Advanced Synthesis of 1-Phenyl-3-Methyl-5-Pyrazolone for High-Value Pharmaceutical Intermediates
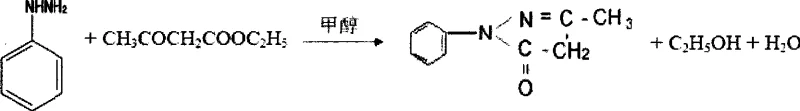
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to produce critical heterocyclic building blocks, and patent CN101367763A presents a significant breakthrough in the synthesis of 1-phenyl-3-methyl-5-pyrazolone. This compound serves as a vital precursor for antipyretic analgesic medicines, dye intermediates, and various agrochemical applications, yet its historical production has been plagued by environmental concerns and quality inconsistencies. The disclosed technology introduces a refined condensation strategy that utilizes a precise pH-controlled environment within a methanol solvent system, effectively bypassing the need for hazardous diazonium salt pre-preparation. By adjusting the reaction medium to a weakly acidic range of pH 5.0 to 6.5 using hydrochloric acid, the process facilitates a direct and high-yielding coupling between phenylhydrazine and methyl acetoacetate. This approach not only simplifies the operational workflow but also ensures the final product exhibits superior physical properties, specifically transforming what was traditionally a yellowish crude material into a high-purity white crystalline solid. For R&D directors and procurement specialists alike, this represents a pivotal shift towards greener chemistry that does not compromise on the stringent purity specifications required for downstream drug synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of 1-phenyl-3-methyl-5-pyrazolone has relied heavily on the ketene dimer method, which involves esterification under sulfuric acid catalysis. This legacy technique is notoriously inefficient from an environmental standpoint, as it consumes vast quantities of strong acids and generates substantial volumes of acid-bearing wastewater and noxious waste gases, creating a heavy burden on waste treatment facilities. Alternatively, existing phenylhydrazine-based routes often necessitate the complex pre-formation of phenylhydrazine hydrochloride diazonium salts, a procedure that requires rigorous cryogenic cooling followed by heating, thereby increasing energy consumption and operational complexity. Furthermore, these traditional pathways frequently employ auxiliary reagents such as sodium nitrite and sulfur dioxide, which contribute to high levels of pollution and pose significant safety risks in a manufacturing setting. Perhaps most critically for quality control, these older methods typically yield products that are flaxen or yellow in color, indicating the presence of conjugated impurities that can interfere with subsequent synthetic steps in API manufacturing. The combination of low atom economy, high pollution potential, and suboptimal product aesthetics has long been a bottleneck for supply chain managers seeking reliable sources of this key intermediate.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the methodology described in CN101367763A streamlines the synthesis into a cohesive, four-step sequence that prioritizes both efficiency and product quality. The core innovation lies in the direct adjustment of the phenylhydrazine methanol solution to a specific weak acidity, which activates the nucleophile sufficiently for condensation without triggering the decomposition or side reactions associated with stronger acidic conditions. By eliminating the separate diazonium salt preparation stage, the process removes the need for hazardous nitrosating agents and the energy-intensive temperature cycling previously required. The reaction proceeds smoothly under reflux conditions where methyl acetoacetate is added dropwise, ensuring controlled exothermicity and maximizing conversion rates. Following the initial condensation, the removal of methanol and subsequent neutralization allows for a secondary thermal treatment that drives the cyclization to completion. This refined approach not only boosts the overall yield to impressive levels exceeding 96% but also fundamentally alters the impurity profile, setting the stage for the production of pristine white crystals rather than the discolored materials typical of prior art.
