Advanced Photocatalytic Synthesis of Organic Selenium Compounds for Commercial Scale-up
Advanced Photocatalytic Synthesis of Organic Selenium Compounds for Commercial Scale-up
The landscape of fine chemical manufacturing is constantly evolving, driven by the need for more sustainable and efficient synthetic routes for complex molecular architectures. A significant breakthrough in this domain is detailed in patent CN114702466A, which discloses a novel preparation method for organic selenium compounds. These compounds are not merely academic curiosities; they serve as critical building blocks in the synthesis of pharmaceutical agents, particularly those targeting cardiovascular diseases and tumor inhibition. The core innovation lies in a photocatalytic radical substitution strategy that bypasses traditional oxidative limitations. By leveraging visible to near-UV light activation, this technology enables the direct functionalization of ether alpha-positions with selenium moieties. For R&D directors and procurement specialists seeking a reliable organic selenium compound supplier, understanding the mechanistic elegance and operational simplicity of this patent is crucial for securing a competitive edge in the supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of organoselenium species has been fraught with significant chemical and logistical challenges that hinder large-scale adoption. Traditional pathways often rely heavily on strong oxidizing agents such as hydrogen peroxide (H2O2), meta-chloroperoxybenzoic acid (MCPBA), or ozone to generate reactive selenoxide intermediates. These reagents are inherently hazardous, posing severe safety risks regarding thermal runaway and explosion potential during commercial scale-up of complex pharmaceutical intermediates. Furthermore, the subsequent elimination reactions required to form double or triple bonds from these selenoxides often suffer from poor regioselectivity and generate substantial quantities of toxic selenium waste. The purification of final products from these messy reaction mixtures typically requires extensive chromatographic separation, driving up both the cost of goods sold (COGS) and the environmental footprint. Additionally, many classical methods require cryogenic conditions or strictly anhydrous environments that are difficult to maintain in multi-ton reactors, leading to batch-to-batch variability and inconsistent purity profiles.
The Novel Approach
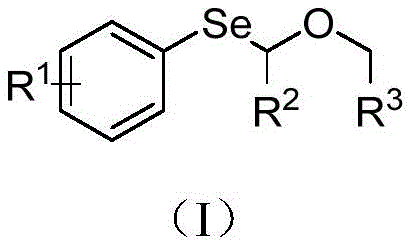
In stark contrast to these archaic techniques, the methodology described in CN114702466A introduces a streamlined, photocatalytic radical pathway that operates under remarkably mild conditions. This novel approach utilizes a benzophenone derivative, specifically 4-chlorobenzophenone, to abstract hydrogen atoms from the alpha-position of ether substrates upon illumination. This generates a transient alkyl radical that directly attacks a diaryl diselenide bond, forging the carbon-selenium linkage with high precision. As demonstrated in the reaction schemes below, this method eliminates the need for dangerous stoichiometric oxidants entirely. The process runs efficiently at room temperature (25°C) in common solvents like tetrahydrofuran (THF), utilizing cesium hydroxide as a benign base. This shift from oxidative chemistry to photocatalytic radical chemistry represents a paradigm shift, offering a route that is not only safer but also significantly more atom-economical. The ability to tolerate various substituents on the aromatic ring, such as methyl or chloro groups, further underscores the versatility of this platform for generating diverse libraries of bioactive scaffolds.
