Scalable Synthesis of Novel Bicyclic Amino Substituted Pyridonecarboxylic Acid Derivatives for Advanced Antibacterial Applications
Scalable Synthesis of Novel Bicyclic Amino Substituted Pyridonecarboxylic Acid Derivatives for Advanced Antibacterial Applications
The pharmaceutical landscape for antibacterial agents is continuously evolving, driven by the urgent need for compounds that exhibit superior in vivo efficacy against resistant bacterial strains. Patent CN1175251A introduces a significant advancement in this domain by disclosing novel pyridonecarboxylic acid derivatives substituted with specific bicyclic amino groups at the 7-position. These compounds, characterized by a 3-azabicyclo[3.2.0]heptane scaffold, represent a strategic evolution from traditional quinolone antibiotics. The innovation lies not merely in the biological activity but in the robust chemical architecture that allows for diverse substitution patterns while maintaining high purity standards required for active pharmaceutical ingredients (APIs). For R&D directors and procurement specialists, understanding the synthetic accessibility and structural novelty of these intermediates is crucial for developing next-generation antimicrobial therapies that can overcome current resistance mechanisms.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional quinolone synthesis often relies on introducing simple cyclic amines such as piperazine or pyrrolidine at the 7-position of the quinolone nucleus. While effective, these structures frequently suffer from metabolic instability or suboptimal pharmacokinetic profiles when administered in vivo. Prior art, such as that described in JP-A-64-56673, explores various substituents but often fails to specifically disclose compounds where the amino group forms a rigid fused ring system like the ethylene-bridged bicyclic structures. Conventional methods may also involve harsh reaction conditions or complex protection-deprotection sequences that lower overall yield and increase impurity profiles. Furthermore, the flexibility of acyclic or monocyclic side chains can lead to multiple conformations in the binding pocket, potentially reducing binding affinity compared to more constrained analogs. These limitations necessitate a shift towards more rigid, structurally defined intermediates that offer both synthetic versatility and enhanced biological performance.
The Novel Approach
The approach detailed in the patent data utilizes a unique bicyclic amine intermediate, specifically derivatives of 3-azabicyclo[3.2.0]heptane, which are coupled to the pyridonecarboxylic acid core. This strategy leverages the inherent rigidity of the [3.2.0] fused ring system to lock the amino substituent into a specific conformation, thereby optimizing interactions with bacterial DNA gyrase and topoisomerase IV. ![General structure of the bicyclic amino group showing the 3-azabicyclo[3.2.0]heptane scaffold](/insights/img/bicyclic-amino-quinolone-intermediate-pharma-supplier-20260309020117-02.webp) The synthesis involves reacting a pyridonecarboxylic acid derivative bearing a leaving group at the 7-position with the novel bicyclic amine. This nucleophilic aromatic substitution is conducted under controlled conditions that minimize side reactions. By focusing on this specific bicyclic framework, the invention provides a pathway to compounds that demonstrate significantly improved antibacterial activity in animal models compared to reference compounds like pipemidic acid. This represents a tangible upgrade in therapeutic potential, offering a compelling value proposition for developers seeking high-efficacy antibacterial candidates.
The synthesis involves reacting a pyridonecarboxylic acid derivative bearing a leaving group at the 7-position with the novel bicyclic amine. This nucleophilic aromatic substitution is conducted under controlled conditions that minimize side reactions. By focusing on this specific bicyclic framework, the invention provides a pathway to compounds that demonstrate significantly improved antibacterial activity in animal models compared to reference compounds like pipemidic acid. This represents a tangible upgrade in therapeutic potential, offering a compelling value proposition for developers seeking high-efficacy antibacterial candidates.
