Advanced Synthesis of Hexadecyl 3,5-Di-Tert-Butyl-4-Hydroxybenzoate for Industrial Polymer Stabilization
The global demand for high-performance polymer additives continues to surge, driven by the need for durable materials in automotive, packaging, and construction sectors. At the forefront of this innovation is patent CN102391124A, which discloses a robust method for preparing hexadecyl 3,5-di-tert-butyl-4-hydroxybenzoate, a critical light stabilizer. This specific compound plays a pivotal role in preventing photo-oxidative degradation in polymers such as polypropylene and polyethylene, ensuring long-term material integrity. The patent outlines a novel synthetic route that leverages phosphorus oxychloride (POCl3) as a superior condensing agent, overcoming the limitations of traditional esterification methods. By shifting the paradigm from weak chlorinating agents to more reactive systems, this technology offers a pathway to significantly enhanced process efficiency and product quality. For industry leaders seeking a reliable polymer additive supplier, understanding the nuances of this patented chemistry is essential for securing a competitive advantage in the specialty chemicals market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of hindered phenol esters like hexadecyl 3,5-di-tert-butyl-4-hydroxybenzoate has relied heavily on direct esterification or the use of thionyl chloride (SOCl2) as an activating agent. However, these conventional pathways are fraught with significant inefficiencies that hinder large-scale commercial viability. Thionyl chloride, while common, often exhibits relatively weak chlorinating ability in this specific steric environment, necessitating the use of substantial excesses to drive the reaction to completion. This overuse not only inflates raw material costs but also complicates the downstream purification process due to the generation of acidic byproducts and unreacted reagents. Furthermore, traditional methods frequently suffer from prolonged reaction times and inconsistent yields, leading to batch-to-batch variability that is unacceptable for high-specification applications. The energy intensity associated with removing these excess reagents and purifying the crude product further erodes profit margins, making cost reduction in light stabilizer manufacturing a critical challenge for producers relying on outdated technologies.
The Novel Approach
In stark contrast, the methodology detailed in patent CN102391124A introduces a transformative approach by employing phosphorus oxychloride (POCl3) as the primary acyl chlorinating agent. This strategic substitution fundamentally alters the reaction kinetics, allowing for a much more efficient conversion of 3,5-di-tert-butyl-4-hydroxybenzoic acid and n-hexadecyl alcohol into the target ester. The new process operates under milder conditions, typically between 90°C and 105°C, which significantly reduces thermal stress on the reactants and minimizes the formation of degradation byproducts. By optimizing the mass ratio of reactants and solvent, specifically utilizing toluene, the reaction achieves a streamlined profile that simplifies the separation and purification stages. The result is a highly reproducible process that consistently delivers product yields exceeding 94%, a benchmark that far surpasses the capabilities of older thionyl chloride-based routes. This technological leap not only enhances operational throughput but also aligns perfectly with the requirements for commercial scale-up of complex polymer additives.
Mechanistic Insights into POCl3-Mediated Esterification
The core of this synthetic breakthrough lies in the mechanistic efficiency of phosphorus oxychloride in activating the carboxylic acid moiety of the hindered phenol. Unlike weaker activators, POCl3 facilitates the rapid formation of a mixed anhydride or acyl chloride intermediate in situ, which is highly susceptible to nucleophilic attack by the long-chain n-hexadecyl alcohol. This activation step is crucial because the steric bulk of the tert-butyl groups adjacent to the hydroxyl and carboxyl functionalities typically impedes reaction progress. The presence of POCl3 effectively overcomes this steric hindrance, ensuring that the condensation proceeds smoothly within the 3.5 to 7.5-hour window specified in the patent. Moreover, the reaction environment is carefully controlled to prevent side reactions, such as the chlorination of the aromatic ring or the dehydration of the alcohol, which are common pitfalls in aggressive esterification protocols. This precise control over the reaction mechanism is what allows manufacturers to achieve high-purity hexadecyl hydroxybenzoate without the need for extensive chromatographic purification.
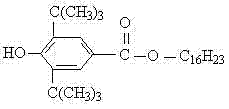
Following the primary condensation, the workup procedure is designed to maximize impurity removal while preserving the structural integrity of the light stabilizer. The addition of an aqueous alkali solution, such as sodium carbonate, serves a dual purpose: it neutralizes any residual acidic species generated during the reaction and hydrolyzes unreacted acylating agents. This step is critical for ensuring the final product meets the rigorous acidity specifications required for polymer processing, where even trace acids can catalyze degradation. Subsequent vacuum distillation recovers the toluene solvent for reuse, enhancing the overall atom economy of the process, while the final recrystallization from absolute ethanol polishes the product to a white powdered solid with a sharp melting point around 60°C. This comprehensive approach to impurity control ensures that the final material provides consistent UV protection and antioxidant performance in end-use applications.