Mechanistic Insights into Photocatalytic Radical Substitution
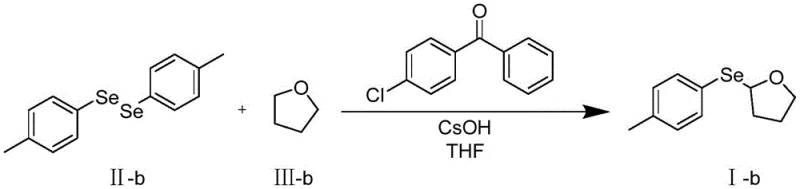
To fully appreciate the value proposition of this technology for cost reduction in pharmaceutical intermediates manufacturing, one must delve into the specific mechanistic steps that govern the reaction efficiency. The cycle initiates when the photocatalyst, 4-chlorobenzophenone, absorbs photons in the 300-390 nm wavelength range, promoting it to an excited triplet state. In this high-energy state, the carbonyl oxygen of the benzophenone acts as a potent hydrogen abstractor. It selectively targets the relatively weak C-H bonds at the alpha-position of the ether substrate (Formula III). This hydrogen atom transfer (HAT) event generates a stabilized alpha-oxy alkyl radical and a ketyl radical species. The resulting carbon-centered radical is nucleophilic in character and rapidly adds to the electrophilic selenium atom of the diaryl diselenide (Formula II). This addition triggers the homolytic cleavage of the weak Se-Se bond, releasing an arylselenyl radical which propagates the chain or terminates to regenerate the catalyst system. The use of cesium hydroxide is critical here; it likely facilitates the deprotonation steps or stabilizes intermediate ionic species, ensuring the reaction proceeds to completion without stalling.
From an impurity control perspective, this mechanism offers distinct advantages over ionic pathways. Because the reaction relies on radical kinetics rather than harsh electrophilic attacks, there is minimal risk of over-oxidation or degradation of sensitive functional groups on the aromatic rings. The mild thermal profile (0-150°C, optimally 25°C) prevents thermal decomposition of the product, which is a common issue in high-temperature selenium chemistry. Furthermore, the specificity of the hydrogen abstraction ensures that substitution occurs exclusively at the desired alpha-position, minimizing the formation of regioisomers that are notoriously difficult to separate. This high level of chemoselectivity translates directly to higher crude purity, reducing the burden on downstream purification units. For quality assurance teams, this means a more robust process capable of consistently meeting stringent purity specifications required for GMP manufacturing of active pharmaceutical ingredients.
How to Synthesize Organic Selenium Compounds Efficiently
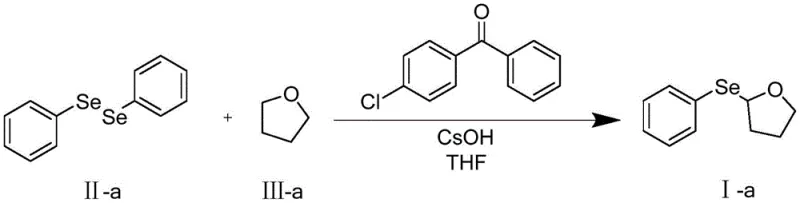
Implementing this photocatalytic protocol in a laboratory or pilot plant setting requires strict adherence to the optimized parameters defined in the patent to ensure maximum yield and reproducibility. The process is designed to be operationally simple, avoiding the need for specialized high-pressure equipment or exotic reagents. The key to success lies in the precise control of the light source wavelength and the maintenance of an inert atmosphere to prevent radical quenching by oxygen. Below is a summary of the standardized procedure derived from the exemplary embodiments, which serves as a foundational guide for process chemists looking to replicate these results.
- Load a reactor with 4-chlorobenzophenone photocatalyst and diaryl diselenide under inert atmosphere.
- Introduce the ether substrate and cesium hydroxide base into the reaction mixture.
