Advanced Five-Step Synthesis of Cyclohexyl Ethylene for High-Performance Liquid Crystal Manufacturing
The global demand for high-performance liquid crystal materials continues to surge, driven by the expansion of display technologies in consumer electronics and automotive sectors. At the heart of this supply chain lies the need for robust, scalable synthesis routes for key intermediates like cyclohexyl ethylene derivatives. Patent CN112321430A introduces a groundbreaking preparation method that addresses long-standing inefficiencies in this domain. By leveraging a sequential five-step transformation involving Wittig-Horner olefination, catalytic hydrogenation, selective reduction, bromination, and final elimination, this technology offers a pathway to produce high-purity cyclohexyl ethylene compounds with exceptional thermal stability and dielectric properties. For R&D directors and procurement strategists, this patent represents a pivotal shift towards more cost-effective and reliable manufacturing protocols for next-generation display mixed liquid crystals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cyclohexyl ethylene compounds has been plagued by significant operational hurdles that impact both cost and quality. Traditional routes often suffer from low product yields and purity, necessitating complex and expensive purification steps such as repeated column chromatography or distillation under high vacuum. Furthermore, many prior art methods rely on raw materials that are either prohibitively expensive or difficult to source in bulk quantities, creating bottlenecks in the supply chain. The use of harsh reaction conditions in older methodologies can also lead to the formation of difficult-to-remove impurities, which is detrimental for electronic grade materials where trace contaminants can compromise device performance. These factors collectively result in elevated production costs and extended lead times, making it challenging for manufacturers to meet the rigorous demands of the display industry.
The Novel Approach
In stark contrast, the method disclosed in CN112321430A utilizes substituted cyclohexyl ketone as a readily accessible starting material, streamlining the entire synthetic trajectory. The process initiates with a Wittig-Horner reaction to construct the carbon framework, followed by a highly efficient hydrogenation step.
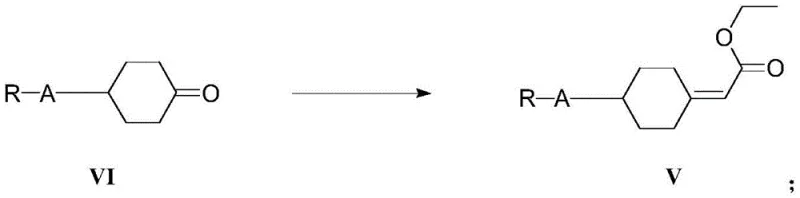
This novel approach eliminates the need for exotic reagents, relying instead on standard industrial chemicals like triethyl phosphonoacetate and common bases. The strategic design of the reaction sequence ensures that each intermediate can be purified through simple crystallization or extraction, drastically reducing solvent consumption and waste. By optimizing reaction parameters such as temperature and molar ratios, the inventors have achieved a process that not only improves overall yield but also enhances the consistency of the final product, making it an ideal solution for cost reduction in electronic chemical manufacturing.
Mechanistic Insights into the Five-Step Synthetic Cascade
The core of this technology lies in its precise control over chemoselectivity across five distinct transformations. The initial Wittig-Horner reaction is conducted at low temperatures (0-15°C) using sodium alkoxides, ensuring the formation of the desired alpha,beta-unsaturated ester with high stereoselectivity. Following this, the hydrogenation step employs a Palladium on Carbon (Pd/C) catalyst under moderate hydrogen pressure (0.4-1.2 MPa) to selectively reduce the olefinic bond without affecting the ester functionality. A critical innovation occurs in the third step, where the ester is reduced to an alcohol.
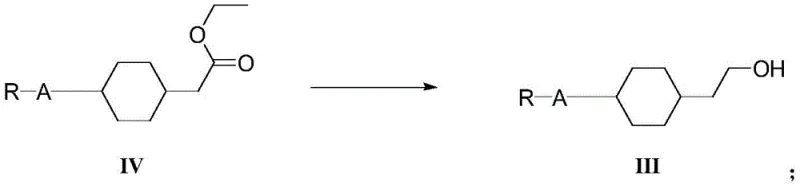
Unlike standard reductions that might require hazardous hydrides alone, this method utilizes a borohydride salt in conjunction with a Lewis acid metal catalyst, specifically Zinc Chloride (ZnCl2). This combination facilitates a smoother reduction profile at moderate temperatures (60-80°C), minimizing side reactions. The subsequent bromination with hydrobromic acid converts the alcohol to an alkyl bromide, which serves as the precursor for the final elimination. The terminal step involves a base-mediated elimination using potassium tert-butoxide at elevated temperatures (90-110°C) to generate the terminal vinyl group.
