Advanced Ionic Liquid Decarboxylation for High-Purity Pyrazinecarboxylic Acid Commercial Production
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic pathways that balance high purity with environmental sustainability. A pivotal advancement in this domain is documented in patent CN101875640A, which details a novel method for preparing pyrazinecarboxylic acid, a critical intermediate used extensively in the synthesis of various bioactive compounds and flavoring agents. This technology replaces traditional volatile organic solvents with task-specific ionic liquids, fundamentally altering the thermodynamics and kinetics of the decarboxylation process. By leveraging the unique solvation properties of imidazolium-based salts, this approach not only mitigates the severe corrosion and odor issues associated with conventional acetic acid methods but also introduces a closed-loop solvent system. For global procurement leaders and R&D directors, understanding this shift is essential, as it represents a move towards greener manufacturing that does not compromise on the rigorous quality standards required for pharmaceutical intermediates. The ability to execute this transformation efficiently positions suppliers to offer more stable pricing and reliable delivery schedules in a volatile market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of pyrazinecarboxylic acid has relied heavily on thermal decarboxylation using glacial acetic acid as the reaction medium. While chemically effective, this legacy process presents substantial operational challenges that impact both safety and cost structures. Acetic acid is highly corrosive, necessitating the use of expensive specialized alloy reactors and piping systems to prevent equipment failure and leakage. Furthermore, its high volatility leads to significant solvent loss during the heating phases, requiring complex condensation and recovery systems to meet environmental emission standards. The strong, pungent odor of acetic acid also creates difficult working conditions, often requiring extensive ventilation infrastructure. From a purification standpoint, separating the product from the acidic solvent can be energy-intensive, often involving multiple distillation or neutralization steps that generate large volumes of saline wastewater. These factors collectively inflate the operational expenditure and introduce supply chain risks related to hazardous material handling and waste disposal compliance.
The Novel Approach
In stark contrast, the methodology outlined in the referenced patent utilizes functionalized ionic liquids, specifically carboxyl-functionalized imidazolium salts, to facilitate the decarboxylation of 2,3-pyrazinedicarboxylic acid. This innovative solvent system operates effectively at temperatures ranging from 40°C to 180°C, with optimal performance observed between 80°C and 140°C. The ionic liquid acts not merely as a solvent but as a stabilizing matrix that promotes the selective removal of the carboxyl group while minimizing side reactions. Because ionic liquids possess negligible vapor pressure, the process eliminates the fugitive emissions typical of volatile organic compounds, drastically reducing the environmental footprint. Moreover, the post-reaction workup is remarkably simplified; the product precipitates upon cooling or addition of anti-solvents, allowing for straightforward filtration. This transition from a corrosive, volatile system to a stable, recyclable ionic medium represents a paradigm shift in process chemistry, offering a cleaner, safer, and potentially more cost-effective route for large-scale manufacturing.
Mechanistic Insights into Ionic Liquid-Mediated Decarboxylation
The core of this technological breakthrough lies in the interaction between the substrate, 2,3-pyrazinedicarboxylic acid, and the ionic liquid catalyst. The ionic liquid, characterized by a cationic imidazolium ring with alkyl and carboxyl substituents and a halide anion, creates a highly polar environment that stabilizes the transition state of the decarboxylation reaction. The mechanism likely involves the formation of a hydrogen-bonding network between the carboxylic acid groups of the substrate and the ionic liquid species, which lowers the activation energy required for carbon dioxide elimination. Unlike traditional thermal decarboxylation which relies solely on high heat to overcome energy barriers, the ionic liquid facilitates a more controlled bond cleavage. This control is crucial for maintaining the integrity of the pyrazine ring, preventing polymerization or degradation that can occur under harsh acidic conditions. The specific structure of the ionic liquid, where R1 is a C1-C10 alkyl group and R2 is a C1-C10 alkylene group, allows for tunability of the solvent properties to match the solubility profile of the reactants and products.
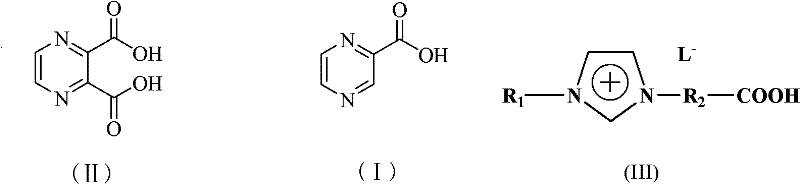
Furthermore, the impurity profile of the final product is significantly improved through this mechanism. In conventional acetic acid processes, side reactions such as esterification or acetylation can occur, leading to difficult-to-remove impurities that require extensive recrystallization. The ionic liquid system, being non-nucleophilic in nature regarding the acyl group, suppresses these competing pathways. The selectivity of the reaction ensures that the primary transformation is the loss of one carboxyl group to yield the mono-acid product. Post-reaction analysis indicates that the melting point of the product remains consistent at 224-225°C, indicative of high purity. For R&D directors, this mechanistic clarity is vital; it suggests that scaling this process will not introduce unpredictable impurity spikes, thereby reducing the burden on quality control laboratories and ensuring batch-to-batch consistency essential for regulatory filings.