Mechanistic Insights into Acid-Catalyzed Condensation and Cyclization
The chemical transformation at the heart of this process is a classic Knorr pyrazolone synthesis, yet its execution here is optimized through precise kinetic control and solvent management. The reaction initiates with the protonation of the carbonyl oxygen in the methyl acetoacetate or the activation of the phenylhydrazine nitrogen species within the pH 5.0-6.5 window, facilitating a nucleophilic attack that forms the hydrazone intermediate. Maintaining the temperature between 40°C and 90°C during the dropwise addition is critical; it provides sufficient thermal energy to overcome the activation barrier for condensation while preventing the thermal degradation of the sensitive hydrazine moiety. As the reaction progresses, the elimination of methanol (or ethanol depending on the specific ester used, though methanol is preferred here) drives the equilibrium forward according to Le Chatelier's principle. The subsequent step involves raising the pH to neutrality and applying heat at 60-80°C, which promotes the intramolecular cyclization and dehydration necessary to close the five-membered pyrazolone ring. This two-stage thermal profile ensures that the reaction mixture remains homogeneous during the initial coupling and then transitions effectively to the precipitation phase upon cooling.
Beyond the primary reaction mechanics, the purification strategy employed in this patent is equally sophisticated in its ability to manage the impurity spectrum. The inclusion of a recrystallization step utilizing activated carbon (GAC) under hot reflux conditions acts as a powerful adsorption mechanism for removing trace colored impurities and organic byproducts that typically cause yellowing. Whether using pure methanol or a mixed solvent system such as methanol-acetone or methanol-ethyl acetate, the solubility differential between the desired pyrazolone and the impurities is exploited to achieve high purity. The hot filtration removes the carbon along with the adsorbed contaminants, while the subsequent cooling induces the formation of a well-defined crystal lattice. This results in a final product with a melting point of 127-127.6°C and purity levels consistently above 99%, demonstrating that the mechanistic control extends beyond mere bond formation to include comprehensive molecular sorting. For R&D teams, this level of control over the solid-state properties is invaluable, as it reduces the need for extensive downstream polishing and ensures compatibility with sensitive catalytic steps in later stages of drug synthesis.
How to Synthesize 1-Phenyl-3-Methyl-5-Pyrazolone Efficiently
Implementing this synthesis route in a pilot or production facility requires strict adherence to the parameter windows defined in the patent to ensure reproducibility and safety. The process begins with the preparation of the reaction vessel, where phenylhydrazine is dissolved in methanol and carefully titrated with hydrochloric acid to reach the target pH range before any heating occurs. Once the weakly acidic environment is established, the methyl acetoacetate is introduced slowly over a period of one to two hours to manage the reaction exotherm, followed by a sustained reflux period to maximize conversion. After the initial reaction phase, the solvent is recovered via distillation, a step that not only isolates the crude product but also allows for the recycling of methanol, contributing to the overall economic viability of the process. The crude solid is then subjected to the critical purification sequence involving hot dissolution and activated carbon treatment, which is the key differentiator for achieving the desired white crystal appearance.
- Adjust the pH of a phenylhydrazine methanol solution to 5.0-6.5 using hydrochloric acid, then dropwise add methyl acetoacetate while maintaining temperature between 40-90°C for reflux.
- Distill off the methanol solvent, adjust the reaction mixture pH to neutral (7.0), and continue stirring with reflux at 60-80°C to complete cyclization.
