Advanced Iridium-Catalyzed Asymmetric Hydrogenation for Scalable Chiral Benzodiazepine Manufacturing
The pharmaceutical industry continuously seeks robust methodologies for constructing complex heterocyclic scaffolds, particularly those exhibiting potent biological activities such as anticancer and anti-inflammatory properties. A significant breakthrough in this domain is documented in patent CN103288829A, which discloses a highly efficient method for synthesizing chiral dihydro-5H-pyrrolo[2,1-c][1,4]-benzodiazepino-5-one derivatives via iridium-catalyzed asymmetric hydrogenation. This technology addresses a critical gap in the availability of optically pure seven-membered ring heterocycles, which are privileged structures in medicinal chemistry. The patented process leverages a sophisticated catalytic system involving an iridium precursor complexed with chiral diphosphine ligands, enabling the direct reduction of cyclic imines with exceptional stereocontrol. By operating under mild temperatures ranging from 20 to 50°C and moderate hydrogen pressures, this method offers a sustainable and atom-economical alternative to classical synthetic routes that often suffer from poor selectivity or require cumbersome resolution steps. For R&D directors and procurement specialists, understanding this technology is paramount as it represents a viable pathway for securing high-purity intermediates essential for next-generation therapeutic agents.
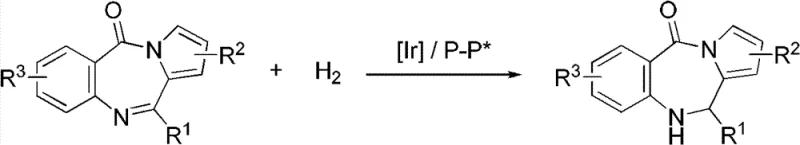
The limitations of conventional methods for synthesizing dihydro-pyrrolo-benzodiazepine derivatives are well-documented in chemical literature, often involving multi-step sequences that rely on stoichiometric chiral auxiliaries or kinetic resolutions. These traditional approaches frequently result in a maximum theoretical yield of 50% during resolution steps, generating substantial waste and increasing the cost of goods sold. Furthermore, classical cyclization strategies may require harsh acidic or basic conditions that compromise sensitive functional groups on the aromatic rings, leading to complex impurity profiles that are difficult to purge. In contrast, the novel approach detailed in the patent utilizes a homogeneous iridium catalytic system that directly targets the prochiral C=N bond within the seven-membered ring. This strategy bypasses the need for resolution entirely, theoretically allowing for 100% atom economy regarding the substrate conversion. The use of specific additives, such as morpholine trifluoroacetate or piperidine hydrochloride, plays a crucial role in activating the catalyst and stabilizing the transition state, thereby ensuring that the reaction proceeds with high turnover frequencies even at room temperature. This shift from stoichiometric chirality sources to catalytic asymmetry fundamentally alters the economic and environmental footprint of the manufacturing process.
Mechanistic Insights into Iridium-Catalyzed Asymmetric Hydrogenation
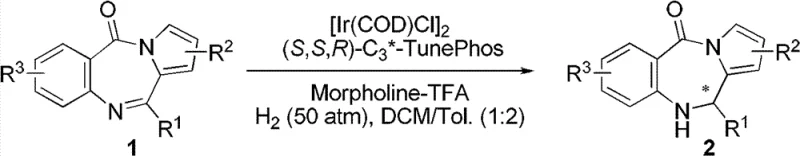
The core of this technological advancement lies in the precise orchestration of the catalytic cycle involving the iridium metal center and the chiral ligand environment. The active catalyst is generated in situ from a dimeric iridium precursor, specifically (1,5-cyclooctadiene)iridium chloride dimer, and a bulky chiral diphosphine ligand such as (S,S,R)-C3*-TunePhos. Mechanistically, the oxidative addition of hydrogen to the iridium center creates a dihydride species, which then coordinates with the nitrogen-containing heterocyclic substrate. The chiral pocket created by the TunePhos ligand dictates the facial selectivity of the hydride transfer to the imine carbon, effectively distinguishing between the two enantiotopic faces of the planar C=N bond. This steric differentiation is further enhanced by the presence of the acid additive, which likely protonates the imine nitrogen to form an iminium ion, increasing its electrophilicity and facilitating coordination to the metal center. The subsequent reductive elimination releases the chiral amine product and regenerates the active iridium species, allowing the cycle to continue with high efficiency. Understanding these mechanistic nuances is vital for process chemists aiming to optimize reaction parameters for specific substrate analogs.
Controlling the impurity profile in the synthesis of complex heterocycles is a primary concern for regulatory compliance, and this catalytic system offers inherent advantages in this regard. The high enantioselectivity reported, reaching up to 96% enantiomeric excess (ee), implies that the formation of the undesired enantiomer is kinetically suppressed, significantly reducing the burden on downstream purification processes like chiral chromatography. Moreover, the mild reaction conditions prevent thermal degradation or polymerization of the sensitive benzodiazepine core, which can occur under the elevated temperatures required by non-catalytic methods. The patent data indicates that side reactions are minimal, as evidenced by clean proton NMR spectra of the crude reaction mixtures, suggesting that the catalyst exhibits excellent chemoselectivity towards the cyclic imine over other reducible functionalities that might be present on the aromatic rings. This level of purity is critical for pharmaceutical applications where trace impurities can have profound toxicological implications. By minimizing the generation of diastereomers and structural isomers, the process ensures a consistent quality of the final intermediate, thereby streamlining the path to clinical trial materials.
