Advanced Manufacturing of Vitamin B1 Phosphate Monoester: A Technical Breakthrough for Global Supply Chains
The global demand for high-quality nutritional intermediates continues to surge, driven by the expanding pharmaceutical and dietary supplement sectors. In this context, patent CN101654464B presents a pivotal advancement in the synthesis of Vitamin B1 phosphate monoester, a critical functional derivative used in treating various metabolic and neurological disorders. This intellectual property outlines a robust manufacturing pathway that transitions away from the harsh, inefficient conditions of legacy technologies toward a more streamlined, environmentally compatible process. By leveraging a specific phosphorylating agent system composed of phosphoric acid and phosphorus pentoxide, the inventors have achieved a synthesis route that not only enhances yield but also fundamentally alters the physical handling characteristics of the reaction mass. For procurement leaders and technical directors alike, understanding the nuances of this patent is essential for securing a reliable vitamin B1 derivative supplier capable of meeting stringent quality and volume requirements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of Vitamin B1 phosphate esters has been plagued by significant operational bottlenecks that hinder efficiency and scalability. Traditional protocols, such as those referenced in GB896089, necessitate the generation of polyphosphoric acid by heating ortho-phosphoric acid to extreme temperatures reaching 270°C. This high-energy requirement not only poses safety risks but also frequently results in the formation of thick, high-viscosity gel products that are notoriously difficult to stir, pump, or process in large reactors. Furthermore, these conventional routes often rely on prolonged hydrolysis periods extending up to seven days to break down mixed phosphate esters into the desired monoester, creating a massive bottleneck in production throughput. The reliance on expensive extraction agents like tri-octylamine and complex ion exchange columns for separation further exacerbates the cost structure, making cost reduction in pharmaceutical intermediate manufacturing a challenging objective for producers adhering to these outdated methodologies.
The Novel Approach
In stark contrast, the methodology disclosed in the patent introduces a sophisticated yet practical solution that circumvents these historical impediments through precise control of the phosphorylating environment. By generating the phosphorylating agent in situ at a much milder temperature range of 80°C to 150°C, the process successfully avoids the formation of the problematic high-viscosity gels that characterize older techniques. This fundamental shift allows for a homogeneous reaction mixture that facilitates efficient heat transfer and mass transport, thereby enabling a drastic simplification of the operational workflow. The subsequent hydrolysis and neutralization steps are executed rapidly, replacing the week-long waiting periods of the past with a continuous flow that can be completed in a matter of hours. This transition represents a paradigm shift for any organization seeking a reliable agrochemical intermediate supplier or pharma partner, as it guarantees a consistent supply of material without the unpredictability associated with gelation and extended reaction times.
Mechanistic Insights into Phosphoric Acid-P2O5 Catalyzed Esterification
The core of this technological breakthrough lies in the specific interaction between phosphoric acid and phosphorus pentoxide (P2O5) to generate a highly reactive phosphorylating species under controlled thermal conditions. When phosphorus pentoxide is introduced to phosphoric acid at temperatures between 80°C and 150°C, it creates a dynamic equilibrium of polyphosphoric acids that are sufficiently electrophilic to attack the hydroxyl group of the Vitamin B1 side chain without degrading the sensitive thiazole or pyrimidine rings. This careful balance of reactivity ensures that the esterification proceeds efficiently within a window of 30 minutes to 8 hours at 100°C to 140°C, maximizing the conversion to the target phosphate monoester while minimizing the formation of pyrophosphate byproducts. The mechanism avoids the chaotic polymerization often seen at higher temperatures, resulting in a cleaner reaction profile that is easier to monitor and control from a quality assurance perspective.
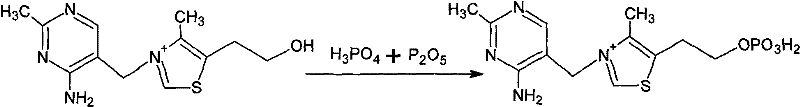
Following the esterification, the process employs a strategic hydrolysis and neutralization sequence that is critical for impurity control and product isolation. The addition of water hydrolyzes any remaining anhydride bonds, while the subsequent introduction of an alkali metal salt, such as calcium carbonate or calcium hydroxide, serves a dual purpose: it neutralizes the excess acid and precipitates insoluble calcium phosphates. This precipitation step is a masterstroke of process chemistry, as it effectively removes the bulk of the inorganic phosphate byproducts through simple filtration, leaving the desired Vitamin B1 phosphate monoester in the aqueous phase. This selective precipitation mechanism eliminates the need for costly and complex ion exchange chromatography, thereby reducing the potential for product loss and ensuring that the final high-purity thiamine monophosphate meets rigorous specifications for heavy metals and residual solvents.
How to Synthesize Vitamin B1 Phosphate Monoester Efficiently
Implementing this synthesis route requires a disciplined approach to reaction parameters and workup procedures to fully realize the benefits of yield and purity described in the patent literature. The process begins with the careful preparation of the phosphorylating agent, followed by the controlled addition of the Vitamin B1 substrate to manage the exotherm and gas evolution. Operators must maintain strict temperature profiles during the esterification phase to ensure complete conversion before proceeding to the quenching and neutralization stages. While the general outline provides a clear path forward, the specific details regarding stoichiometry, agitation rates, and crystallization conditions are vital for reproducibility. For a comprehensive understanding of the standardized operating procedures required to execute this synthesis safely and effectively, please refer to the detailed guide below.
