Scalable Metal-Free Synthesis of Alpha-Amino Acid Esters for Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking robust, environmentally benign methodologies for constructing essential molecular scaffolds, particularly C-N bonds which are ubiquitous in bioactive molecules. A significant breakthrough in this domain is detailed in patent CN111747858A, which discloses a novel, non-metal-catalyzed insertion reaction of sulfur ylides with aromatic amines. This technology represents a paradigm shift from traditional transition-metal-dependent protocols to a greener, aqueous-based system operating under remarkably mild conditions. By utilizing pure water as the solvent and avoiding hazardous oxidants or expensive metal catalysts, this method offers a sustainable pathway for synthesizing diverse α-amino acid ester derivatives. For R&D directors and process chemists, this innovation addresses critical pain points regarding impurity profiles and regulatory compliance, while simultaneously presenting compelling economic advantages for procurement and supply chain stakeholders seeking reliable pharmaceutical intermediate supplier partnerships.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of C-N bonds has relied heavily on transition-metal-catalyzed cross-coupling reactions, such as the classic Ullmann or Goldberg couplings, which necessitate the use of copper or palladium complexes. These traditional pathways often require pre-activated starting materials, typically aryl halides, which generate stoichiometric amounts of halide salt by-products, thereby increasing the burden on waste treatment facilities. Furthermore, alternative methods involving carbene N-H insertion or direct C-H amination frequently depend on unstable diazo compounds or expensive rhodium and iridium catalysts. The reliance on these precious metals not only inflates the raw material costs but also introduces the risk of heavy metal contamination in the final active pharmaceutical ingredients (APIs), necessitating complex and costly purification steps to meet stringent regulatory limits. Additionally, many of these conventional processes require harsh reaction conditions, including high temperatures and the use of volatile organic solvents, which pose significant safety hazards and environmental liabilities in a commercial manufacturing setting.
The Novel Approach
In stark contrast to these legacy technologies, the method disclosed in the referenced patent utilizes sulfur ylides as safe and effective surrogates for diazo compounds, enabling a direct N-H insertion reaction with aromatic amines. This innovative approach operates in pure water at a mild temperature of 35°C, completely eliminating the need for organic solvents and transition metal catalysts. The reaction is facilitated by simple acid additives, such as nitric acid or acetic acid, which activate the system without introducing toxic residues. As illustrated in the general reaction scheme below, this methodology allows for the efficient synthesis of various α-amino acid ester derivatives by simply varying the aromatic amine substrate. This simplicity translates directly into operational efficiency, as the absence of metal catalysts removes the need for specialized ligands and complex scavenging procedures, streamlining the entire production workflow.
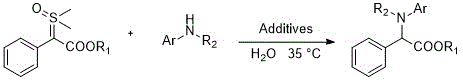
Mechanistic Insights into Non-Metal-Catalyzed N-H Insertion
The core of this technological advancement lies in the unique reactivity of sulfur ylides under acidic aqueous conditions. Unlike traditional carbene precursors that often require metal coordination to generate the reactive species, sulfur ylides in this system undergo protonation or activation by the acid additive to facilitate the nucleophilic attack on the electrophilic center. This mechanism bypasses the formation of free metal-carbene complexes, thereby avoiding the side reactions and decomposition pathways often associated with transition metal catalysis. The mild thermal energy provided at 35°C is sufficient to drive the insertion reaction to completion over a 24-hour period, suggesting a low activation energy barrier that is highly favorable for energy conservation in large-scale reactors. The use of water as a solvent further stabilizes the transition states through hydrogen bonding interactions, enhancing the selectivity of the N-H insertion over potential competing reactions such as cyclopropanation or epoxidation which might occur in non-polar media.
From an impurity control perspective, this metal-free mechanism offers a distinct advantage by inherently preventing the incorporation of heavy metals into the product matrix. In conventional syntheses, trace amounts of catalysts like palladium or copper can persist through multiple purification stages, requiring additional treatments with activated carbon or specialized resins to reduce levels to parts-per-million (ppm) specifications. By eliminating the source of these contaminants entirely, the new process simplifies the impurity profile, making it easier to characterize and control the quality of the final α-amino acid ester. Furthermore, the specificity of the reaction towards aromatic amines ensures that side products arising from solvent participation or reagent degradation are minimized. This high level of chemical fidelity is crucial for pharmaceutical applications where the identity and quantity of every impurity must be rigorously documented to satisfy global regulatory agencies.
