Advanced Catalytic Synthesis of 2,4,5-Trifluoro-3-Methoxybenzoyl Chloride for Global Pharma Supply Chains
The pharmaceutical industry's relentless pursuit of efficient fluoroquinolone synthesis has found a significant breakthrough in the methodology disclosed in patent CN111635307B. This document details a robust, catalytic pathway for producing 2,4,5-trifluoro-3-methoxybenzoyl chloride, a critical building block for advanced antibiotics such as gatifloxacin, balofloxacin, and moxifloxacin. Unlike traditional methods that struggle with excessive reagent consumption and complex isolation steps, this novel approach leverages metal halide catalysis to streamline the transformation of N-methyltetrafluorophthalimide into the target acyl chloride. The technical implications are profound, offering a total molar yield of 82.5 percent and product purity exceeding 99.6 percent, thereby addressing the stringent quality demands of modern API manufacturing while simultaneously optimizing the economic footprint of the supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,4,5-trifluoro-3-methoxybenzoic acid, the precursor to the target chloride, has been plagued by operational inefficiencies and high material costs. As illustrated in prior art such as US 05380926, conventional routes often necessitate the isolation of intermediate hydroxy-acids before proceeding to subsequent steps, preventing continuous one-pot operations and increasing labor intensity. Furthermore, alternative pathways utilizing tetrafluorophthalic acid as a starting material involve prohibitively expensive raw materials and require substantial quantities of sodium hydroxide. This excessive alkali usage not only drives up raw material costs but also generates massive amounts of inorganic salt waste, complicating downstream purification and creating significant environmental disposal burdens for manufacturing facilities.
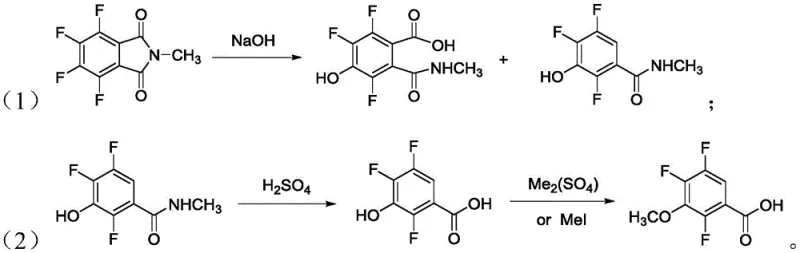
The Novel Approach
The innovative strategy presented in the patent data fundamentally reengineers this chemical landscape by introducing a metal halide-catalyzed hydrolysis that enables a seamless one-pot synthesis for the first three critical steps. By employing catalysts such as zinc chloride, aluminum trichloride, or ferric trichloride, the process accelerates the hydrolytic defluorination and hydroxylation reactions without requiring the isolation of intermediates. This continuity drastically shortens the overall process route and reduces reaction times to approximately 8-10 hours for the initial conversion. Moreover, the catalytic efficiency allows for a reduction in alkali consumption by more than 50 percent compared to state-of-the-art methods, directly translating to lower waste generation and simplified workup procedures that are highly attractive for industrial scale-up.
Mechanistic Insights into Metal Halide-Catalyzed Hydrolysis
The core of this technological advancement lies in the specific role of the metal halide catalyst during the initial hydrolysis and defluorination stages. In the absence of a catalyst, the amide bonds within the N-methyltetrafluorophthalimide structure are resistant to cleavage, leading to prolonged reaction times and incomplete conversions. The introduction of Lewis acidic metal halides facilitates the breaking of these amide bonds through the formation of reactive intermediates, as depicted in the reaction mechanism. This catalytic promotion ensures that the conversion to the 4-hydroxy salt-3,5,6-trifluorophthalate proceeds rapidly and comprehensively, establishing a solid foundation for the subsequent decarboxylation and methylation steps within the same reactor vessel.
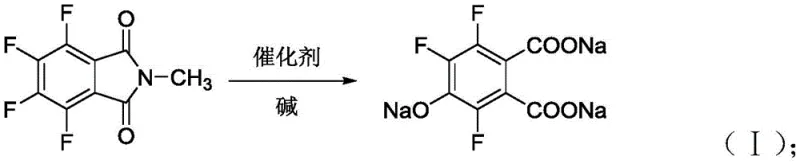
Beyond accelerating the primary reaction, the catalyst plays a pivotal role in impurity control, specifically by suppressing the formation of double-hydroxylated side products. Without catalytic guidance, the harsh alkaline conditions required for defluorination can lead to over-reaction, generating difficult-to-remove impurities that compromise the final API quality. The optimized catalytic environment moderates the reactivity, ensuring selective mono-hydroxylation and preserving the integrity of the fluorine substituents essential for the biological activity of the final fluoroquinolone drug. This precise control over the reaction trajectory results in a final product with a pH value greater than or equal to 1.5 and minimal levels of chlorinated by-products, demonstrating superior selectivity.