Mechanistic Insights into Nucleophilic Aromatic Substitution and Cyclization
The core chemical transformation driving this technology is the nucleophilic displacement of a leaving group (typically a halogen such as fluorine or chlorine) at the 7-position of the quinolone ring system. 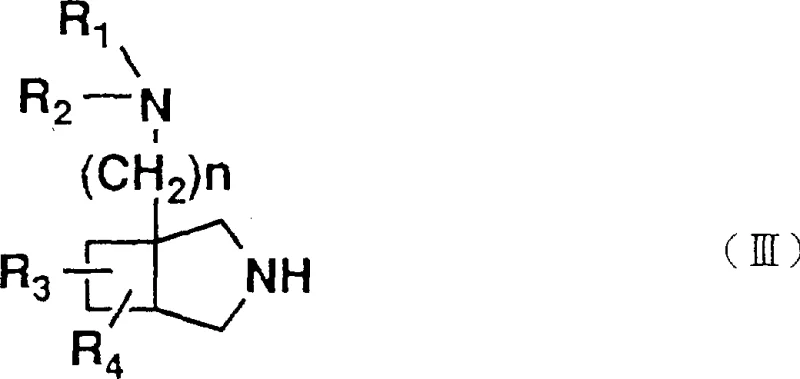 The mechanism proceeds via an addition-elimination pathway where the electron-rich nitrogen of the bicyclic amine attacks the electron-deficient carbon at the 7-position, activated by the adjacent carbonyl group at the 4-position. The presence of acid acceptors such as triethylamine or DBU is critical to neutralize the hydrogen halide byproduct, driving the equilibrium towards the desired product. The patent specifies reaction temperatures ranging from 10°C to 180°C, allowing for optimization based on the specific reactivity of the leaving group and the steric bulk of the amine. This mechanistic understanding is vital for process chemists aiming to scale the reaction, as controlling the exotherm and ensuring complete conversion are key to minimizing unreacted starting materials which can be difficult to separate from the final API.
The mechanism proceeds via an addition-elimination pathway where the electron-rich nitrogen of the bicyclic amine attacks the electron-deficient carbon at the 7-position, activated by the adjacent carbonyl group at the 4-position. The presence of acid acceptors such as triethylamine or DBU is critical to neutralize the hydrogen halide byproduct, driving the equilibrium towards the desired product. The patent specifies reaction temperatures ranging from 10°C to 180°C, allowing for optimization based on the specific reactivity of the leaving group and the steric bulk of the amine. This mechanistic understanding is vital for process chemists aiming to scale the reaction, as controlling the exotherm and ensuring complete conversion are key to minimizing unreacted starting materials which can be difficult to separate from the final API.
Furthermore, the preparation of the bicyclic amine intermediate itself involves sophisticated cyclization strategies. 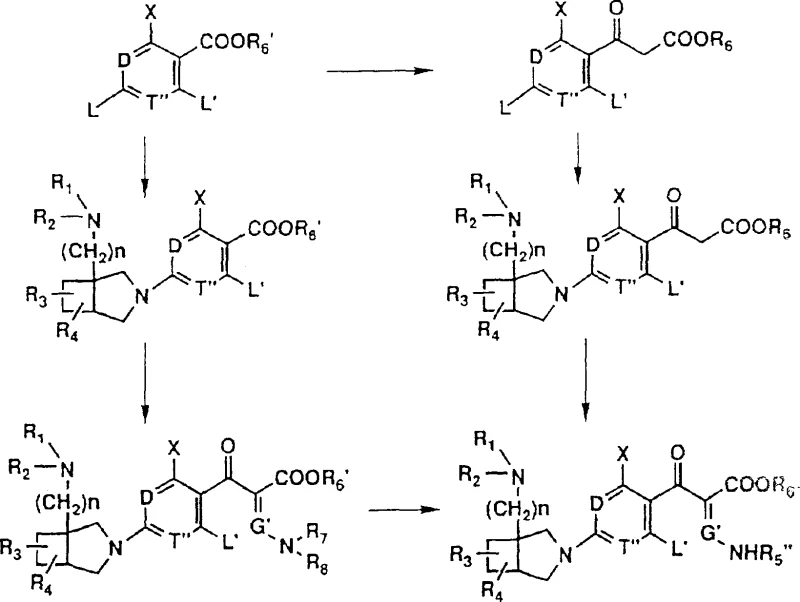 The synthesis of the 3-azabicyclo[3.2.0]heptane core often begins with cyclobutene derivatives, undergoing cycloaddition or ring-closing reactions to establish the fused ring system. Subsequent functionalization introduces the necessary amino group, often protected as a carbamate (e.g., Boc) to prevent self-reaction during the coupling step. The ability to manipulate substituents on the bicyclic ring (R3, R4) allows for fine-tuning of the physicochemical properties, such as solubility and lipophilicity, which are critical for oral bioavailability. Impurity control is managed through careful selection of protecting groups that can be orthogonally removed, ensuring that the final deprotection step yields the free amine without compromising the integrity of the quinolone core or the carboxylic acid functionality.
