Scalable Synthesis of O-Amino-P-Tert-Butyl Phenol via Low-Pressure Catalytic Hydrogenation for Global Supply Chains
The chemical landscape for producing high-value aromatic amines has evolved significantly with the disclosure of patent CN101696173B, which details a robust and environmentally benign synthesis route for o-amino-p-tert-butyl phenol. This compound serves as a pivotal building block in the fine chemical industry, particularly as a key intermediate for the synthesis of fluorescent whitening agents like OB and various pharmaceutical compounds. The traditional reliance on energy-intensive electrolytic reduction or pollution-heavy iron powder methods has created a bottleneck for manufacturers seeking sustainable and cost-effective supply chains. The patented methodology introduces a sophisticated catalytic hydrogenation protocol utilizing Raney Nickel in the presence of ammonia, fundamentally altering the economic and technical feasibility of large-scale production. By shifting the paradigm from stoichiometric reductants to catalytic systems, this innovation addresses the critical triad of modern chemical manufacturing: operational simplicity, environmental compliance, and superior product quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of o-amino-p-tert-butyl phenol has been plagued by significant technical and environmental drawbacks inherent to legacy technologies. The electrolytic reduction method, while capable of achieving moderate yields, suffers from exorbitant energy consumption due to the high electrical load required to drive the reduction of the nitro group, rendering it economically unviable in regions with high utility costs. Alternatively, the iron powder reduction method, though chemically straightforward, generates massive quantities of hazardous iron sludge waste that requires complex and costly disposal procedures, severely impacting the environmental footprint of the facility. Furthermore, the product purity obtained from iron powder reduction is often insufficient for high-end pharmaceutical applications, limiting its market utility to lower-grade dye intermediates. Even noble metal catalytic methods using Platinum or Palladium often necessitate high-pressure conditions exceeding 3.0 MPa and incur prohibitive catalyst costs, creating barriers to entry for mid-sized chemical producers.
The Novel Approach
The innovative process described in the patent data revolutionizes this synthesis by employing a Raney Nickel catalyst system augmented with ammonia under controlled hydrogenation conditions. This approach operates effectively at moderate pressures ranging from 1.0 to 3.0 MPa and temperatures between 90 and 150°C, significantly lowering the equipment specifications and safety risks associated with high-pressure reactors. The strategic inclusion of ammonia into the reaction matrix serves to stabilize the reaction environment, preventing the formation of undesirable byproducts and enhancing the overall selectivity towards the target amine. Utilizing common lower aliphatic alcohols such as methanol or ethanol as solvents further simplifies the downstream processing, allowing for easy recovery and recycling of materials. This method achieves yields exceeding 95% with product purity reaching 98%, demonstrating a clear superiority over traditional techniques in both efficiency and output quality.
Mechanistic Insights into Raney Nickel-Catalyzed Hydrogenation
The core of this synthetic breakthrough lies in the heterogeneous catalysis mechanism facilitated by the high surface area of the Raney Nickel alloy. In this system, molecular hydrogen is adsorbed onto the active nickel sites, where it dissociates into atomic hydrogen species that are highly reactive towards the nitro group of the substrate. The o-nitro-p-tert-butylphenol molecule coordinates with the catalyst surface, allowing for the stepwise reduction of the nitro functionality through nitroso and hydroxylamine intermediates before finally arriving at the stable amino group. The porous structure of the Raney Nickel provides an abundance of active centers, ensuring rapid reaction kinetics even at the relatively mild temperatures specified in the protocol. This efficient activation of hydrogen minimizes the residence time required in the reactor, thereby increasing the throughput capacity of the manufacturing plant without compromising conversion rates.
Crucially, the addition of ammonia plays a definitive role in controlling the impurity profile and maximizing the yield of the desired o-amino-p-tert-butyl phenol. In the absence of a basic modifier, the reduction of nitro-phenols can often lead to condensation side reactions, such as the formation of azo or azoxy compounds, which are difficult to separate and degrade the quality of the final product. Ammonia acts as a competitive base that neutralizes acidic species generated during the reaction and blocks the active sites responsible for these coupling pathways. 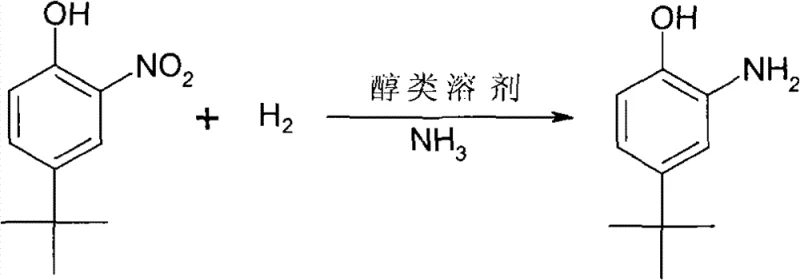
Furthermore, the basic environment provided by ammonia helps to maintain the catalyst activity by preventing the adsorption of acidic byproducts that could otherwise poison the nickel surface. This synergistic effect between the catalyst and the ammonia additive ensures a clean reaction trajectory, resulting in a crude product that requires minimal purification efforts. The ability to suppress these complex side reactions is what allows the process to achieve yields that are 3 to 5 percentage points higher than traditional hydrotreating methods, directly translating to better material utilization and reduced waste generation.
