Advanced Synthesis of Dimer Diamide: A Scalable Route for High-Performance DDI Manufacturing
The landscape of high-performance polymer additives and propellant binders is undergoing a significant transformation, driven by the urgent need for safer, more scalable synthetic routes. Patent CN101648885A introduces a groundbreaking methodology for the preparation of Dimer Diamide (DDA), a critical intermediate in the synthesis of Dimer Acid Diisocyanate (DDI). This technology addresses long-standing bottlenecks in the production of curing agents for composite solid propellants, specifically those based on hydroxyl-terminated polybutadiene (HTPB). By shifting away from hazardous high-pressure hydrogenation and toxic carbonyl azide rearrangements, this patent outlines a robust thermal amidation process utilizing urea and dimerized fatty acids. For R&D directors and procurement strategists, this represents a pivotal opportunity to secure a supply chain that is not only cost-effective but also inherently safer and more environmentally compliant. The ability to produce high-purity DDA with yields exceeding 80% under relatively mild thermal conditions marks a substantial leap forward in fine chemical manufacturing efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Dimer Acid Diisocyanate (DDI) and its precursors has been plagued by severe operational hazards and complex engineering requirements. Traditional pathways, such as the dinitrile method, necessitate the conversion of dimer acid to dimer nitrile followed by high-pressure hydrogenation over Raney nickel catalysts to produce dimer amine. This process demands specialized high-pressure reactors capable of withstanding extreme conditions, leading to prohibitive capital expenditures and significant safety liabilities associated with handling hydrogen gas and pyrophoric catalysts. Furthermore, the alternative carbonyl azide rearrangement method, while offering high yields, involves the generation and thermal decomposition of unstable azide intermediates, posing unacceptable explosion risks for large-scale industrial facilities. These legacy methods create fragile supply chains vulnerable to regulatory shutdowns and equipment failures, making them ill-suited for the consistent, high-volume production required by the aerospace and advanced materials sectors.
The Novel Approach
In stark contrast, the novel approach detailed in CN101648885A leverages a direct thermal amidation strategy that fundamentally simplifies the manufacturing workflow. By reacting dimerized linoleic acid or tungoleic acid directly with urea in a weight ratio ranging from 10:1 to 10:3, the process bypasses the need for hazardous nitrile intermediates entirely. The reaction proceeds through a controlled temperature ramp, initiating at 140-150°C and stabilizing at 170-200°C for a duration of 2 to 8 hours. This solvent-free initial phase minimizes waste generation and reduces the complexity of downstream processing. The resulting deep yellow transparent reaction liquid is then subjected to a straightforward workup involving neutralization, solvent extraction, and vacuum distillation. This streamlined protocol not only enhances operator safety by eliminating high-pressure steps but also drastically reduces the energy footprint and equipment maintenance costs associated with traditional high-severity chemical transformations.
Mechanistic Insights into Thermal Urea Amidation
The core chemical transformation driving this innovation is the thermal condensation of carboxylic acid groups on the dimer acid backbone with urea to form the corresponding diamide linkages. Unlike conventional amidation which often requires activating agents or coupling reagents, this thermal process relies on the inherent reactivity of urea at elevated temperatures to facilitate the nucleophilic attack on the carbonyl carbon of the fatty acid. As the temperature rises to the 170-200°C range, urea decomposes in situ to generate reactive isocyanic acid species or ammonia, which subsequently react with the carboxylic acid moieties. This mechanism effectively converts the dimer acid into the target Dimer Diamide structure while releasing volatile byproducts like carbon dioxide and ammonia, which are easily vented from the reactor system. The simplicity of this mechanistic pathway ensures high atom economy and minimizes the formation of complex side products that typically complicate purification in multi-step syntheses.
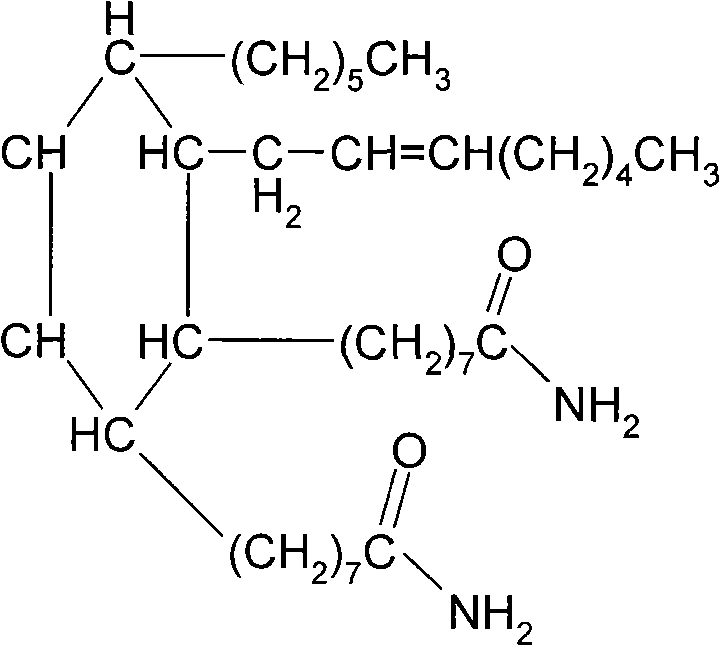
Controlling the impurity profile in this synthesis is critical for ensuring the downstream performance of the derived DDI curing agent. The patent specifies a rigorous purification sequence designed to remove unreacted starting materials and colored byproducts. Following the thermal reaction, the crude mixture is treated with aqueous sodium carbonate to neutralize any residual acidity, followed by multiple extractions with organic solvents such as ethyl acetate or petroleum ether. This liquid-liquid extraction phase is crucial for separating the lipophilic Dimer Diamide from polar impurities and inorganic salts. Subsequent treatment with activated carbon effectively adsorbs conjugated organic impurities responsible for discoloration, ensuring the final product meets the stringent aesthetic and purity specifications required for high-end applications. The final vacuum distillation step, conducted at mild temperatures of 20-80°C under reduced pressure (0.5-20 mmHg), guarantees the removal of trace solvents and low-boiling volatiles, delivering a product with exceptional chemical integrity.
