Advanced Synthesis of 4-Amino-2,6-Dichloropyridine via Safe Curtius Rearrangement for Commercial Scale-up
Introduction to Advanced Pyridine Functionalization
The pharmaceutical and agrochemical industries constantly demand robust synthetic routes for halogenated heterocycles, specifically 4-amino-2,6-dichloropyridine, which serves as a critical building block for bioactive molecules such as 1H-pyrrolo[3,2-b]pyridines. A recent technological breakthrough detailed in patent CN111960996A introduces a refined preparation method that addresses long-standing safety and efficiency challenges associated with traditional Curtius rearrangements. This innovation leverages 2,6-dihydroxyisonicotinic acid as a commercially accessible starting material, transforming it through a sequence of chlorination, hydrazinolysis, and controlled rearrangement. By eliminating the need for hazardous isolated azide reagents, this protocol offers a pathway to high-purity intermediates that aligns with modern green chemistry principles and industrial safety standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-amino-2,6-dichloropyridine has relied on converting carboxylic acids to acyl chlorides followed by reaction with sodium azide (NaN3) to effect a Curtius rearrangement. As illustrated in the reaction scheme below, this classical approach involves significant operational hazards, including the generation of toxic carbon monoxide gas when using oxalyl chloride and the handling of highly explosive sodium azide.
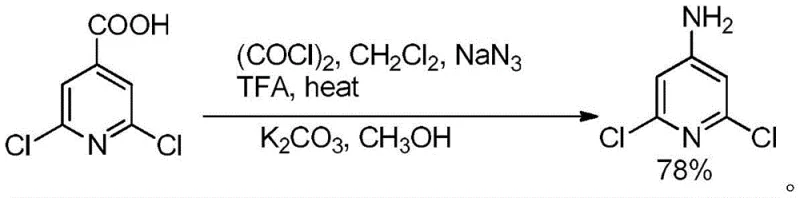
Furthermore, the subsequent hydrolysis steps often require strong alkaline conditions to remove protecting groups, which poses a severe risk of hydrolyzing the sensitive chlorine atoms on the pyridine ring. This side reaction not only reduces the overall yield but also complicates the impurity profile, necessitating costly and time-consuming purification processes that are detrimental to large-scale manufacturing economics.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent utilizes a hydrazide-mediated pathway that circumvents the direct use of explosive azide salts. By first converting the chlorinated pyridine carboxylic acid or ester into a hydrazide using hydrazine hydrate, the process creates a stable precursor that can be safely diazotized in situ. This strategic shift allows for the generation of the reactive acyl azide species under controlled acidic conditions, immediately followed by thermal rearrangement. The result is a streamlined process that maintains the integrity of the halogen substituents while delivering superior yields and simplified downstream processing.
Mechanistic Insights into Hydrazide-Mediated Curtius Rearrangement
The core of this synthetic advancement lies in the precise control of the diazotization and rearrangement steps. Initially, the 2,6-dichloroisonicotinic acid derivative reacts with hydrazine to form the corresponding hydrazide. Upon treatment with sodium nitrite and hydrochloric acid at low temperatures (0-20°C), the hydrazide undergoes diazotization to form the acyl azide intermediate. Unlike traditional methods where the azide might be isolated, here it is immediately subjected to heating (40-150°C), triggering the loss of nitrogen gas and the migration of the pyridyl group to form a highly reactive isocyanate species.
