Advanced Synthetic Route for 3-Amino-7-Chloroquinoline Enables Scalable Pharmaceutical Manufacturing
Introduction to the Novel Synthetic Pathway
The pharmaceutical industry continuously seeks robust and scalable methodologies for constructing complex heterocyclic scaffolds, particularly those serving as critical building blocks for antimalarial therapeutics. Patent CN108484497B discloses a groundbreaking synthetic method for 3-amino-7-chloroquinoline, a pivotal intermediate in the development of next-generation antimalarial agents targeting the plasmodium parasite lifecycle. This technology represents a significant departure from traditional approaches by leveraging a concise five-step sequence that initiates from the inexpensive and commercially abundant 4-chloro-1-methyl-2-nitrobenzene. The process integrates formylation, reductive cyclization, hydrolysis, and a strategic Curtius rearrangement to install the crucial amino functionality at the 3-position with high fidelity. For R&D directors and procurement specialists, this innovation addresses long-standing challenges regarding raw material availability and process economics, offering a viable pathway for the reliable supply of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-amino-7-chloroquinoline has been hindered by a reliance on 7-chloro-3-nitroquinoline as the primary starting material, a compound that is neither commercially available nor cost-effective to produce in bulk. Conventional routes typically involve the high-temperature reduction of this nitro precursor, a process that is notoriously catalyst-intensive and prone to generating complex impurity profiles that are difficult to purge. The scarcity of the nitro starting material creates a bottleneck in the supply chain, forcing manufacturers to synthesize the precursor through multi-step sequences that further erode overall yield and increase production costs. Furthermore, the harsh conditions required for nitro reduction often compromise the structural integrity of sensitive functional groups, necessitating additional protection and deprotection steps that lengthen the manufacturing timeline. These cumulative inefficiencies render traditional methods unsuitable for the demands of modern, large-scale API manufacturing where cost reduction in pharmaceutical intermediate manufacturing is paramount.
The Novel Approach
In stark contrast, the methodology outlined in the patent utilizes a convergent strategy that constructs the quinoline core while simultaneously installing the amino group through a rearrangement mechanism. By starting with 4-chloro-1-methyl-2-nitrobenzene, the process bypasses the need for expensive specialty reagents, utilizing commodity chemicals that ensure consistent supply chain reliability. The sequence employs a mild formylation followed by a tin-mediated reductive cyclization to establish the heterocyclic ring system under controlled thermal conditions. Subsequent hydrolysis and Curtius rearrangement allow for the precise introduction of the nitrogen atom without the formation of hazardous byproducts associated with direct nitration or reduction. This streamlined approach not only enhances the total yield but also simplifies the workup procedures, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into the Multi-Step Transformation
The chemical elegance of this synthesis lies in its sequential manipulation of functional groups to achieve regioselective amination. The process begins with the formylation of the methyl group on the nitrobenzene ring using N,N-dimethylformamide dimethyl acetal (DMFDMA), followed by oxidative cleavage with sodium periodate to yield 4-chloro-2-nitrobenzaldehyde. This aldehyde then undergoes a reductive cyclization with ethyl 3,3-diethoxypropionate in the presence of stannous chloride, a reaction that simultaneously reduces the nitro group and closes the quinoline ring to form ethyl 7-chloro-3-quinolinecarboxylate. The ester is subsequently hydrolyzed under alkaline conditions to generate the free carboxylic acid, which serves as the substrate for the critical Curtius rearrangement. In this key step, activation with diphenyl phosphorazidate (DPPA) generates an acyl azide that thermally rearranges to an isocyanate, which is then trapped by tert-butanol to form the Boc-protected amine. Finally, acidic deprotection reveals the target 3-amino-7-chloroquinoline, completing the transformation with high atom economy.
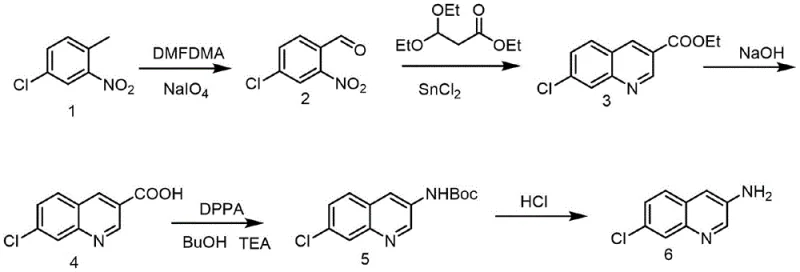
From an impurity control perspective, this route offers distinct advantages by avoiding the radical mechanisms often associated with high-temperature reductions. The use of the Boc protecting group during the rearrangement step acts as a temporary mask that prevents unwanted side reactions at the amino center, ensuring a cleaner reaction profile. The intermediate 3-tert-butoxycarbonylamino-7-chloroquinoline can be purified via standard crystallization or extraction techniques before the final deprotection, effectively acting as a purification checkpoint. This level of control is essential for meeting the stringent purity specifications required for downstream coupling reactions, such as the diazotization and fluorination steps needed to produce 7-chloro-3-fluoro-4-aminoquinoline. The mechanistic clarity of each step allows process chemists to fine-tune reaction parameters, such as the molar ratio of DPPA to acid (1:1 to 1:1.5), to maximize conversion and minimize waste.