How to Synthesize Pyrazinecarboxylic Acid Efficiently
Implementing this synthesis route requires precise control over stoichiometry and thermal parameters to maximize yield and solvent recovery. The process begins with the charging of the reactor with 2,3-pyrazinedicarboxylic acid and the selected ionic liquid, typically at a mass ratio ranging from 1:5 to 1:50, with a preferred range of 1:5 to 1:50 to ensure adequate solvation without excessive dilution. The mixture is then heated to the target temperature, ideally between 80°C and 140°C, and maintained for a duration of 3 to 8 hours depending on the specific thermal profile desired. Upon completion, the reaction mixture is cooled to induce crystallization of the pyrazinecarboxylic acid. The solid is collected via suction filtration, and the filtrate containing the ionic liquid is retained for reuse. The crude product is subsequently purified through recrystallization using water or aqueous ethanol solutions. Detailed standardized operating procedures and specific parameter optimizations for different reactor scales are provided in the technical guide below.
- Charge the reactor with 2,3-pyrazinedicarboxylic acid and the specific ionic liquid catalyst at a mass ratio between 1: 5 and 1:50.
- Heat the reaction mixture to a temperature range of 80°C to 140°C and maintain stirring for 3 to 8 hours to ensure complete decarboxylation.
- Cool the reaction mixture to precipitate the solid product, followed by suction filtration and recrystallization using water or ethanol-water mixtures.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ionic liquid technology translates into tangible strategic benefits beyond mere chemical efficiency. The most significant advantage lies in the drastic simplification of the downstream processing workflow. By eliminating the need for complex solvent recovery distillation columns required for acetic acid, the capital expenditure for new production lines is reduced, and the operational complexity of existing lines is minimized. This simplicity directly correlates to enhanced supply chain reliability, as fewer unit operations mean fewer potential points of failure or bottlenecking during high-volume production campaigns. Additionally, the recyclability of the ionic liquid means that the consumption of auxiliary materials is substantially lowered over the lifecycle of the plant. This reduction in material throughput not only lowers the variable cost per kilogram but also insulates the manufacturing process from fluctuations in raw solvent markets. Consequently, suppliers utilizing this technology can offer more stable long-term pricing contracts, a critical factor for multinational corporations managing annual budgets.
- Cost Reduction in Manufacturing: The economic model of this process is driven by the elimination of expensive corrosion-resistant infrastructure and the reduction of waste treatment costs. Since the ionic liquid does not volatilize, there is no loss of solvent to the atmosphere, and the ability to reuse the catalytic medium for multiple cycles without significant degradation means the effective cost of the solvent per batch approaches zero after the initial charge. Furthermore, the simplified workup, which relies on filtration and recrystallization rather than energy-intensive distillation, results in significantly lower utility consumption for steam and cooling water. These cumulative efficiencies allow for a leaner cost structure, enabling competitive pricing strategies without sacrificing margin. The removal of hazardous waste streams associated with acidic wastewater treatment further reduces compliance costs and liability risks.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by regulatory crackdowns on environmental emissions and the availability of hazardous raw materials. This green chemistry approach mitigates those risks by operating within a closed system with minimal emissions. The raw materials, specifically the imidazolium precursors and the pyrazine dicarboxylic acid, are commercially available and stable, ensuring that production is not held hostage by obscure reagent shortages. The robustness of the reaction conditions, which tolerate a broad temperature window, also means that minor fluctuations in utility supply (such as steam pressure) are less likely to cause batch failures. This resilience ensures that delivery schedules can be met consistently, even during periods of high market demand or logistical strain, providing peace of mind to supply chain planners.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to multi-ton production often reveals hidden complexities, particularly regarding heat transfer and mixing in viscous media. However, the fluid nature of the ionic liquids at reaction temperatures facilitates excellent heat transfer, making the scale-up from 100 kgs to 100 MT annual commercial production technically feasible and predictable. From an environmental perspective, this process aligns perfectly with increasingly stringent global regulations regarding volatile organic compound (VOC) emissions and hazardous waste generation. By adopting this technology, manufacturers future-proof their operations against tightening environmental laws, avoiding costly retrofits or shutdowns. The "green" label associated with ionic liquid chemistry also enhances the brand value of the final product, appealing to end-users who prioritize sustainable sourcing in their own supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ionic liquid decarboxylation technology. These answers are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this method into their existing manufacturing portfolios. The focus is on practical application, yield expectations, and the specific advantages over legacy methods.
Q: What are the primary advantages of using ionic liquids over acetic acid for this decarboxylation?
A: Unlike traditional acetic acid methods which involve strong corrosiveness and irritating odors, the ionic liquid method described in patent CN101875640A offers a non-volatile, recyclable medium that significantly simplifies post-treatment and reduces environmental hazards.
Q: Can the ionic liquid catalyst be recovered and reused in subsequent batches?
A: Yes, the patent explicitly states that the ionic liquid filtrate obtained after suction filtration does not require complex post-treatment and can be directly reused for the next reaction cycle, enhancing process economics.
Q: What is the optimal temperature range for maximizing yield in this specific pathway?
A: While the reaction can proceed between 40°C and 180°C, experimental data suggests that maintaining the temperature between 80°C and 140°C provides the best balance between reaction rate and product yield, avoiding thermal degradation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrazinecarboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic methodologies requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO and supplier in the fine chemical sector, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped to handle complex chemistries, including ionic liquid systems, with stringent purity specifications and rigorous QC labs that ensure every batch meets international pharmacopeial standards. We understand the critical nature of pyrazinecarboxylic acid as a building block for high-value pharmaceuticals and flavors, and we are committed to delivering this key intermediate with the reliability and quality your operations demand. Our team is ready to collaborate on process optimization to further enhance efficiency and sustainability.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain insights into the potential economic impact of switching to this greener manufacturing method. We encourage you to contact us to obtain specific COA data from our pilot runs and to discuss route feasibility assessments tailored to your volume needs. Let us partner with you to secure a sustainable and cost-effective supply of high-purity pyrazinecarboxylic acid for your global operations.
