Advanced Manufacturing of 2-Chloro-6-Fluorobenzaldehyde: A Solid Superacid Catalytic Breakthrough for Commercial Scale-Up
The pharmaceutical and agrochemical industries are constantly seeking robust synthetic routes for critical intermediates such as 2-chloro-6-fluorobenzaldehyde, a key precursor for antibiotics like flucloxacillin and various high-efficiency pesticides. Recent intellectual property developments, specifically patent CN102617312B, have introduced a transformative methodology that replaces traditional corrosive liquid acid hydrolysis with a heterogeneous ferric solid superacid catalytic system. This innovation addresses long-standing challenges in reactor corrosion, environmental pollution, and yield optimization that have plagued conventional manufacturing processes for decades. By leveraging photo-chlorination followed by solid acid-catalyzed hydrolysis, this technology offers a pathway to achieve purity levels exceeding 99.7% while drastically simplifying the downstream purification workflow. For global supply chain leaders and R&D directors, understanding the mechanistic advantages of this solid-state catalysis is essential for evaluating next-generation sourcing strategies and ensuring long-term supply continuity for complex aromatic aldehydes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of 2-chloro-6-fluorobenzaldehyde has relied heavily on harsh chemical environments that pose significant operational and economic burdens. Traditional protocols typically involve the use of radical initiators such as benzoyl peroxide or diisopropyl azodicarboxylate to facilitate the initial chlorination of 2-chloro-6-fluorotoluene, followed by hydrolysis in concentrated 85% sulfuric acid at elevated temperatures around 90°C. While these methods can achieve yields of approximately 90%, they necessitate the use of specialized acid-resistant reactors and extensive safety measures due to the highly corrosive nature of hot concentrated sulfuric acid. Furthermore, the disposal of spent acid waste streams creates substantial environmental compliance costs and complicates the overall manufacturing footprint. The reliance on liquid strong acids also introduces risks of equipment degradation over time, leading to potential contamination issues and increased maintenance downtime that can disrupt supply chains for critical pharmaceutical intermediates.
The Novel Approach
In stark contrast to legacy methods, the novel approach detailed in the patent utilizes a combination of metal-halide lamp irradiation for chlorination and a ferric solid superacid (SO4 2-/Fe3O4) for the subsequent hydrolysis step. This shift from homogeneous liquid acid catalysis to heterogeneous solid acid catalysis represents a paradigm shift in process safety and efficiency. The reaction proceeds by first converting 2-chloro-6-fluorotoluene into mixed benzyl chlorides under illumination, followed by the addition of the solid superacid and controlled water dripping at temperatures between 100°C and 200°C. This methodology not only eliminates the hazards associated with transporting and storing large volumes of concentrated sulfuric acid but also enhances the reaction yield to approximately 95%. The use of a solid catalyst simplifies the separation process, as the catalyst can be easily managed without the complex neutralization and washing steps required for liquid acids, thereby streamlining the production of high-purity 2-chloro-6-fluorobenzaldehyde.
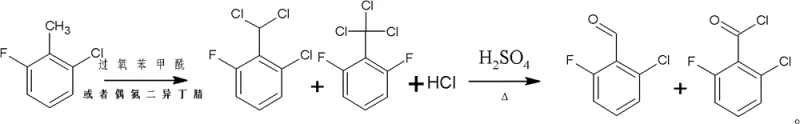
Mechanistic Insights into Photo-Chlorination and Solid Superacid Hydrolysis
The core of this technological advancement lies in the precise control of the chlorination degree and the unique activation properties of the ferric solid superacid. During the initial phase, the substrate 2-chloro-6-fluorotoluene undergoes free-radical chlorination driven by photon energy from a metal-halide lamp rather than thermal decomposition of chemical initiators. This photo-induced mechanism allows for a more uniform distribution of radical species, minimizing the formation of polychlorinated byproducts that often complicate purification. The process is meticulously monitored via gas chromatography to ensure that the content of the intermediate 2-chloro-6-fluorobenzyl chloride drops below 0.5% before proceeding, ensuring that the subsequent hydrolysis step operates on a optimized mixture of benzyl chlorides. This rigorous control point is critical for maintaining the structural integrity of the fluorine and chlorine substituents on the aromatic ring, preventing defluorination or dechlorination side reactions that would compromise the quality of the final API intermediate.
Following the chlorination phase, the introduction of the ferric solid superacid acts as a powerful Lewis acid catalyst that facilitates the hydrolysis of the benzyl chloride moieties into the corresponding aldehyde functionality. Unlike liquid sulfuric acid, which can promote sulfonation or charring of organic materials at high temperatures, the solid superacid provides active sites for water activation without excessive oxidative degradation. The reaction conditions involve dripping water into the mixture at temperatures ranging from 100°C to 200°C and maintaining this heat for approximately 4 hours, allowing for complete conversion of the dichloride and trichloride intermediates. The heterogeneity of the catalyst system means that post-reaction workup involves a simple phase separation after alkaline neutralization, rather than the dilution and extraction nightmares associated with spent sulfuric acid. This mechanistic elegance results in a cleaner crude product profile, reducing the load on the final vacuum distillation column and ensuring consistent batch-to-batch reproducibility.
