Advanced One-Pot Synthesis of N-cyclohexyl-2-aminoethanesulfonic acid for Commercial Scale Production
Introduction to Next-Generation CHES Manufacturing
The landscape of biochemical buffer manufacturing is undergoing a significant transformation driven by the need for cost-effective and high-purity intermediates. Patent CN112479938A introduces a groundbreaking preparation method for N-cyclohexyl-2-aminoethanesulfonic acid (CHES), a critical zwitterionic buffer widely used in protein stabilization and electrophoresis due to its biocompatibility and lack of metal interaction. This innovation shifts the paradigm from complex, multi-step syntheses relying on expensive precursors to a streamlined, one-pot process utilizing readily available commodity chemicals. By leveraging cyclohexylamine, 1,2-dichloroethane, and sodium sulfite, this technology addresses the longstanding economic and operational bottlenecks in the supply chain of high-performance buffering agents.
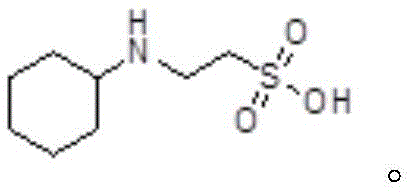
The strategic value of this patent lies in its ability to deliver product purity exceeding 99 percent while drastically simplifying the operational workflow. For R&D directors and procurement managers alike, this represents a dual opportunity: securing a reliable source of high-specification material while simultaneously driving down the cost basis through raw material optimization. The method eliminates the need for harsh reaction conditions and difficult-to-source reagents, positioning it as a superior alternative for the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
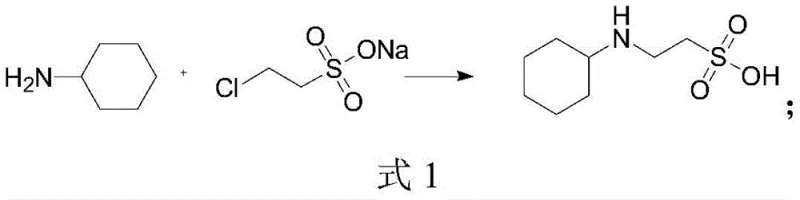
Historically, the industrial production of N-cyclohexyl-2-aminoethanesulfonic acid has been plagued by inefficiencies inherent in traditional synthetic routes. One common approach involves the reaction of cyclohexylamine with 2-chloroethyl sodium sulfonate, as depicted in the reaction scheme below. While conceptually straightforward, this pathway suffers from high raw material costs and relatively low yields. More critically, the reduction steps often associated with generating the necessary precursors introduce significant impurity profiles that complicate downstream purification and compromise the final product quality required for sensitive biological applications.
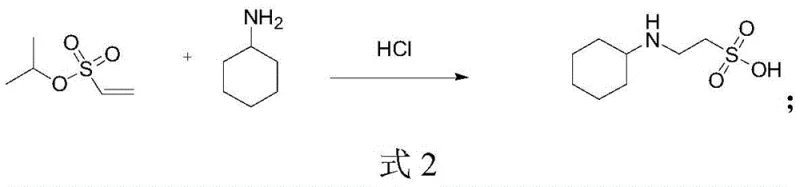
Another reported method utilizes isopropyl vinylsulfonate reacting with cyclohexylamine, followed by addition and hydrolysis steps. This route, illustrated in the following formula, presents even more severe logistical challenges. The precursor, isopropyl vinylsulfonate, is not only expensive but also difficult to procure in bulk quantities, creating a fragile supply chain vulnerable to disruptions. Furthermore, the multi-step nature of this synthesis extends reaction times and requires harsh conditions, increasing energy consumption and safety risks, which are untenable for modern sustainable manufacturing practices.
The Novel Approach
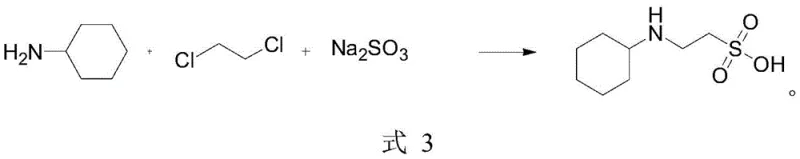
In stark contrast to these legacy methods, the invention disclosed in CN112479938A proposes a robust, one-pot synthesis that fundamentally restructures the production economics. By reacting cyclohexylamine directly with 1,2-dichloroethane and sodium sulfite in the presence of a copper powder catalyst, the process achieves a direct construction of the sulfonic acid backbone. This approach, shown in the reaction scheme below, bypasses the need for pre-functionalized sulfonate esters entirely. The reaction proceeds under mild reflux conditions (60-70°C), ensuring safety and ease of control while delivering a total yield exceeding 85 percent.
The operational simplicity of this novel approach cannot be overstated. It consolidates what were previously multiple unit operations into a single reactor vessel, significantly reducing capital expenditure requirements and floor space. The use of a water-ethanol mixed solvent system further enhances the green chemistry profile of the process, facilitating easier product isolation through crystallization. For supply chain heads, this translates to a more resilient manufacturing process that is less dependent on niche chemical suppliers and more adaptable to fluctuating market demands for high-purity biochemical buffers.