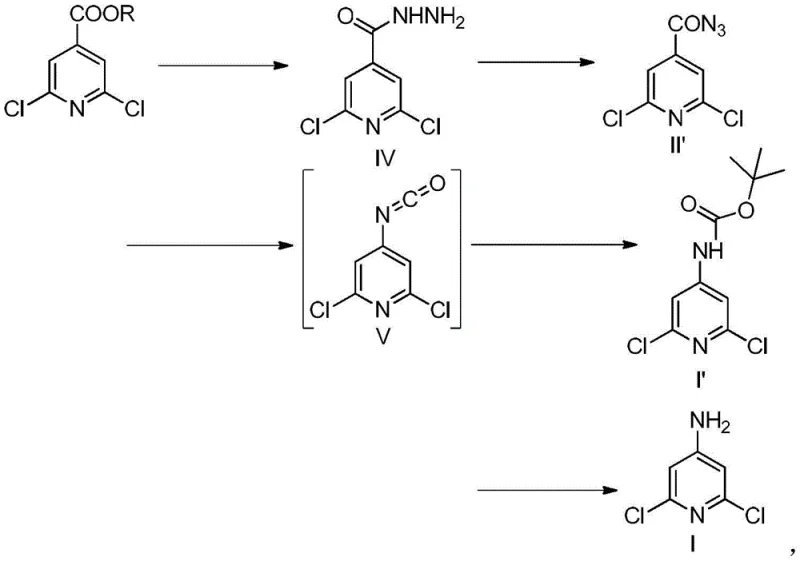
This isocyanate intermediate can then be trapped with an alcohol, such as tert-butanol, to form a carbamate-protected amine, or directly hydrolyzed to the free amine depending on the desired workflow. The use of a protecting group strategy, specifically the tert-butyloxycarbonyl (Boc) group, provides an additional layer of control, preventing potential side reactions during the rearrangement phase. The final deprotection step under mild acidic conditions cleanly reveals the target 4-amino-2,6-dichloropyridine with minimal degradation of the chloro-substituents, ensuring a high-purity final product suitable for sensitive pharmaceutical applications.
How to Synthesize 4-Amino-2,6-Dichloropyridine Efficiently
The synthesis protocol described in the patent offers a versatile framework adaptable to both batch and continuous flow processing. The procedure begins with the conversion of the starting acid to a hydrazide, followed by careful temperature-controlled diazotization. For organizations looking to implement this chemistry, the detailed standardized synthesis steps see the guide below.
- React 2,6-dichloroisonicotinic acid or its ester with hydrazine hydrate in an alcohol solvent to form the hydrazide intermediate.
- Perform diazotization on the hydrazide using sodium nitrite and hydrochloric acid at 0-20°C to generate the acyl azide in situ.
- Heat the azide to induce Curtius rearrangement into an isocyanate, then trap with tert-butanol or hydrolyze directly to obtain the amine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this hydrazide-based route represents a significant opportunity for cost reduction in pharmaceutical intermediate manufacturing. By removing the requirement for specialized safety infrastructure needed to handle bulk sodium azide, facilities can lower their operational overhead and insurance costs. Furthermore, the avoidance of toxic gas evolution simplifies waste treatment protocols, leading to substantial cost savings in environmental compliance and effluent management.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents like oxalyl chloride and sodium azide directly lowers the raw material bill of costs. Additionally, the high selectivity of the reaction minimizes the formation of de-chlorinated byproducts, which reduces the burden on purification teams and increases the overall mass efficiency of the plant.
- Enhanced Supply Chain Reliability: The starting materials, such as 2,6-dihydroxyisonicotinic acid and hydrazine hydrate, are commodity chemicals with robust global supply chains. This ensures consistent availability and reduces the risk of production stoppages due to raw material shortages, thereby stabilizing the supply of this critical intermediate for downstream API synthesis.
- Scalability and Environmental Compliance: The patent explicitly highlights the compatibility of this chemistry with microchannel reactor technology. This capability allows for the safe intensification of the exothermic diazotization and rearrangement steps, facilitating a smoother transition from laboratory scale to multi-ton commercial production while adhering to strict environmental regulations regarding hazardous waste.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and technical disclosures within the patent documentation to provide clarity on process feasibility and safety.
Q: Why is this synthesis method safer than traditional approaches?
A: This method avoids the use of isolated, explosive sodium azide (NaN3) by generating the acyl azide intermediate in situ from a hydrazide precursor, significantly enhancing operational safety.
Q: What yields and purity levels can be expected?
A: The patent data indicates that the process can achieve yields exceeding 95% with product purity greater than 99% without the need for secondary purification steps.
Q: Is this process suitable for continuous manufacturing?
A: Yes, the reaction sequence, particularly the diazotization and rearrangement steps, is compatible with microchannel reactor technology, allowing for precise temperature control and continuous production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Amino-2,6-Dichloropyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the drug development lifecycle. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex chemistries like the hydrazide-mediated Curtius rearrangement are executed with precision. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 4-amino-2,6-dichloropyridine meets the exacting standards required for GMP manufacturing.
We invite you to contact our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain. Request a Customized Cost-Saving Analysis today, and let us provide you with specific COA data and route feasibility assessments tailored to your project's unique requirements.