How to Synthesize Alpha-Amino Acid Esters Efficiently
The practical implementation of this synthesis route is designed for ease of operation, requiring standard laboratory or plant equipment without the need for inert atmosphere techniques or high-pressure vessels. The protocol involves a straightforward mixing of the sulfur ylide, the chosen aromatic amine, and a catalytic amount of acid in water, followed by gentle heating. This accessibility makes it an ideal candidate for rapid process development and scale-up, allowing chemists to quickly generate libraries of derivatives for structure-activity relationship (SAR) studies. The detailed standardized synthesis steps for replicating this high-yielding transformation are provided in the guide below, ensuring consistent results across different batches and facilities.
- Combine sulfur ylide compound, aromatic amine, and an acid additive (such as nitric acid or acetic acid) in pure water within a clean reactor.
- Maintain the reaction mixture in an oil bath at a温和 temperature of 35°C for approximately 24 hours to facilitate the N-H insertion.
- Upon completion, remove the solvent under reduced pressure and purify the resulting residue using silica gel column chromatography to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free technology offers substantial strategic benefits that extend beyond mere technical feasibility. The elimination of precious metal catalysts directly impacts the cost structure of the manufacturing process, removing the volatility associated with the market prices of rhodium, palladium, or copper. Moreover, the use of water as the primary solvent drastically reduces the expenditure on organic solvents and the associated costs of solvent recovery or hazardous waste disposal. This shift towards greener chemistry not only lowers operational expenses but also enhances the company's sustainability profile, which is increasingly becoming a key criterion in vendor selection processes for major multinational pharmaceutical corporations.
- Cost Reduction in Manufacturing: The most significant economic driver of this technology is the complete removal of expensive transition metal catalysts and their配套 ligands from the bill of materials. In traditional C-N bond formation, catalyst loading can range from 1 mol% to 5 mol%, representing a substantial portion of the raw material cost, especially when using noble metals. By replacing these with inexpensive acid additives and stable sulfur ylides, the direct material cost is significantly reduced. Additionally, the simplified workup procedure, which avoids complex metal scavenging steps, reduces labor hours and consumable usage, leading to a lower overall cost of goods sold (COGS) for the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, specifically sulfur ylides and various aromatic amines, are commercially available and easy to source from multiple suppliers, reducing the risk of supply chain disruptions. Unlike specialized organometallic reagents that may have long lead times or limited availability, the precursors for this reaction are commodity chemicals with robust global supply networks. This abundance ensures a steady flow of materials for continuous production, allowing manufacturers to maintain high inventory turnover rates and meet tight delivery schedules for their clients without the fear of bottlenecking on critical reagents.
- Scalability and Environmental Compliance: The use of water as a reaction medium inherently improves the safety and scalability of the process, as it eliminates the fire hazards and toxicity concerns associated with large volumes of flammable organic solvents. This makes the technology highly adaptable for commercial scale-up of complex pharmaceutical intermediates, from pilot plant trials to multi-ton production campaigns. Furthermore, the reduction in hazardous waste generation aligns with increasingly stringent environmental regulations, minimizing the permitting hurdles and compliance costs associated with chemical manufacturing. This environmental compatibility ensures long-term operational continuity and protects the manufacturer from future regulatory shocks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this non-metal-catalyzed synthesis method. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Does this synthesis method leave heavy metal residues in the final product?
A: No, the process described in patent CN111747858A is explicitly metal-free. It utilizes sulfur ylides and aromatic amines without any transition metal catalysts, ensuring the final alpha-amino acid esters are free from toxic metal impurities.
Q: What are the environmental benefits of using water as a solvent in this reaction?
A: Using water eliminates the need for volatile organic compounds (VOCs) and hazardous organic solvents. This significantly reduces waste disposal costs and environmental impact, aligning with green chemistry principles for sustainable manufacturing.
Q: Can this method be scaled up for industrial production of API intermediates?
A: Yes, the mild reaction conditions (35°C) and the use of inexpensive, readily available raw materials make this process highly suitable for commercial scale-up. The simple workup procedure further enhances its feasibility for large-scale operations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Amino Acid Esters Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthesis technology for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent, high-quality supply regardless of volume requirements. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of alpha-amino acid esters meets the exacting standards required for drug substance manufacturing. We are committed to leveraging this green chemistry innovation to deliver superior value to our global partners.
We invite you to collaborate with us to explore how this efficient synthesis route can optimize your supply chain and reduce your overall manufacturing costs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs, demonstrating the tangible economic benefits of switching to this metal-free protocol. Please contact our technical procurement team today to request specific COA data for our available intermediates and to discuss detailed route feasibility assessments for your upcoming development programs.