Mechanistic Insights into Copper-Catalyzed Sulfonation
The core of this technological advancement lies in the efficient nucleophilic substitution mechanism facilitated by the copper catalyst. In this system, sodium sulfite acts as the sulfur source, generating the sulfonate anion which attacks the electrophilic carbon of the 1,2-dichloroethane. The presence of copper powder is critical; it likely serves to activate the halide or stabilize intermediate species, thereby lowering the activation energy required for the substitution reaction. This catalytic effect allows the reaction to proceed rapidly at relatively low temperatures (60-70°C), minimizing thermal degradation of the sensitive amine functionality and preventing the formation of polymeric byproducts often seen in uncatalyzed alkylations.
Impurity control is meticulously managed through the stoichiometry and addition protocol. By controlling the dropwise addition of 1,2-dichloroethane over a period of approximately 2 hours, the concentration of the di-halide is kept low, favoring mono-substitution by the amine and sulfite rather than bis-alkylation or polymerization. Following the reaction, the removal of the solid copper catalyst via filtration ensures that no heavy metal residues contaminate the final API intermediate. Subsequent acidification to a pH of 5-7 precipitates the zwitterionic product in its purest form, effectively separating it from unreacted starting materials and inorganic salts, thus guaranteeing the stringent purity specifications demanded by the pharmaceutical industry.
How to Synthesize N-cyclohexyl-2-aminoethanesulfonic acid Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for implementing this technology at scale. The process begins with the preparation of a specific solvent matrix, typically a mixture of water and ethanol in a mass ratio of roughly 3:4, which optimizes the solubility of both the inorganic sulfite and the organic amine. Into this medium, cyclohexylamine, the copper catalyst, and sodium sulfite are introduced to form a homogeneous reactive phase. The key operational parameter is the controlled addition of 1,2-dichloroethane, which must be dosed slowly to maintain reaction selectivity and thermal stability throughout the exothermic substitution process.
- Prepare a mixed solvent of water and ethanol (ratio 3: 4), add cyclohexylamine, copper powder catalyst, and sodium sulfite, then heat to reflux.
- Slowly dropwise add 1,2-dichloroethane over approximately 2 hours while maintaining reflux temperature (60-70°C) for reaction.
- Filter to remove catalyst, distill off solvent, dissolve residue in absolute ethanol, adjust pH to 5-7 with HCl, and crystallize to obtain high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this synthesis route offers compelling economic and logistical benefits. The primary driver of value is the substitution of high-cost, specialty precursors with bulk commodity chemicals. 1,2-dichloroethane and sodium sulfite are produced on a massive global scale, ensuring consistent availability and shielding the manufacturer from the price volatility associated with fine chemical intermediates. This raw material shift fundamentally alters the cost structure, enabling significant cost reduction in pharmaceutical intermediate manufacturing without compromising on quality or yield.
- Cost Reduction in Manufacturing: The elimination of expensive reagents like isopropyl vinylsulfonate removes a major cost center from the bill of materials. Furthermore, the one-pot nature of the reaction reduces utility consumption by shortening overall processing time and eliminating the need for intermediate isolation steps. The ability to recover and reuse the ethanol-water solvent mixture through distillation further contributes to operational expenditure savings, creating a leaner and more profitable production model.
- Enhanced Supply Chain Reliability: Relying on commodity feedstocks significantly de-risks the supply chain. Unlike specialized sulfonate esters which may have limited supplier bases and long lead times, the raw materials for this process are readily accessible from multiple global sources. This diversification ensures business continuity and allows for rapid scaling of production capacity to meet surges in demand for CHES buffer, a critical component in biopharmaceutical formulation and diagnostic kits.
- Scalability and Environmental Compliance: The mild reaction conditions (60-70°C) and the use of a heterogeneous copper catalyst simplify the engineering requirements for scale-up. Standard stainless steel reactors are sufficient, avoiding the need for exotic alloys required for highly corrosive or high-pressure processes. Additionally, the process generates fewer hazardous byproducts compared to traditional routes, simplifying waste treatment and aligning with increasingly strict environmental regulations regarding industrial effluent and heavy metal discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and process descriptions provided in the patent documentation, offering clarity on yield expectations, purity profiles, and operational parameters for potential licensees or manufacturing partners.
Q: What are the primary advantages of the new synthesis route for CHES compared to traditional methods?
A: The new route utilizes inexpensive, commodity-grade raw materials like 1,2-dichloroethane and sodium sulfite instead of costly vinyl sulfonates. It operates as a one-pot synthesis at mild temperatures (60-70°C), eliminating complex multi-step procedures and significantly reducing production costs while achieving over 99% purity.
Q: How is the catalyst removed and solvent recovered in this process?
A: The process employs solid copper powder as a catalyst, which is easily removed via simple filtration after the reaction is complete. The water-ethanol solvent mixture is subsequently removed through reduced pressure distillation, allowing for potential recycling and reuse, thereby enhancing environmental compliance and operational efficiency.
Q: What represents the critical quality attribute for the final CHES product in this patent?
A: The critical quality attribute is the chromatographic purity, which consistently exceeds 99% (e.g., 99.2% to 99.5% in examples). This high purity is achieved through a controlled crystallization step involving pH adjustment to 5-7 using concentrated hydrochloric acid followed by rectification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-cyclohexyl-2-aminoethanesulfonic acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this advanced synthesis route for the global supply of biochemical buffers. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of N-cyclohexyl-2-aminoethanesulfonic acid meets the exacting standards required for research and therapeutic applications.
We invite pharmaceutical and biotech companies to leverage our technical expertise to optimize their supply chains. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your project benefits from the highest quality intermediates delivered with speed and reliability.