How to Synthesize Dimer Diamide Efficiently
Implementing this synthesis route requires precise control over thermal profiles and stoichiometric ratios to maximize yield and minimize degradation. The process begins with the careful charging of dimer acid and urea into a reactor equipped with efficient stirring and heating capabilities. Operators must monitor the exothermic nature of the initial reaction phase closely as the temperature ramps to 150°C. Once the initial vigorous reaction subsides, the system is held at the target temperature of 170-200°C for a defined period, typically between 2 to 8 hours depending on the specific batch size and desired conversion. The subsequent workup involves cooling the reaction mass and performing a series of extractions and washes to isolate the organic phase. For a comprehensive understanding of the exact operational parameters, safety protocols, and quality control checkpoints required for successful execution, please refer to the standardized synthesis guide below.
- Charge reactor with dimer acid and urea (10: 1 to 10:3 weight ratio), heat to 140-150°C for initial reaction, then maintain at 170-200°C for 2-8 hours.
- Cool reaction mixture to 100-120°C, neutralize with sodium carbonate solution, and perform multiple extractions using organic solvents like ethyl acetate or petroleum ether.
- Dry the combined organic extracts, decolorize with activated carbon, and perform vacuum distillation at 20-80°C to isolate the pure Dimer Diamide product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this urea-based amidation technology offers profound advantages in terms of cost stability and supply chain resilience. By eliminating the dependency on precious metal catalysts like Raney nickel and avoiding the need for high-pressure hydrogenation infrastructure, manufacturers can significantly reduce both fixed capital investment and variable operating costs. The raw materials involved, specifically dimerized fatty acids and urea, are commodity chemicals with robust global supply networks, insulating production from the volatility often seen with specialized reagents. This shift towards simpler, atmospheric pressure chemistry translates directly into lower manufacturing overheads and reduced risk premiums, allowing for more competitive pricing structures in the final DDI market without compromising on product quality or performance metrics.
- Cost Reduction in Manufacturing: The elimination of high-pressure hydrogenation steps removes the need for expensive autoclaves and specialized safety systems, leading to substantial capital expenditure savings. Furthermore, the use of urea as a nitrogen source is markedly cheaper than the multi-step nitrile route, driving down the raw material cost per kilogram of the final intermediate. The simplified purification process also reduces solvent consumption and waste disposal costs, contributing to a leaner and more economically efficient production model that enhances overall margin potential for downstream users.
- Enhanced Supply Chain Reliability: Relying on widely available commodity feedstocks rather than specialized catalysts or hazardous gases ensures a more stable and continuous supply of the Dimer Diamide intermediate. The robustness of the thermal reaction conditions means that production is less susceptible to interruptions caused by equipment maintenance or regulatory inspections associated with high-risk chemical processes. This reliability is critical for long-term contracts in the aerospace and defense sectors, where consistency of supply is often prioritized over marginal price fluctuations, ensuring that customers receive their orders on time without unexpected delays.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing standard batch reactor technology that can be easily expanded from pilot scale to multi-ton commercial production without significant re-engineering. The absence of heavy metal catalysts simplifies wastewater treatment and reduces the environmental burden of the manufacturing facility, aligning with increasingly stringent global environmental regulations. This green chemistry profile not only mitigates regulatory risk but also enhances the brand value of the final product for eco-conscious customers in the specialty chemicals market who prioritize sustainable sourcing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Dimer Diamide as described in the patent literature. These insights are derived directly from the experimental data and process descriptions provided in CN101648885A, offering a transparent view into the feasibility and benefits of this synthetic route. Understanding these details is essential for technical teams evaluating the integration of this intermediate into their existing formulation workflows or supply chains.
Q: What are the primary advantages of the urea amidation method over traditional dinitrile routes?
A: The urea amidation method eliminates the need for hazardous high-pressure hydrogenation and expensive Raney nickel catalysts required in the dinitrile route. It operates at atmospheric pressure with simpler equipment, significantly reducing capital expenditure and safety risks while maintaining high yields above 80%.
Q: How does this Dimer Diamide intermediate contribute to propellant performance?
A: As a precursor to Dimer Acid Diisocyanate (DDI), this intermediate enables the production of curing agents with lower N=C=O reactivity. This results in extended pot life for hydroxyl-terminated polybutadiene (HTPB) propellant slurries and improved mechanical properties in the final composite solid propellant.
Q: Is the purification process scalable for commercial production?
A: Yes, the purification involves standard unit operations such as liquid-liquid extraction, drying, and vacuum distillation. These processes are easily scalable from pilot plants to multi-ton reactors without requiring specialized or exotic separation technologies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dimer Diamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of advanced materials and propellant systems. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering Dimer Diamide and related fine chemicals with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify every batch. Our capability to adapt and optimize synthetic routes allows us to meet the unique demands of global clients, providing a level of technical support and supply security that is unmatched in the industry.
We invite you to collaborate with us to explore how this innovative synthesis route can enhance your product portfolio and reduce your overall manufacturing costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized production capabilities can serve as a cornerstone for your supply chain strategy, ensuring you have access to reliable, high-performance chemical solutions.