How to Synthesize 2,4,5-Trifluoro-3-Methoxybenzoyl Chloride Efficiently
Implementing this synthesis route requires careful attention to the sequential addition of reagents and temperature control to maximize the benefits of the one-pot design. The process begins with the catalytic hydrolysis, followed by in-situ acidification for decarboxylation, and concludes with methylation before the final acylation step. This integrated approach minimizes material transfer losses and exposure to atmospheric moisture, which is critical for maintaining the stability of the fluorinated intermediates. For a detailed breakdown of the specific operational parameters, reagent ratios, and safety protocols required to execute this synthesis effectively, please refer to the standardized guide below.
- Hydrolytic Defluorination: React N-methyltetrafluorophthalimide with alkali and a metal halide catalyst (e.g., ZnCl2) to form the sodium salt intermediate.
- Decarboxylation: Add acid to the system to induce decarboxylation, yielding 2,4,5-trifluoro-3-hydroxybenzoic acid.
- Methylation & Acidification: Under alkaline conditions, add a methylating agent followed by acid to obtain 2,4,5-trifluoro-3-methoxybenzoic acid.
- Acylation: React the benzoic acid derivative with thionyl chloride to finalize the synthesis of 2,4,5-trifluoro-3-methoxybenzoyl chloride.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalytic synthesis method offers compelling strategic advantages that extend beyond simple yield improvements. The ability to execute multiple synthetic transformations in a single reactor vessel significantly reduces the capital expenditure required for plant infrastructure, as fewer reactors and isolation units are needed to achieve the same output volume. Additionally, the drastic reduction in alkali consumption lowers the cost of goods sold (COGS) by minimizing the purchase of bulk caustic soda and reducing the logistical costs associated with the disposal of saline wastewater. These factors combine to create a more resilient and cost-effective supply chain capable of withstanding fluctuations in raw material pricing.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation steps and the significant decrease in alkali usage directly lower operational expenditures. By avoiding the generation of excessive inorganic salts, the facility reduces the burden on wastewater treatment systems, leading to substantial long-term savings in environmental compliance and waste management costs without compromising on throughput.
- Enhanced Supply Chain Reliability: Utilizing N-methyltetrafluorophthalimide as a starting material leverages a widely available and stable feedstock, reducing the risk of supply disruptions associated with more exotic or expensive precursors like tetrafluorophthalic acid. The robustness of the one-pot method ensures consistent batch-to-batch quality, fostering greater reliability in meeting the rigorous delivery schedules demanded by global pharmaceutical partners.
- Scalability and Environmental Compliance: The streamlined nature of this process facilitates easier commercial scale-up from pilot plants to multi-ton production lines. The reduced chemical footprint, characterized by lower reagent volumes and minimized side reactions, aligns perfectly with modern green chemistry principles, helping manufacturers meet increasingly strict environmental regulations while maintaining high production efficiency.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the process capabilities. Understanding these nuances is essential for R&D teams evaluating process feasibility and procurement teams assessing vendor qualifications for high-purity intermediate sourcing.
Q: How does the metal halide catalyst improve the synthesis efficiency?
A: The metal halide catalyst promotes the cleavage of the amide bond during hydrolysis, significantly accelerating the reaction rate and allowing for a reduction in alkali usage by over 50%, which minimizes salt waste.
Q: What is the purity profile of the final product using this method?
A: The process achieves a target product purity of greater than 99.6%, with specific impurities like 2,4-difluoro-5-chloro-3-methoxybenzoyl chloride controlled to less than 0.1%.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the first three steps can be completed in a single reaction vessel (one-pot method), which simplifies operation, reduces equipment requirements, and facilitates continuous commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4,5-Trifluoro-3-Methoxybenzoyl Chloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and scalable infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the efficiencies demonstrated in patent CN111635307B can be fully realized in a GMP-compliant manufacturing environment. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2,4,5-trifluoro-3-methoxybenzoyl chloride meets the exacting standards required for fluoroquinolone synthesis, providing our clients with peace of mind regarding quality and consistency.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this advanced catalytic technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your next project benefits from the highest standards of chemical manufacturing and supply chain security.