How to Synthesize Hexadecyl 3,5-Di-Tert-Butyl-4-Hydroxybenzoate Efficiently
Implementing this patented synthesis requires precise adherence to the specified stoichiometric ratios and thermal profiles to replicate the high yields reported in the literature. The process begins with the careful charging of 3,5-di-tert-butyl-4-hydroxybenzoic acid, n-hexadecyl alcohol, and the acyl chlorinating agent into a reactor equipped with efficient agitation and reflux capabilities. Maintaining the reaction temperature strictly within the 90-105°C range is vital to balance reaction rate against potential thermal decomposition. Detailed standardized operating procedures regarding the specific addition rates of the alkali quench and the parameters for vacuum distillation are essential for operators to ensure safety and consistency. For a complete breakdown of the experimental protocol, please refer to the step-by-step guide below.
- Charge 3,5-di-tert-butyl-4-hydroxybenzoic acid, n-hexadecyl alcohol, and phosphorus oxychloride into a reactor with toluene solvent.
- Heat the mixture to 90-105°C and maintain condensation reaction for 3.5 to 7.5 hours under reflux.
- Quench the reaction with aqueous alkali, recover solvent via vacuum distillation, and recrystallize the crude product in absolute ethanol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this POCl3-mediated synthesis route offers tangible benefits that extend far beyond simple chemical yield improvements. The shift away from excessive reagent usage directly translates to a leaner bill of materials, reducing the volume of hazardous chemicals that must be sourced, stored, and handled on-site. This simplification of the input supply chain mitigates risks associated with raw material volatility and enhances overall operational resilience. Furthermore, the shortened reaction time and simplified workup procedure mean that production cycles are faster, allowing facilities to increase throughput without requiring significant capital investment in new reactor capacity. These factors combine to create a more agile manufacturing process capable of responding rapidly to fluctuating market demands for high-performance stabilizers.
- Cost Reduction in Manufacturing: The elimination of excessive thionyl chloride and the associated waste treatment costs results in substantial cost savings per kilogram of finished product. By utilizing a more efficient activating agent, the process minimizes the generation of acidic waste streams that require expensive neutralization and disposal, thereby lowering the total cost of ownership for the manufacturing facility. Additionally, the ability to recover and reuse the toluene solvent further drives down variable production costs, making the final light stabilizer more price-competitive in the global market. This economic efficiency is achieved without compromising on quality, ensuring that cost reductions do not come at the expense of product performance.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures a stable and continuous supply of critical polymer additives, reducing the risk of production stoppages due to quality failures. Because the process tolerates standard industrial-grade raw materials and does not rely on exotic or hard-to-source catalysts, supply chain bottlenecks are significantly minimized. The high yield consistency means that production planning becomes more predictable, allowing supply chain heads to optimize inventory levels and reduce the need for safety stock. This reliability is paramount for serving multinational clients who require just-in-time delivery of specialty chemicals to keep their own polymer production lines running smoothly.
- Scalability and Environmental Compliance: The mild reaction conditions and straightforward purification steps make this process inherently scalable from pilot plant to full commercial production volumes. The reduced environmental footprint, characterized by lower energy consumption and minimized chemical waste, aligns with increasingly stringent global environmental regulations. This compliance advantage future-proofs the manufacturing asset against tightening emissions standards and reduces the regulatory burden on the site. Consequently, companies adopting this technology position themselves as sustainable partners, appealing to end-users who prioritize green chemistry and responsible sourcing in their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specialized light stabilizer. These insights are derived directly from the technical specifications and experimental data provided in the underlying patent documentation. Understanding these details is crucial for R&D teams evaluating process feasibility and procurement teams assessing supplier capabilities. The answers reflect the consensus on best practices for handling and synthesizing hindered phenol esters in an industrial setting.
Q: Why is phosphorus oxychloride preferred over thionyl chloride in this synthesis?
A: Phosphorus oxychloride acts as a more efficient acyl chlorinating agent compared to thionyl chloride, which often exhibits weak chlorinating ability requiring excessive usage. The patented method utilizes POCl3 to drive the condensation reaction more effectively, resulting in significantly higher yields exceeding 94% while minimizing raw material waste.
Q: What are the critical purity specifications for this light stabilizer?
A: The process described in patent CN102391124A ensures high product purity through a specific workup procedure involving alkaline quenching and absolute ethanol recrystallization. This removes unreacted acids and alcohols, ensuring the final white powdered solid meets stringent quality standards required for high-performance polymer applications like polypropylene and polyethylene.
Q: How does this method impact environmental compliance in manufacturing?
A: By optimizing the stoichiometric ratio of reactants and utilizing a recoverable solvent system like toluene, the process drastically reduces chemical waste generation. The simplified separation and purification steps lower energy consumption during vacuum distillation, aligning with modern green chemistry principles for sustainable polymer additive production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hexadecyl 3,5-Di-Tert-Butyl-4-Hydroxybenzoate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory patent to commercial reality requires more than just chemical knowledge; it demands engineering excellence and unwavering commitment to quality. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of hexadecyl 3,5-di-tert-butyl-4-hydroxybenzoate meets the highest industry standards. We understand the critical nature of light stabilizers in protecting polymer assets, and our team is dedicated to delivering products that perform consistently in the most demanding applications.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. Our technical experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our advanced synthesis capabilities can become a cornerstone of your strategic sourcing strategy for high-performance polymer additives.