How to Synthesize 2-Chloro-6-Fluorobenzaldehyde Efficiently
Implementing this advanced synthesis route requires careful attention to the transition between the photo-chlorination and hydrolysis stages to maximize yield and safety. The process begins with the illumination of the starting toluene derivative in the presence of chlorine gas, where the reaction temperature is maintained between 100°C and 200°C to ensure optimal radical generation without thermal runaway. Once the monitoring systems confirm the depletion of the mono-chlorinated intermediate, the system is purged with nitrogen to remove excess chlorine before the solid catalyst is introduced. Water is then added dropwise over a period of 2 to 4 hours, a critical parameter that controls the exotherm and ensures steady hydrolysis kinetics.
- Perform photo-chlorination on 2-chloro-6-fluorotoluene using a metal-halide lamp at 100-200°C until mono-benzyl chloride content drops below 0.5%.
- Add ferric solid superacid (SO4 2-/Fe3O4) and drip water at 100-200°C, maintaining temperature for approximately 4 hours to complete hydrolysis.
- Neutralize with alkali solution, separate the organic layer, and purify via vacuum distillation to obtain the final aldehyde product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this solid superacid technology translates directly into tangible operational efficiencies and risk mitigation strategies. The elimination of concentrated sulfuric acid from the process removes a major bottleneck related to hazardous material handling, storage requirements, and regulatory compliance, thereby simplifying the logistics of raw material sourcing. By switching to a solid catalyst system, manufacturers can utilize standard stainless steel reactors instead of expensive glass-lined or Hastelloy vessels designed to withstand extreme acid corrosion, leading to significant capital expenditure savings and reduced maintenance intervals. Furthermore, the improved yield profile and simplified workup procedure mean that less raw material is wasted per kilogram of finished product, enhancing overall resource efficiency and reducing the volume of chemical waste requiring treatment. These factors collectively contribute to a more resilient supply chain capable of delivering high-purity intermediates with greater consistency and lower total cost of ownership.
- Cost Reduction in Manufacturing: The transition away from liquid strong acids fundamentally alters the cost structure of production by removing the need for specialized corrosion-resistant infrastructure and expensive acid neutralization agents. Without the requirement for 85% sulfuric acid, facilities can avoid the high costs associated with acid-resistant linings and the continuous replacement of degraded equipment parts. Additionally, the solid catalyst system generates significantly less acidic wastewater, which drastically lowers the operational expenses related to effluent treatment and environmental compliance fees. The qualitative improvement in yield from approximately 90% to 95% further amplifies these savings by maximizing the output from every ton of starting material purchased, effectively lowering the unit cost of the final aldehyde without compromising on quality standards.
- Enhanced Supply Chain Reliability: Relying on solid catalysts and photo-chlorination reduces dependency on volatile liquid acid supply chains that can be subject to transportation restrictions and market fluctuations. The use of a metal-halide lamp for initiation removes the need for unstable radical initiators like benzoyl peroxide, which often require cold chain logistics and have limited shelf lives. This stability in raw material requirements ensures that production schedules are less likely to be disrupted by supplier delays or regulatory hurdles associated with hazardous chemical transport. Consequently, partners can expect more predictable lead times and a steadier flow of inventory, which is crucial for maintaining uninterrupted production lines for downstream antibiotics and agrochemical formulations.
- Scalability and Environmental Compliance: The inherent safety of the solid superacid process makes it exceptionally well-suited for scaling up from pilot batches to multi-ton commercial production without exponential increases in safety risks. The absence of large volumes of hot concentrated acid minimizes the potential for catastrophic spills or exposure incidents, aligning perfectly with modern ESG (Environmental, Social, and Governance) goals and stricter global safety regulations. Waste streams generated from this process are easier to manage and treat, as they lack the high acidity and sulfate load of traditional methods, facilitating easier discharge permits and community acceptance. This environmental compatibility ensures long-term operational viability and reduces the risk of shutdowns due to non-compliance, securing the supply of this critical pharmaceutical intermediate for years to come.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this solid superacid catalytic route for 2-chloro-6-fluorobenzaldehyde. These insights are derived directly from the patented methodology and are intended to clarify the operational benefits for potential manufacturing partners. Understanding these details is vital for assessing the feasibility of integrating this technology into existing production portfolios.
Q: What are the primary advantages of using ferric solid superacid over sulfuric acid in this synthesis?
A: Using ferric solid superacid eliminates the need for highly corrosive liquid sulfuric acid, significantly reducing equipment corrosion risks and waste treatment costs while improving yield from approximately 90% to 95%.
Q: How does the photo-chlorination step impact product purity?
A: Photo-chlorination using a metal-halide lamp allows for precise control over the chlorination degree without traditional radical initiators, resulting in fewer side reactions and a final product purity exceeding 99.7%.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process avoids hazardous liquid acids and complex initiator handling, making it safer and more scalable for industrial manufacturing of pharmaceutical intermediates like flucloxacillin precursors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Chloro-6-Fluorobenzaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the solid superacid hydrolysis process are translated into robust, industrial-grade manufacturing realities. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-chloro-6-fluorobenzaldehyde meets the exacting standards required for antibiotic and pesticide synthesis. Our commitment to technical excellence allows us to navigate complex regulatory landscapes while delivering products that support your downstream drug development and commercialization goals.
We invite you to collaborate with us to leverage these technological advancements for your specific supply chain needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your operational efficiency and product quality.