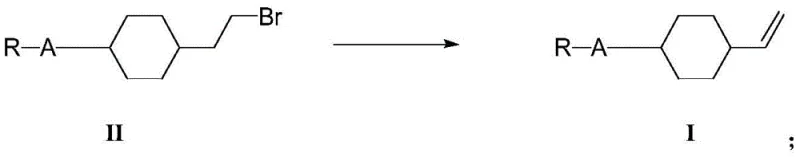
This mechanistic pathway is meticulously engineered to prevent the degradation of the cyclohexyl rings, preserving the structural integrity required for liquid crystal applications. The careful selection of solvents, such as toluene and THF, further aids in maintaining reaction homogeneity and facilitating easy workup, ensuring that the final product meets the stringent purity specifications demanded by high-end display manufacturers.
How to Synthesize Cyclohexyl Ethylene Efficiently
Implementing this synthesis route requires adherence to specific operational parameters to maximize yield and safety. The process begins with the preparation of the unsaturated ester under inert atmosphere, followed by high-pressure hydrogenation which necessitates appropriate autoclave equipment. The reduction and bromination steps require careful temperature control to manage exotherms, while the final elimination demands anhydrous conditions to prevent hydrolysis of the base. For a comprehensive guide on reactor setup, reagent addition rates, and quenching procedures, please refer to the standardized protocol below.
- Perform a Wittig-Horner reaction on substituted cyclohexyl ketone (Formula VI) with triethyl phosphonoacetate and a base to form the unsaturated ester (Formula V).
- Subject the unsaturated ester to catalytic hydrogenation using Pd/C to saturate the double bond, yielding the saturated ester (Formula IV).
- Reduce the ester group to an alcohol (Formula III) using a borohydride salt in the presence of a metal catalyst like Zinc Chloride.
- Convert the alcohol to a bromide (Formula II) via bromination reaction using hydrobromic acid.
- Execute a final elimination reaction using a strong organic base to generate the target vinyl compound (Formula I).
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patent offers substantial value by addressing key pain points in the supply chain for display materials. The reliance on commodity chemicals rather than specialized intermediates significantly de-risks the sourcing strategy, ensuring continuity of supply even during market fluctuations. The simplified purification workflow translates directly into lower operational expenditures, as less energy and fewer solvents are required to achieve pharmaceutical or electronic grade purity.
- Cost Reduction in Manufacturing: The elimination of complex purification steps such as extensive column chromatography results in significant cost savings. By utilizing high-yield reactions that produce crude products amenable to simple recrystallization, manufacturers can reduce solvent waste disposal costs and increase throughput. The use of recoverable catalysts like Pd/C further contributes to long-term economic efficiency, making this route highly competitive for large-scale production.
- Enhanced Supply Chain Reliability: The raw materials identified in this process, including substituted cyclohexyl ketones and triethyl phosphonoacetate, are widely available from multiple global suppliers. This diversification of the supply base mitigates the risk of single-source dependency. Furthermore, the robustness of the reaction conditions means that production is less susceptible to minor variations in raw material quality, ensuring consistent output and reliable delivery schedules for downstream clients.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively in multi-gram to kilogram scales in the patent examples. The use of standard organic solvents like toluene and ethanol simplifies solvent recovery and recycling systems, aligning with modern environmental compliance standards. The reduction in hazardous waste generation compared to traditional methods supports corporate sustainability goals and reduces the regulatory burden associated with chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cyclohexyl ethylene synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on feasibility and performance metrics for potential adopters.
Q: What are the key advantages of this cyclohexyl ethylene preparation method over prior art?
A: This method utilizes a streamlined five-step sequence starting from readily available substituted cyclohexyl ketones. It avoids complex purification steps often associated with traditional routes, achieving product purity greater than 99% GC through standard recrystallization and extraction techniques, thereby significantly lowering production costs and operational complexity.
Q: Which catalysts are critical for the reduction and hydrogenation steps in this process?
A: The process employs Palladium on Carbon (Pd/C) for the hydrogenation of the double bond under mild pressure (0.4-1.2 MPa). For the subsequent reduction of the ester to an alcohol, the method uniquely utilizes a combination of borohydride salts (such as Sodium Borohydride) and a Lewis acid metal catalyst, preferably Zinc Chloride (ZnCl2), to ensure high chemoselectivity and yield.
Q: Is this synthesis route scalable for industrial production of liquid crystal materials?
A: Yes, the route is designed for scalability. It uses common organic solvents like toluene and ethanol, operates at moderate temperatures (mostly below 110°C), and utilizes robust reagents like hydrobromic acid and potassium tert-butoxide. The high yields reported in the examples (often exceeding 90% per step) indicate strong potential for commercial scale-up with minimal waste generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyclohexyl Ethylene Supplier
As the display industry evolves, the need for high-purity intermediates produced via efficient routes becomes paramount. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging deep expertise in organic synthesis to bring patented technologies like CN112321430A to commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of cyclohexyl ethylene meets the exacting standards required for liquid crystal mixtures.
We invite you to collaborate with us to optimize your material sourcing strategy. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can enhance your product performance and bottom line.