- Cool the solution to crystallize the crude product, filter, and purify via hot dissolution in methanol or mixed solvents with activated carbon treatment followed by recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis technology offers tangible benefits that extend far beyond simple chemical yield improvements. The elimination of the diazonium salt preparation step removes the requirement for storing and handling hazardous reagents like sodium nitrite and sulfur dioxide, thereby reducing regulatory compliance costs and insurance premiums associated with dangerous goods. Furthermore, the simplified workflow reduces the total batch cycle time, allowing manufacturing facilities to increase throughput without the need for additional capital investment in reactor capacity. The ability to recover and reuse methanol solvent significantly lowers the raw material consumption per kilogram of product, directly impacting the variable cost of goods sold. Additionally, the production of a white crystal product rather than a yellow one reduces the risk of batch rejection by downstream customers who have strict color specifications for their API intermediates, thus enhancing supply chain reliability and reducing the incidence of costly returns or reworks.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the removal of expensive and hazardous auxiliary reagents that were previously mandatory for diazonium salt formation. By operating in a weakly acidic methanol medium, the process avoids the corrosion issues associated with strong mineral acids like sulfuric acid, leading to extended equipment lifespan and reduced maintenance downtime. The high yield reported in the embodiments, consistently reaching above 96%, means that less raw material is wasted per unit of output, optimizing the atom economy of the production line. Moreover, the energy profile is more favorable as it eliminates the need for cryogenic cooling baths, relying instead on standard heating and reflux conditions that are cheaper to maintain in a large-scale plant. These factors combine to create a substantially lower cost base for the manufacture of this critical pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: From a logistics and planning perspective, the robustness of this synthetic route ensures a more stable supply of 1-phenyl-3-methyl-5-pyrazolone to the market. The reliance on readily available commodity chemicals like phenylhydrazine and methyl acetoacetate minimizes the risk of raw material shortages that can plague specialized reagent supply chains. The simplicity of the operation, involving standard unit processes like distillation and filtration, means that the technology can be easily transferred between different manufacturing sites or scaled up rapidly to meet surges in demand. The consistent production of high-purity white crystals reduces the variability in product quality, allowing customers to streamline their own incoming quality control processes and reduce safety stock levels. This predictability is crucial for maintaining continuous production schedules in the highly regulated pharmaceutical industry.
- Scalability and Environmental Compliance: As global regulations on chemical manufacturing become increasingly stringent, the environmental profile of this process offers a distinct competitive advantage. The significant reduction in acid waste and the absence of nitrosating agents mean that effluent treatment is simpler and less costly, aligning with modern green chemistry principles. The process is inherently scalable, as the heat transfer and mixing requirements are manageable even in large reactors, avoiding the hotspots and safety risks associated with highly exothermic diazonium reactions. This scalability ensures that suppliers can meet the growing global demand for pyrazolone derivatives without facing bottlenecks related to waste disposal capacity or safety permits. Consequently, this technology supports a sustainable supply chain that is resilient to regulatory changes and capable of long-term growth.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this specific synthesis technology for 1-phenyl-3-methyl-5-pyrazolone. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this intermediate into their broader supply networks.
Q: How does this synthesis method improve product color compared to traditional phenylhydrazine routes?
A: Traditional methods often result in yellowish crystals due to complex diazonium salt preparation involving sodium nitrite and sulfur dioxide. This novel process eliminates the diazonium step and incorporates an activated carbon reflux recrystallization, ensuring the final product is a high-quality white crystal with purity exceeding 99%.
Q: What are the environmental advantages of this pH-controlled methanol process?
A: Unlike the industrial ketene dimer method which generates significant acid-bearing wastewater and waste gas, this process operates in a weakly acidic methanol environment (pH 5.0-6.5) without requiring strong mineral acids like sulfuric acid in large quantities, drastically reducing hazardous waste generation and simplifying effluent treatment.
Q: Can this process be scaled for commercial production of pharmaceutical intermediates?
A: Yes, the process is designed for scalability. It utilizes standard unit operations such as dropwise addition, distillation, and recrystallization, avoiding complex cryogenic cooling or hazardous gas handling, making it highly suitable for commercial scale-up of complex heterocyclic intermediates in a GMP environment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Phenyl-3-Methyl-5-Pyrazolone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields and purity profiles demonstrated in patent CN101367763A can be reliably replicated on an industrial scale. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to verify that every batch of 1-phenyl-3-methyl-5-pyrazolone meets the exacting standards required for pharmaceutical applications. Our commitment to quality assurance means that we do not just supply chemicals; we deliver validated solutions that integrate seamlessly into your drug development and manufacturing pipelines.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our manufacturing efficiencies can translate into better margins for your final products. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your volume needs. Let us collaborate to build a more efficient, sustainable, and high-quality supply chain for your critical pharmaceutical intermediates.