How to Synthesize Chiral Dihydro-benzodiazepine Efficiently
The implementation of this synthesis route requires careful attention to the preparation of the catalyst and the control of reaction atmosphere to ensure reproducibility and safety. The protocol involves generating the active iridium complex under an inert nitrogen atmosphere to prevent oxidation of the phosphine ligands, followed by the introduction of the substrate and the acidic additive in a mixed solvent system of dichloromethane and toluene. The reaction is typically conducted in a stainless steel autoclave capable of withstanding hydrogen pressures up to 50 atm, although effective conversion is observed at lower pressures as well. Detailed operational procedures, including specific molar ratios of catalyst to substrate and precise workup methods involving column chromatography, are essential for achieving the reported yields and selectivity. For a comprehensive understanding of the standardized synthetic steps and safety protocols required for execution, please refer to the structured guide below.
- Prepare the catalyst precursor by mixing (1,5-cyclooctadiene)iridium chloride dimer with a chiral diphosphine ligand such as (S,S,R)-C3*-TunePhos in a dichloromethane-toluene solvent system.
- Combine the catalyst solution with the pyrrolo-[2,1-c][1,4]-benzodiazepine-5-one substrate and an additive like morpholine trifluoroacetate in a stainless steel autoclave.
- Pressurize the reactor with hydrogen gas to 13-50 atm and maintain the temperature between 20-50°C for 20-24 hours to achieve high enantiomeric excess.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this iridium-catalyzed hydrogenation technology presents significant opportunities for cost optimization and supply chain resilience in the manufacturing of pharmaceutical intermediates. The elimination of stoichiometric chiral resolving agents drastically reduces the raw material costs associated with producing single-enantiomer compounds, as the catalyst loading is typically low (substrate-to-catalyst ratio of 50:1 or better). Furthermore, the high atom economy of the hydrogenation reaction means that less waste is generated per kilogram of product, which translates to lower waste disposal fees and a reduced environmental compliance burden for manufacturing facilities. The use of common industrial solvents like toluene and dichloromethane ensures that the process can be easily integrated into existing infrastructure without requiring specialized equipment investments. These factors collectively contribute to a more sustainable and economically viable supply chain for high-value chiral building blocks.
- Cost Reduction in Manufacturing: The transition from resolution-based methods to direct asymmetric hydrogenation eliminates the inherent 50% yield loss associated with discarding the unwanted enantiomer, effectively doubling the theoretical output from the same amount of starting material. Additionally, the recovery and potential recycling of the precious iridium catalyst can further drive down the variable costs of production over time. By simplifying the synthetic sequence and reducing the number of unit operations required to achieve high optical purity, manufacturers can realize substantial savings in labor, energy, and solvent consumption. This economic efficiency makes the technology particularly attractive for the large-scale production of generic drug intermediates where margin compression is a constant challenge.
- Enhanced Supply Chain Reliability: The robustness of the catalytic system, which tolerates a variety of substituents on the aromatic rings including halogens and alkyl groups, ensures a consistent supply of diverse intermediates from a single platform technology. The mild reaction conditions reduce the risk of batch failures due to thermal runaways or decomposition, thereby improving the predictability of delivery schedules for downstream customers. Sourcing raw materials for this process is straightforward, as the substrates and ligands are commercially accessible or can be synthesized via established routes, mitigating the risk of supply disruptions. This reliability is crucial for pharmaceutical companies managing tight development timelines and requiring guaranteed continuity of supply for clinical and commercial batches.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated in autoclaves that mimic industrial high-pressure hydrogenation reactors, facilitating a smooth transition from gram-scale laboratory optimization to multi-kilogram pilot runs. The high selectivity of the reaction minimizes the formation of hazardous byproducts, aligning with green chemistry principles and simplifying the permitting process for new manufacturing lines. Reduced solvent usage and the potential for solvent recycling further enhance the environmental profile of the process, helping companies meet increasingly stringent corporate sustainability goals. This alignment with environmental, social, and governance (ESG) criteria adds intangible value to the supply chain, appealing to stakeholders who prioritize responsible manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric hydrogenation technology, derived from the specific details and advantages outlined in the patent documentation. These insights are intended to clarify the operational feasibility and strategic benefits for stakeholders evaluating this synthetic route for their pipeline projects. Understanding these aspects is essential for making informed decisions about process adoption and vendor selection.
Q: What is the primary advantage of using iridium catalysis for benzodiazepine synthesis?
A: The primary advantage is the ability to achieve high enantioselectivity (up to 96% ee) under relatively mild conditions (20-50°C), which avoids the harsh reagents often required in traditional resolution methods.
Q: Which ligands are most effective for this asymmetric hydrogenation?
A: Chiral diphosphine ligands such as (S,S,R)-C3*-TunePhos, (R)-SynPhos, and (R,R)-Me-DuPhos have demonstrated superior performance in generating the desired chiral center with high fidelity.
Q: Is this process scalable for industrial production?
A: Yes, the process utilizes standard hydrogenation equipment (autoclaves) and common solvents like toluene and dichloromethane, making it highly amenable to scale-up from laboratory to commercial tonnage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Benzodiazepine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-quality chiral intermediates for the development of life-saving medications, and we possess the technical expertise to bring complex synthetic routes like this iridium-catalyzed hydrogenation to fruition. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to verify the enantiomeric excess and chemical purity of every batch we produce. Our commitment to quality assurance guarantees that the chiral dihydro-benzodiazepine intermediates we supply meet the exacting standards required by global regulatory agencies.
We invite pharmaceutical partners to collaborate with us to leverage this advanced technology for their specific drug development programs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that evaluates the economic benefits of switching to this catalytic route for your specific target molecule. We encourage you to reach out today to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring that your supply chain is optimized for both performance and cost-efficiency.
