Revolutionizing ADC Payload Production: A Safe, Scalable Route to High-Purity Maytansinol
Revolutionizing ADC Payload Production: A Safe, Scalable Route to High-Purity Maytansinol
The landscape of Antibody Drug Conjugate (ADC) manufacturing is undergoing a critical transformation, driven by the urgent need for safer and more scalable synthesis routes for potent cytotoxic payloads. Patent CN113825759A introduces a groundbreaking methodology for the preparation of maytansinol, a pivotal intermediate in the construction of maytansinoid-based ADCs, by leveraging organometallic chemistry to overcome longstanding safety and purity hurdles. This innovation shifts the paradigm from hazardous aluminum hydride reductions to a controlled nucleophilic substitution strategy using readily available Grignard reagents. For pharmaceutical manufacturers, this represents not just a chemical optimization but a strategic supply chain advantage, enabling the reliable production of high-purity pharmaceutical intermediates essential for next-generation oncology therapies. The technical depth of this disclosure offers a robust framework for scaling complex natural product derivatives while mitigating the severe process safety risks inherent in traditional reduction protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
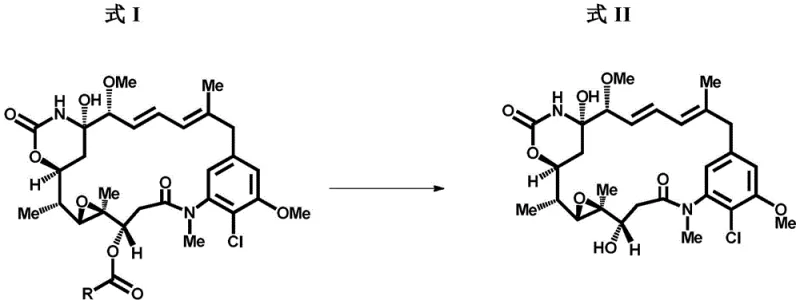
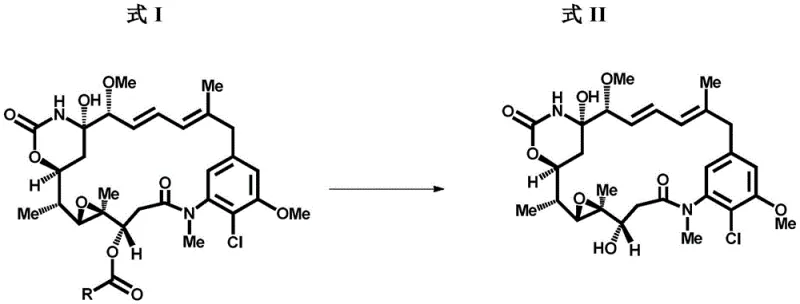
Historically, the conversion of ansamitocin esters, such as Ansamitocin P-3 (AP-3), into the free alcohol maytansinol has relied heavily on aluminum hydride reagents like lithium aluminum hydride (LAH) or its modified variants. While chemically feasible on a small scale, these conventional methods present catastrophic challenges when translated to industrial manufacturing environments. The primary drawback is the uncontrollable evolution of hydrogen gas during both the reaction initiation and the quenching phases, necessitating expensive, specialized explosion-proof equipment and rigorous safety monitoring systems that drastically inflate capital expenditure. Furthermore, the aggressive reducing nature of aluminum hydrides often leads to non-selective reduction, generating difficult-to-remove impurities such as over-deoxygenated by-products and des-chloro-maytansinol, which compromise the purity profile required for clinical-grade ADC payloads. The direct quenching of these reactive mixtures with protic solvents is inherently dangerous at scale, creating a bottleneck that limits throughput and increases the risk of thermal runaway incidents.
The Novel Approach
In stark contrast, the process detailed in CN113825759A utilizes organometallic reagents, specifically organomagnesium compounds like methyl magnesium bromide, to effect the cleavage of the ester moiety through a nucleophilic mechanism rather than a hydride transfer. This fundamental shift in reaction chemistry eliminates the generation of hydrogen gas entirely, thereby removing the most significant safety barrier to commercial scale-up. The reaction proceeds under mild, controlled low-temperature conditions, typically between -40°C and -15°C, which preserves the sensitive structural integrity of the maytansinoid macrocycle and prevents the formation of chloro-removed impurities. Moreover, the quenching step can be safely managed using common carbonyl compounds like acetone or alcohols like tert-butanol, resulting in a homogeneous or easily filterable mixture that simplifies downstream processing. This approach not only enhances operator safety but also streamlines the isolation of the target compound, making it an ideal candidate for reliable pharmaceutical intermediates supplier operations aiming for high efficiency.
Mechanistic Insights into Organometallic Ester Cleavage
The core of this technological advancement lies in the precise mechanistic pathway where the organometallic reagent acts as a nucleophile attacking the carbonyl carbon of the C-3 ester group on the ansamitocin scaffold. Unlike hydride reagents which deliver a hydrogen anion, the alkyl group from the Grignard reagent adds to the carbonyl, forming a tetrahedral intermediate that subsequently collapses to release the maytansinol alcohol and a corresponding ketone by-product. This mechanism is highly selective for the ester functionality and does not aggressively reduce other sensitive groups within the complex molecule, such as the epoxide or the amide linkages, provided the temperature is strictly controlled. The use of polar aprotic solvents like tetrahydrofuran (THF) further stabilizes the organometallic species, ensuring consistent reactivity and minimizing side reactions that could lead to degradation. By tuning the stoichiometry and addition rate, manufacturers can achieve near-quantitative conversion while maintaining a clean reaction profile that reduces the burden on purification columns.