How to Synthesize 3-Amino-7-Chloroquinoline Efficiently
Implementing this synthetic route requires careful attention to reaction stoichiometry and thermal management, particularly during the exothermic oxidation and rearrangement phases. The patent details a robust protocol where the initial formylation is conducted at elevated temperatures (120-140°C) to ensure complete conversion of the methyl group, followed by a controlled oxidative workup. The subsequent cyclization step utilizes ethanol as a green solvent, and the hydrolysis is performed under mild alkaline conditions to prevent degradation of the quinoline core. For the Curtius rearrangement, maintaining the temperature between 80-120°C is critical to drive the migration of the acyl group while preventing the decomposition of the azide intermediate. The detailed standardized synthesis steps below outline the precise operational parameters required to replicate this high-yielding process in a pilot or production environment.
- Perform formylation and oxidation on 4-chloro-1-methyl-2-nitrobenzene using DMFDMA and sodium periodate to generate 4-chloro-2-nitrobenzaldehyde.
- Execute reductive cyclization with ethyl 3,3-diethoxypropionate and stannous chloride to form the quinoline core structure.
- Hydrolyze the resulting ester under alkaline conditions to obtain 7-chloro-3-quinolinecarboxylic acid.
- Conduct Curtius rearrangement using diphenyl phosphorazidate (DPPA) and tert-butanol to introduce the protected amino group.
- Remove the tert-butyloxycarbonyl (Boc) protecting group using acid to yield the final 3-amino-7-chloroquinoline product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers substantial strategic benefits beyond mere technical feasibility. The shift away from non-commercial starting materials to commodity chemicals drastically reduces the risk of supply disruption, ensuring a continuous flow of raw materials even during market fluctuations. The elimination of expensive transition metal catalysts and the reduction in processing steps translate directly into lower manufacturing overheads, allowing for more competitive pricing structures in the global marketplace. Furthermore, the simplified workup procedures, which rely on filtration and extraction rather than resource-intensive chromatography, reduce solvent consumption and waste generation, aligning with modern environmental compliance standards.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the utilization of low-cost raw materials like 4-chloro-1-methyl-2-nitrobenzene and the avoidance of precious metal catalysts often required in cross-coupling alternatives. By streamlining the synthesis into five high-yielding steps, the overall material throughput is maximized, significantly reducing the cost per kilogram of the final intermediate. The ability to perform reactions in common solvents like ethanol and DMF further lowers operational expenses related to solvent recovery and disposal. Consequently, this route enables significant cost savings in API manufacturing without compromising on the quality or purity of the output.
- Enhanced Supply Chain Reliability: Dependence on obscure or custom-synthesized starting materials is a major vulnerability in pharmaceutical supply chains, but this method mitigates that risk by relying on widely available bulk chemicals. The robustness of the reaction conditions means that production is less susceptible to minor variations in raw material quality, ensuring consistent batch-to-batch performance. This stability allows for better inventory planning and shorter lead times for high-purity pharmaceutical intermediates, facilitating just-in-time delivery models for downstream API producers. The scalability of the process ensures that supply can be rapidly ramped up to meet surging demand for antimalarial therapies.
- Scalability and Environmental Compliance: The process design inherently supports large-scale production, with reaction conditions that are easily transferable from laboratory glassware to industrial reactors. The absence of highly toxic reagents and the generation of manageable byproducts simplify waste treatment protocols, reducing the environmental footprint of the manufacturing facility. Efficient solvent usage and the potential for solvent recycling contribute to a greener manufacturing profile, which is increasingly important for regulatory approval and corporate sustainability goals. This combination of scalability and environmental responsibility makes the technology a sustainable long-term solution for the production of complex quinoline derivatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. These insights are derived directly from the patent data and are intended to clarify the operational benefits and chemical rationale behind the new route. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this process for their specific supply chain needs.
Q: What are the primary advantages of this synthetic route over conventional nitro-reduction methods?
A: This novel route circumvents the reliance on 7-chloro-3-nitroquinoline, a non-commercial and costly reagent often requiring harsh reduction conditions. By utilizing readily available 4-chloro-1-methyl-2-nitrobenzene and employing a Curtius rearrangement strategy, the process achieves higher total yields and significantly simplifies post-treatment operations.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the process is explicitly designed for scalability, utilizing common industrial solvents like ethanol and DMF and avoiding highly toxic reagents. The operational simplicity, characterized by straightforward filtration and extraction steps rather than complex chromatography, ensures robust performance from kilogram to multi-ton scales.
Q: How does the Curtius rearrangement step impact the purity profile of the final intermediate?
A: The Curtius rearrangement facilitates a clean migration of the acyl group to the amine, minimizing side reactions common in direct amination strategies. This mechanistic pathway, combined with the crystallization-friendly nature of the Boc-protected intermediate, allows for stringent impurity control and high-purity specifications essential for API synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Amino-7-Chloroquinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving antimalarial medications. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 3-amino-7-chloroquinoline meets the exacting standards required for pharmaceutical applications. We are committed to delivering not just a product, but a comprehensive supply solution that enhances your R&D velocity and commercial success.
We invite you to engage with our technical procurement team to discuss how this advanced synthetic route can optimize your production costs and supply security. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume requirements. We encourage potential partners to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on real-world performance metrics and our proven track record in fine chemical synthesis.