- Prepare the phosphorylating agent by reacting phosphoric acid with phosphorus pentoxide at 80-150°C for 30 minutes to 2 hours.
- Add Vitamin B1 to the phosphorylating agent and conduct esterification at 100-140°C for 30 minutes to 8 hours to form the phosphate ester solution.
- Hydrolyze the mixture with water, neutralize with an alkali metal salt (such as calcium carbonate), and filter to obtain the aqueous solution.
- Concentrate the aqueous solution and perform recrystallization using organic solvents like ethyl acetate or ethanol to isolate the final pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis method translates directly into tangible improvements in cost structure and logistical reliability. By eliminating the need for extreme high-temperature equipment capable of sustaining 270°C, manufacturers can utilize standard glass-lined or stainless steel reactors, significantly lowering capital expenditure and maintenance costs. The removal of expensive extraction agents and ion exchange resins from the process flow further reduces the variable cost per kilogram, allowing for more competitive pricing strategies in the global market. Additionally, the ability to recycle alkali metal phosphates and organic solvents used in recrystallization creates a closed-loop system that minimizes waste disposal fees and aligns with increasingly strict environmental regulations, offering substantial cost savings without compromising on output quality.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived primarily from the simplification of the unit operations and the elimination of high-cost consumables. By avoiding the use of tri-octylamine and ion exchange columns, the process removes two of the most significant cost drivers associated with traditional purification methods. Furthermore, the shorter reaction cycles mean that the same production assets can generate significantly more batches per year, effectively increasing capacity without the need for new infrastructure. This efficiency gain allows suppliers to offer more attractive pricing tiers for long-term contracts, providing a distinct competitive advantage in the marketplace.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the unpredictability of complex chemical syntheses, particularly those prone to forming intractable gels or requiring week-long processing times. This novel approach mitigates those risks by establishing a robust, predictable reaction timeline that can be tightly scheduled. The use of readily available raw materials like phosphoric acid and phosphorus pentoxide ensures that supply is not dependent on niche reagents that might face market shortages. Consequently, partners can rely on consistent lead times and steady inventory levels, which is crucial for maintaining uninterrupted production schedules in downstream pharmaceutical applications.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to commercial production often reveals hidden challenges, but this method is inherently designed for industrial suitability. The absence of viscous gels ensures that mixing and heat transfer remain efficient even in large-scale reactors, preventing hot spots that could degrade the product. Moreover, the recycling of solvents and the generation of saleable or reusable phosphate salts demonstrate a commitment to green chemistry principles. This environmental stewardship not only reduces the ecological footprint but also simplifies regulatory compliance, making it easier to obtain necessary permits for commercial scale-up of complex vitamin derivatives.
Frequently Asked Questions (FAQ)
To assist technical teams and procurement specialists in evaluating this technology, we have compiled a set of frequently asked questions based on the specific technical disclosures and advantages found within the patent documentation. These inquiries address common concerns regarding process safety, product quality, and the feasibility of transitioning from legacy methods to this improved synthesis route. Understanding these details is key to making informed decisions about sourcing strategies and partnership opportunities in the fine chemical sector.
Q: How does this new synthesis method improve upon traditional high-temperature phosphorylation?
A: Traditional methods often require heating ortho-phosphoric acid to extreme temperatures around 270°C to generate polyphosphoric acid, which leads to the formation of viscous, difficult-to-handle gel products. The novel approach described in patent CN101654464B utilizes a milder phosphorylating agent generated at 80-150°C, effectively eliminating the formation of high-viscosity gels and significantly simplifying the downstream separation and purification processes.
Q: What are the primary cost drivers reduced in this manufacturing process?
A: The process eliminates the need for expensive extraction agents like tri-octylamine and avoids the capital-intensive ion exchange columns required in older separation techniques. Furthermore, the ability to recycle alkali metal phosphates and organic recrystallization solvents substantially lowers raw material consumption and waste disposal costs, driving down the overall cost of goods sold (COGS).
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the method is explicitly designed for industrial suitability. By avoiding the 7-day hydrolysis periods associated with legacy methods and utilizing standard neutralization and filtration steps, the production cycle is drastically shortened. The robust nature of the reaction conditions allows for seamless scaling from pilot batches to multi-ton commercial production without compromising yield or purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vitamin B1 Phosphate Monoester Supplier
At NINGBO INNO PHARMCHEM, we recognize that the theoretical advantages of a patent must be translated into practical, commercial reality to deliver true value to our clients. As a leading CDMO and manufacturer, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the efficiencies promised by patent CN101654464B are fully realized in our facilities. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize advanced analytical techniques to verify every batch. We understand that for a critical nutritional ingredient like Vitamin B1 phosphate monoester, consistency is paramount, and our state-of-the-art infrastructure is designed to deliver that consistency reliably, batch after batch.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain and reduce your overall manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the specific economic benefits applicable to your volume requirements. We encourage potential partners to contact us directly to索取 specific COA data and route feasibility assessments, allowing you to validate our capabilities against your exacting standards. Let us collaborate to bring high-quality, cost-effective Vitamin B1 derivatives to the global market with speed and precision.
