Advanced Metal-Free Synthesis of 5-Iodoindole Compounds for Commercial API Manufacturing
Introduction to Patent CN112479972A
The pharmaceutical and agrochemical industries rely heavily on functionalized indole scaffolds, yet accessing specific substitution patterns remains a persistent synthetic challenge. Patent CN112479972A introduces a groundbreaking synthesis method for 5-iodoindole compounds that addresses the critical need for site-selective functionalization on the benzene ring of the indole core. Unlike traditional methods that often struggle with regiocontrol or require harsh conditions, this invention leverages a Lewis acid-catalyzed radical reaction using N-iodosuccinimide (NIS) or elemental iodine as the iodine source. The process operates under remarkably mild conditions—specifically at normal temperature and pressure—eliminating the need for energy-intensive heating or cryogenic cooling. This technological advancement represents a significant leap forward for manufacturers seeking reliable pharmaceutical intermediate supplier partnerships, as it offers a robust pathway to high-value aryl iodides essential for downstream cross-coupling reactions like Suzuki-Miyaura or Sonogashira couplings.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of the indole ring has been heavily biased towards the pyrrole moiety, specifically the C2 and C3 positions, due to their inherent high nucleophilicity. Achieving substitution on the benzene ring, particularly at the C5 position, has traditionally required complex strategies involving the installation and subsequent removal of directing groups at the C3 or Nitrogen positions. These conventional routes are often plagued by multi-step sequences, low atom economy, and the necessity for expensive transition metal catalysts which introduce difficult-to-remove heavy metal impurities. Furthermore, many existing protocols demand stringent anhydrous conditions or extreme temperatures, posing significant safety risks and operational costs during commercial scale-up of complex pharmaceutical intermediates. The lack of a general, effective strategy for direct C5 functionalization has long been a bottleneck in the efficient production of diverse indole-based active pharmaceutical ingredients (APIs).
The Novel Approach
The methodology disclosed in the patent revolutionizes this landscape by enabling direct iodination at the C5 position through a streamlined, one-pot procedure. By employing a Lewis acid catalyst in a halogenated organic solvent, the reaction system effectively activates the iodine source to target the C5 position with high specificity, bypassing the need for pre-functionalized directing groups. This approach not only simplifies the synthetic route but also dramatically improves the overall process efficiency and environmental profile. The versatility of the method is demonstrated by its tolerance to a wide array of functional groups, including aldehydes, ketones, esters, and cyano groups, making it universally applicable across diverse chemical spaces. 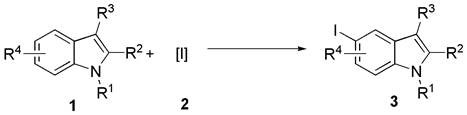
Mechanistic Insights into Lewis Acid-Catalyzed Electrophilic Iodination
The core mechanism driving this transformation involves the activation of the iodine source (NIS or I2) by the Lewis acid, generating a highly reactive electrophilic iodine species in situ. This activated species interacts with the electron-rich indole system, where the Lewis acid likely coordinates with the nitrogen atom or the carbonyl oxygen of the C3 substituent, thereby modulating the electron density distribution across the aromatic rings. This electronic modulation deactivates the typically more reactive C3 position and enhances the nucleophilicity of the C5 position on the benzene ring, facilitating selective electrophilic aromatic substitution. The reaction proceeds via a radical or cationic pathway depending on the specific Lewis acid used, such as BF3·Et2O or trifluoroacetic acid, ensuring that the iodination occurs exclusively at the desired site without significant formation of di-iodinated byproducts or C2/C3 isomers.
From an impurity control perspective, the absence of transition metals is a paramount advantage for R&D directors focused on purity specifications. Traditional palladium-catalyzed halogenations often leave trace metal residues that require costly scavenging steps to meet ICH Q3D guidelines. In contrast, this metal-free protocol utilizes organic or boron-based acids that are easily removed during standard aqueous workup and silica gel purification. The high regioselectivity minimizes the formation of structural isomers, simplifying the purification process and maximizing the yield of the target 5-iodoindole. This clean reaction profile ensures that the resulting high-purity pharmaceutical intermediates are ready for subsequent coupling reactions without extensive remediation, thereby accelerating the drug development timeline.