- Illuminate the mixture with 300-390 nm light at 25°C for 12 hours, then purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this photocatalytic methodology offers tangible strategic benefits that extend beyond mere technical novelty. The primary advantage lies in the drastic simplification of the raw material portfolio. By eliminating the need for hazardous oxidants like MCPBA or ozone generators, facilities can reduce their inventory of dangerous chemicals, thereby lowering insurance premiums and regulatory compliance costs. The reliance on commodity chemicals such as diaryl diselenides, simple ethers, and cesium hydroxide ensures a stable and resilient supply chain, mitigating the risk of shortages associated with specialty reagents. Furthermore, the reaction's ability to proceed at ambient temperature significantly reduces energy consumption compared to processes requiring reflux or cryogenic cooling, contributing to long-term operational expenditure (OpEx) savings.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the elimination of expensive stoichiometric oxidants and the simplification of the workup procedure. Traditional selenium chemistry often generates heavy metal waste or requires complex extraction protocols to remove selenium byproducts, which drives up waste disposal costs. In this new method, the byproduct is essentially a selenol or diselenide derivative that can potentially be recycled or is easily separated via standard silica gel chromatography. The high atom economy of the radical substitution means that a larger proportion of the starting mass ends up in the final product, directly improving the theoretical yield ceiling. Additionally, the use of a catalytic amount of benzophenone derivative, rather than stoichiometric reagents, further lowers the material cost per kilogram of product produced.
- Enhanced Supply Chain Reliability: Securing a consistent supply of high-purity intermediates is critical for maintaining uninterrupted API production schedules. This photocatalytic process enhances reliability by utilizing robust, shelf-stable reagents that do not require cold-chain logistics or special handling permits. Diaryl diselenides and ether solvents are widely available from multiple global vendors, reducing dependency on single-source suppliers. The mild reaction conditions also mean that the process is less susceptible to failure due to minor fluctuations in utility supplies, such as steam or chilled water availability. This inherent robustness makes the technology ideal for continuous manufacturing setups, where steady-state operation is paramount for meeting just-in-time delivery commitments to downstream pharmaceutical clients.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been a bottleneck due to light penetration issues (Beer-Lambert law); however, modern flow chemistry reactors have largely solved this, making this technology ready for ton-scale production. The absence of toxic oxidants and the generation of minimal hazardous waste align perfectly with increasingly strict environmental regulations (EHS). Facilities adopting this method can expect a smoother permitting process and reduced liability associated with chemical storage and waste treatment. The ability to run the reaction in green solvents like THF or even potentially greener alternatives in future iterations further cements its status as a sustainable manufacturing solution. This environmental stewardship is increasingly becoming a key differentiator when qualifying suppliers for major multinational pharmaceutical contracts.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic selenium synthesis. These answers are derived directly from the experimental data and claims within patent CN114702466A, providing a reliable reference for technical due diligence. Understanding these specifics helps stakeholders evaluate the feasibility of integrating this chemistry into their existing production portfolios.
Q: What is the preferred photocatalyst for this selenium synthesis?
A: The patent specifies 4-chlorobenzophenone as the preferred photocatalyst due to its superior applicability to the substrate and high catalytic efficiency compared to other benzophenone derivatives.
Q: What are the optimal reaction conditions for high yield?
A: The optimal conditions involve using cesium hydroxide as the base, reacting at 25°C under 300-390 nm illumination for approximately 12 hours in anhydrous THF.
Q: Can this method be scaled for industrial production?
A: Yes, the method utilizes mild temperatures and readily available reagents, making it highly suitable for commercial scale-up without the need for extreme pressure or cryogenic conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Organic Selenium Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of photocatalytic technologies in modernizing the synthesis of high-value fine chemicals. Our team of expert process chemists has extensively evaluated the methodology described in CN114702466A and possesses the technical capability to adapt and optimize this route for your specific project needs. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to manufacturing plant is seamless and efficient. Our state-of-the-art facilities are equipped with advanced photoreactors and rigorous QC labs capable of maintaining stringent purity specifications required for pharmaceutical grade intermediates. We are committed to delivering not just a product, but a comprehensive supply solution that guarantees consistency and quality.
We invite you to collaborate with us to leverage this cutting-edge synthesis for your next development program. By partnering with our technical team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity targets. We encourage you to contact our technical procurement team today to request specific COA data for our selenium catalog and to discuss route feasibility assessments for your custom synthesis projects. Let us help you secure a competitive advantage through superior chemistry and reliable supply chain execution.