Impurity control is another critical aspect where this mechanism outperforms traditional hydride reductions. In conventional LAH processes, the high reactivity often leads to the opening of the sensitive epoxide ring or the removal of the chlorine atom at the C-9 position, creating toxicological concerns for the final drug product. The organometallic route described here operates under kinetic control at low temperatures, effectively suppressing these competing pathways. The resulting crude product contains significantly fewer structurally related impurities, which simplifies the chromatographic purification steps and improves the overall yield of the isolated maytansinol. This level of chemical precision is essential for meeting the stringent purity specifications demanded by regulatory agencies for ADC payloads, ensuring that the final conjugate maintains its therapeutic index without unexpected toxicity from process-related impurities.
How to Synthesize Maytansinol Efficiently
The implementation of this novel synthesis route requires careful attention to reaction parameters, particularly temperature control and reagent addition rates, to maximize yield and safety. The process begins with the dissolution of the starting ansamitocin material in an anhydrous ether solvent under an inert atmosphere, followed by cooling to sub-zero temperatures to prepare for the exothermic addition of the organometallic reagent. Detailed standardized synthetic steps for executing this transformation, including specific molar ratios, quenching protocols, and purification gradients, are outlined in the technical guide below to assist process chemists in replicating these results.
- Dissolve Ansamitocin P-3 (AP-3) in anhydrous THF under an inert atmosphere and cool the mixture to approximately -30°C.
- Slowly add an excess of methyl magnesium bromide (MeMgBr) solution dropwise while maintaining the low temperature to initiate nucleophilic attack.
- Quench the reaction carefully with acetone to consume excess reagent, followed by aqueous workup and purification via normal phase chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this organometallic process translates into tangible operational efficiencies and risk mitigation strategies that directly impact the bottom line. By replacing hazardous hydride reagents with stable, commercially abundant Grignard solutions, companies can significantly reduce the costs associated with specialized storage, handling, and disposal of dangerous chemicals. The elimination of hydrogen gas evolution removes the need for costly engineering controls and explosion-proof reactors, allowing for the utilization of standard glass-lined or stainless steel equipment that is already prevalent in most multipurpose pharmaceutical manufacturing facilities. This compatibility with existing infrastructure accelerates technology transfer timelines and reduces the capital investment required to bring new ADC programs to commercial production.
- Cost Reduction in Manufacturing: The shift to organometallic reagents offers substantial cost savings by simplifying the reaction workup and purification stages. Traditional hydride reductions often require complex, multi-step quenching procedures involving hazardous acids or careful water addition to manage gas release, which increases labor time and waste disposal costs. In contrast, the new method allows for a straightforward quench with acetone or alcohols, generating benign by-products that are easily separated. Furthermore, the improved selectivity of the reaction reduces the loss of valuable starting material to side products, thereby enhancing the overall mass balance and lowering the cost of goods sold for this high-value intermediate.
- Enhanced Supply Chain Reliability: Sourcing organomagnesium reagents is far more robust compared to specialized aluminum hydride variants, as Grignard reagents are commodity chemicals produced by numerous global suppliers in large volumes. This diversity of supply sources mitigates the risk of raw material shortages that could disrupt production schedules for critical oncology drugs. Additionally, the stability of these reagents allows for longer shelf-life and easier logistics management, ensuring that manufacturing campaigns can proceed without interruption due to reagent degradation or delivery delays, thus securing the continuity of supply for downstream ADC assembly.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this process aligns perfectly with modern green chemistry principles by minimizing the generation of hazardous waste streams. The absence of aluminum salts and hydrogen gas simplifies wastewater treatment and reduces the environmental footprint of the manufacturing site. The inherent safety of the procedure facilitates easier regulatory approval for scale-up, as safety assessments are more favorable without the risks of pyrophoric materials or pressurized gas release. This enables a smoother path from pilot plant to full commercial scale, ensuring that production capacity can be expanded rapidly to meet market demand without encountering regulatory bottlenecks.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this patented synthesis route, providing clarity on its advantages over legacy methods. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, offering a factual basis for decision-making.
Q: Why is the organometallic method superior to traditional lithium aluminum hydride reduction?
A: Traditional methods using lithium aluminum hydride often produce hazardous hydrogen gas, require dangerous quenching protocols, and generate significant over-reduced by-products like des-chloro-maytansinol. The organometallic approach described in CN113825759A avoids hydrogen evolution, allows for safer quenching with ketones or alcohols, and provides a cleaner impurity profile suitable for GMP manufacturing.
Q: What specific organometallic reagents are effective for this transformation?
A: The patent identifies organomagnesium reagents, particularly methyl magnesium bromide (MeMgBr), as highly effective. Other viable options include ethyl, propyl, or butyl magnesium halides, as well as nucleophilic organolithium reagents like methyllithium or n-butyllithium, offering flexibility in reagent sourcing for large-scale production.
Q: How does this process impact the scalability of ADC payload manufacturing?
A: By eliminating the exothermic risks associated with hydride reductions and hydrogen gas release, this process significantly enhances operational safety at scale. The use of standard Grignard reagents and manageable quenching procedures facilitates easier technology transfer from laboratory to multi-kilogram or ton-scale commercial production without requiring specialized explosion-proof infrastructure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Maytansinol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and safe manufacturing processes for high-potency intermediates like maytansinol. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of materials for their ADC programs. Our state-of-the-art facilities are equipped to handle sensitive organometallic chemistry under strict inert conditions, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the demanding requirements of global pharmaceutical regulators.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your project volume. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to delivering excellence in the manufacturing of complex pharmaceutical intermediates.