How to Synthesize 5-Iodoindole Efficiently
The practical implementation of this synthesis is designed for ease of operation, requiring standard laboratory equipment and readily available reagents. The general procedure involves dissolving the indole substrate and the iodine source in a solvent such as dichloromethane or chloroform, followed by the controlled addition of the Lewis acid catalyst at room temperature. The reaction typically reaches completion within 4 hours, after which standard extraction and purification techniques yield the desired product. For detailed operational parameters and specific stoichiometric ratios tailored to different substrates, please refer to the standardized synthesis steps provided below.
- Charge a reaction vessel with the indole substrate (e.g., indole-3-carbaldehyde) and the iodine source (NIS or I2) in a halogenated solvent like dichloromethane.
- Add a Lewis acid catalyst such as BF3·Et2O, TFA, or TfOH dropwise to the mixture under stirring at room temperature.
- Stir the reaction for approximately 4 hours, then perform extraction, solvent removal, and silica gel column purification to isolate the 5-iodoindole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis method translates into tangible strategic benefits regarding cost stability and supply continuity. The elimination of precious metal catalysts removes a major variable cost driver and mitigates the risk associated with the volatile pricing of metals like palladium or rhodium. Furthermore, the use of commodity chemicals such as N-iodosuccinimide, elemental iodine, and common Lewis acids ensures a resilient supply chain that is not dependent on specialized or scarce reagents. The mild reaction conditions also reduce energy consumption and infrastructure requirements, allowing for safer and more flexible manufacturing schedules.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by removing the need for expensive transition metal catalysts and the associated downstream purification steps required to meet regulatory limits. The high atom utilization rate and simplified workup procedure reduce solvent usage and waste disposal costs, leading to substantial overall savings in the production budget. Additionally, the ability to run the reaction at room temperature eliminates the capital and operational expenditures linked to heating or cooling systems, further enhancing the economic viability of large-scale production.
- Enhanced Supply Chain Reliability: By relying on widely available and stable raw materials, manufacturers can secure a consistent supply of critical intermediates without the risk of shortages often associated with specialized catalytic systems. The robustness of the reaction across a wide range of substrates means that a single production line can be adapted to synthesize various 5-iodoindole derivatives, increasing asset utilization and flexibility. This adaptability ensures reducing lead time for high-purity pharmaceutical intermediates and allows for rapid response to changing market demands or pipeline requirements.
- Scalability and Environmental Compliance: The metal-free nature of the reaction significantly simplifies waste treatment and aligns with increasingly stringent environmental regulations regarding heavy metal discharge. The use of halogenated solvents, while requiring proper management, is well-established in industrial settings with mature recovery and recycling protocols. The simplicity of the operation facilitates seamless scale-up from kilogram to tonne quantities, ensuring that the process remains efficient and safe regardless of the production volume, thus supporting sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of 5-iodoindole compounds, based on the detailed experimental data and advantages outlined in the patent documentation. These insights are intended to clarify the feasibility and benefits of adopting this novel methodology for your specific production needs.
Q: How does this method achieve C5 regioselectivity without directing groups?
A: The method utilizes specific Lewis acid activation which modulates the electron density of the indole ring, favoring electrophilic attack at the C5 position over the traditionally more reactive C2/C3 positions, even without additional directing groups.
Q: Are there heavy metal residues in the final 5-iodoindole product?
A: No, the process is completely metal-free, utilizing organic Lewis acids or boron-based catalysts instead of transition metals like palladium or copper, ensuring a cleaner impurity profile suitable for pharmaceutical applications.
Q: What is the substrate scope for this iodination reaction?
A: The reaction demonstrates broad compatibility with various substituents at the C3 position (aldehyde, ketone, ester, cyano) and N-position (H, alkyl, sulfonyl, acyl), tolerating halogens and other functional groups effectively.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Iodoindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development programs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 5-iodoindole delivered meets the highest international standards for pharmaceutical applications.
We invite you to contact our technical procurement team to discuss how this advanced synthesis technology can be integrated into your supply chain. Request a Customized Cost-Saving Analysis today to understand the specific economic benefits for your project, and ask for specific COA data and route feasibility assessments to validate the quality and reliability of our offerings.
