeneous organic solvent system. This approach completely circumvents the need for high-pressure equipment or explosive reducing agents, fundamentally altering the risk profile of the manufacturing process. The reaction proceeds smoothly in a mixture of DMSO and acetonitrile under acid catalysis, allowing for precise control over reaction kinetics at moderate temperatures ranging from 50°C to 80°C. This one-pot strategy not only simplifies the operational procedure but also significantly enhances the total yield and reduces the generation of three wastes (wastewater, waste gas, and solid waste). By eliminating the hydrogenation step, manufacturers can achieve a more streamlined production flow that is inherently safer and more conducive to large-scale industrialization, making it an ideal candidate for cost reduction in insecticide manufacturing.
Mechanistic Insights into Acid-Catalyzed Benzylic Amination
The core of this technological advancement lies in the efficient activation of the benzylic position of 4-phenoxytoluene through acid catalysis in a polar aprotic environment. The presence of DMSO and acetonitrile creates a highly polar medium that stabilizes the transition states involved in the amination process, facilitating the nucleophilic attack required to install the amino group. Unlike radical-based reductions which can lead to indiscriminate side reactions, this ionic mechanism offers superior selectivity. The acid catalyst, whether it be concentrated hydrochloric acid or HCl gas, protonates the intermediate species, lowering the activation energy barrier for the substitution reaction. This mechanistic pathway ensures that the ether linkage remains intact while the benzylic position is selectively functionalized, a critical factor in maintaining the structural integrity of the final agrochemical intermediate. The robustness of this mechanism allows for a wide tolerance of reaction conditions, providing process chemists with a flexible window for optimization.
Impurity control is another critical aspect where this mechanism excels, particularly for R&D teams focused on purity profiles. Traditional hydrogenation methods often produce over-reduced byproducts or leave behind trace metal contaminants that are difficult to purge. In the acid-catalyzed system, the primary impurities are typically unreacted starting materials or minor regio-isomers, which are far easier to separate via standard crystallization techniques. The patent data highlights that by monitoring the reaction via liquid chromatography until the starting material content drops below 1%, operators can ensure maximum conversion before workup. The subsequent crystallization step, triggered by pH adjustment and water addition, effectively purges soluble impurities, resulting in a product with a chemical purity exceeding 98%. This high level of intrinsic purity reduces the need for extensive chromatographic purification, directly translating to lower processing costs and higher throughput.
How to Synthesize 4-Tolyloxy Benzylamine Efficiently
Implementing this synthesis route requires careful attention to solvent ratios and temperature control to maximize the benefits of the one-pot design. The process begins with the sequential addition of the organic solvent, substrate, DMSO, acetonitrile, and acid under an inert nitrogen atmosphere to prevent oxidation. Heating the mixture to a set temperature, optimally between 50°C and 80°C, initiates the amination reaction which is typically maintained for 12 to 24 hours depending on the scale and specific acid catalyst used. Once the reaction reaches completion, the valuable solvent system is recovered under reduced pressure for reuse, a step that significantly contributes to the economic viability of the process. The detailed standardized synthesis steps, including specific molar ratios and workup procedures, are outlined in the guide below.
- Charge the reactor with organic solvent, 4-phenoxytoluene, DMSO, acetonitrile, and acid under nitrogen protection, then heat to 50-80°C.
- Maintain reaction temperature until HPLC analysis confirms 4-phenoxytoluene content is below 1%, typically requiring 12-24 hours.
- Recover solvents under reduced pressure, add water to the residue, adjust pH to 7, and crystallize to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers compelling advantages that extend beyond mere technical feasibility. The elimination of high-pressure hydrogenation equipment drastically reduces capital expenditure (CAPEX) requirements for manufacturing facilities, allowing for production in standard glass-lined or stainless steel reactors that are more readily available. Furthermore, the ability to recover and reuse the DMSO and acetonitrile solvent mixture at rates exceeding 90% creates a closed-loop system that minimizes raw material consumption and waste disposal fees. This efficiency is crucial for maintaining competitive pricing in the volatile agrochemical market. By adopting this method, companies can secure a more stable supply of 4-tolyloxy benzylamine, insulated from the regulatory and safety disruptions that often plague facilities relying on hazardous hydrogenation processes.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the reaction setup and the high recovery rate of solvents. By avoiding the use of expensive transition metal catalysts like palladium on carbon, the method eliminates the costly and time-consuming step of heavy metal scavenging and filtration. Additionally, the use of commodity chemicals such as hydrochloric acid and acetonitrile ensures that raw material costs remain low and predictable. The high isolated yield of approximately 86% to 89% means that less starting material is wasted per kilogram of product, further driving down the cost of goods sold (COGS) and enhancing overall profit margins for the final insecticide formulation.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the strict regulatory oversight surrounding high-risk chemical processes. Since this method operates under atmospheric pressure and avoids explosive reagents, it faces fewer regulatory hurdles and safety inspections, leading to more consistent production schedules. The raw material, 4-phenoxytoluene, is widely available from multiple sources, reducing the risk of supply bottlenecks associated with specialized precursors. This robustness ensures that procurement teams can rely on a steady flow of high-purity intermediates, minimizing the risk of production stoppages due to raw material shortages or safety-related shutdowns at the supplier's facility.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to the pilot plant and finally to commercial production is often fraught with unforeseen challenges, particularly with exothermic hydrogenation reactions. This acid-catalyzed amination is inherently easier to scale due to its mild thermal profile and lack of gas evolution, making the commercial scale-up of complex agrochemical intermediates significantly more predictable. Moreover, the reduction in hazardous waste generation aligns with increasingly stringent global environmental regulations. The ability to recycle solvents and minimize aqueous waste discharge not only lowers environmental compliance costs but also enhances the corporate sustainability profile of the manufacturing entity, a key factor for modern supply chain partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 4-tolyloxy benzylamine using this patented methodology. These insights are derived directly from the experimental data and process descriptions found in the source patent, providing a transparent view of the technology's capabilities. Understanding these details helps stakeholders make informed decisions about integrating this intermediate into their supply chains.
Q: How does this new method improve safety compared to traditional hydrogenation?
A: Traditional methods often rely on high-pressure hydrogenation or explosive reagents like sodium borohydride. This patented acid-catalyzed process operates at atmospheric pressure and moderate temperatures (50-80°C), eliminating explosion risks and simplifying equipment requirements.
Q: What is the expected yield and purity for commercial scale production?
A: Pilot plant data indicates a chemical purity of over 98% with an isolated yield ranging from 86% to 89%. The process also allows for approximately 90-92% recovery of the acetonitrile and DMSO solvent system.
Q: Which acid catalysts are most effective for this transformation?
A: While various acids like sulfuric acid and phosphoric acid were tested, concentrated hydrochloric acid and HCl gas dissolved in acetonitrile demonstrated superior performance, achieving yields of 85% and 92% respectively in comparative studies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Tolyloxy Benzylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting safer and more efficient synthetic routes for key agrochemical intermediates. Our technical team has thoroughly analyzed the potential of this acid-catalyzed amination process and is fully prepared to implement it for our global partners. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale innovation to industrial reality is seamless. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 4-tolyloxy benzylamine meets the exacting standards required for Tolfenpyrad synthesis. We are committed to delivering high-purity agrochemical intermediates that empower your R&D and production teams.
We invite you to collaborate with us to leverage this advanced technology for your supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this process can optimize your budget. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can enhance your production efficiency and supply security.