How to Synthesize O-Amino-P-Tert-Butyl Phenol Efficiently
Implementing this synthesis route in a production environment requires precise control over the stoichiometric ratios and reaction parameters to ensure consistent batch-to-batch quality. The process begins with the charging of the autoclave with the nitro precursor, the alcohol solvent, and the Raney Nickel catalyst, followed by the careful introduction of ammonia to establish the protective basic atmosphere. Detailed standardized operating procedures regarding the pressurization sequence, temperature ramping rates, and workup protocols are essential for operators to replicate the high yields reported in the patent literature safely. The following guide outlines the critical operational phases necessary to execute this transformation effectively on an industrial scale.
- Charge the autoclave with o-nitro-p-tert-butylphenol, a lower aliphatic alcohol solvent, and Raney Nickel catalyst.
- Add liquid ammonia to the mixture to suppress side reactions and improve selectivity.
- Pressurize with hydrogen to 1.0-3.0 MPa and heat to 90-150°C for 1-4 hours, then isolate product via filtration and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this Raney Nickel-based hydrogenation process offers tangible strategic benefits that extend beyond simple chemical conversion. The shift away from precious metal catalysts and stoichiometric reducing agents fundamentally alters the cost structure of the supply chain, removing dependencies on volatile commodity markets for platinum or palladium. Additionally, the elimination of heavy metal sludge waste streams drastically reduces the regulatory burden and disposal costs associated with environmental compliance, freeing up capital for other operational investments. The robustness of the reaction conditions also implies a higher reliability of supply, as the process is less susceptible to minor fluctuations in utility inputs compared to sensitive electrolytic methods.
- Cost Reduction in Manufacturing: The replacement of expensive noble metal catalysts with economical Raney Nickel results in a drastic reduction in direct material costs per kilogram of product. By eliminating the need for iron powder and the subsequent costly treatment of iron mud waste, the overall variable cost of production is significantly lowered, enhancing profit margins. The improved yield efficiency means that less raw material is required to produce the same amount of finished goods, optimizing the utilization of the o-nitro-p-tert-butylphenol feedstock. These cumulative savings create a highly competitive pricing structure that can be leveraged in negotiations with downstream pharmaceutical and agrochemical clients.
- Enhanced Supply Chain Reliability: The use of standard lower aliphatic alcohols as solvents ensures that the supply chain is not vulnerable to shortages of exotic or specialized reagents. The moderate pressure requirements allow the reaction to be performed in widely available standard autoclaves rather than specialized high-pressure vessels, increasing the number of qualified contract manufacturing organizations capable of producing this intermediate. This flexibility in manufacturing infrastructure reduces the risk of supply disruptions caused by equipment maintenance or capacity bottlenecks at single-source suppliers. Consequently, buyers can secure a more stable and continuous flow of high-purity intermediates essential for their own production schedules.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with reaction parameters that translate linearly from laboratory bench scale to multi-ton commercial production without significant re-engineering. The absence of toxic heavy metal waste and the reduction in energy consumption align perfectly with modern green chemistry principles and stringent environmental regulations. This eco-friendly profile simplifies the permitting process for new production lines and enhances the corporate social responsibility standing of the supply chain partners. Companies prioritizing sustainable sourcing will find this manufacturing route particularly attractive for long-term strategic partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of o-amino-p-tert-butyl phenol using this advanced catalytic method. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this intermediate into their synthesis pipelines. The answers provided are derived directly from the technical specifications and experimental data disclosed in the relevant patent documentation to ensure accuracy and reliability.
Q: Why is ammonia added during the hydrogenation of o-nitro-p-tert-butylphenol?
A: Ammonia acts as a critical modifier that suppresses side reactions such as coupling and aminolysis, significantly improving product selectivity and yield compared to traditional methods without ammonia.
Q: What are the advantages of using Raney Nickel over Platinum or Palladium catalysts?
A: Raney Nickel offers a substantial cost advantage over precious metals like Pt/C or Pd/C while maintaining high activity, allowing the reaction to proceed effectively at lower pressures (1.0-3.0 MPa).
Q: How does this method compare to the traditional iron powder reduction process?
A: Unlike the iron powder method which generates large amounts of toxic iron sludge and yields lower purity product unsuitable for pharma, this catalytic method is environmentally friendly and produces high-purity crystals.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable O-Amino-P-Tert-Butyl Phenol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of complex pharmaceutical and fine chemical syntheses. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of o-amino-p-tert-butyl phenol meets the exacting standards required for downstream applications. Our commitment to process excellence allows us to deliver consistent quality that supports your regulatory filings and product development timelines.
We invite you to collaborate with us to optimize your supply chain for this essential building block. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. Please contact us to request specific COA data and route feasibility assessments that demonstrate how our advanced manufacturing capabilities can enhance your operational efficiency. By partnering with us, you gain access to a reliable source of premium chemicals backed by deep technical expertise and a dedication to long-term supply security.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