The synthesis of the 3-azabicyclo[3.2.0]heptane core often begins with cyclobutene derivatives, undergoing cycloaddition or ring-closing reactions to establish the fused ring system. Subsequent functionalization introduces the necessary amino group, often protected as a carbamate (e.g., Boc) to prevent self-reaction during the coupling step. The ability to manipulate substituents on the bicyclic ring (R3, R4) allows for fine-tuning of the physicochemical properties, such as solubility and lipophilicity, which are critical for oral bioavailability. Impurity control is managed through careful selection of protecting groups that can be orthogonally removed, ensuring that the final deprotection step yields the free amine without compromising the integrity of the quinolone core or the carboxylic acid functionality.
How to Synthesize 7-(1-Amino-3-azabicyclo[3.2.0]heptan-3-yl) Quinolones Efficiently
The efficient synthesis of these high-value intermediates requires a disciplined approach to reaction engineering and purification. The process generally begins with the preparation of the protected bicyclic amine, followed by its coupling with a suitably activated quinolone ester or acid. The patent outlines specific solvent systems, such as pyridine, acetonitrile, or DMSO, which facilitate the dissolution of both reactants and stabilize the transition state. Following the coupling reaction, the crude product typically undergoes hydrolysis to convert ester groups to the free carboxylic acid, a critical step for biological activity. Purification is often achieved through recrystallization or column chromatography, yielding products with high melting points indicative of high crystallinity and purity. For detailed operational parameters and specific stoichiometric ratios, refer to the standardized synthesis guide below.
- Preparation of the bicyclic amine intermediate via cycloaddition and subsequent functional group manipulation to introduce the amino moiety.
- Nucleophilic displacement of the leaving group at the 7-position of the pyridonecarboxylic acid derivative using the bicyclic amine.
- Hydrolysis or deprotection steps to yield the final free carboxylic acid form of the antibacterial agent.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain and procurement perspective, the adoption of this synthetic route offers distinct advantages over legacy methods for producing quinolone antibiotics. The reliance on well-defined chemical transformations reduces the risk of batch-to-batch variability, a common pain point in the sourcing of complex heterocyclic intermediates. The use of stable, commercially available starting materials for the bicyclic amine synthesis ensures a reliable supply chain, mitigating the risks associated with exotic or single-source reagents. Moreover, the robustness of the nucleophilic substitution step allows for processing in standard stainless steel reactors without the need for specialized high-pressure or cryogenic equipment, significantly lowering capital expenditure requirements for manufacturing partners.
- Cost Reduction in Manufacturing: The synthetic pathway eliminates the need for expensive transition metal catalysts often required in cross-coupling reactions, replacing them with cost-effective organic bases and thermal activation. This simplification of the catalyst system removes the necessity for rigorous heavy metal scavenging steps, which are both costly and time-consuming in a GMP environment. Additionally, the high crystallinity of the intermediate products facilitates purification through simple recrystallization rather than complex chromatographic separations, leading to substantial reductions in solvent consumption and processing time. These factors collectively contribute to a lower cost of goods sold (COGS), making the final API more competitive in the global market.
- Enhanced Supply Chain Reliability: The intermediates described, such as the 3-azabicyclo[3.2.0]heptane derivatives, are synthesized from robust precursors like cyclobutene carboxylic acids, which are accessible through established industrial chemical channels. This reduces dependency on fragile supply lines for niche building blocks. The chemical stability of the protected amine intermediates allows for extended storage and transportation without significant degradation, providing flexibility in inventory management. By securing a supply of these key building blocks, pharmaceutical companies can ensure continuous production schedules and avoid delays caused by raw material shortages, thereby enhancing overall supply chain resilience.
- Scalability and Environmental Compliance: The reaction conditions described operate within moderate temperature ranges and utilize solvents that are amenable to recovery and recycling, aligning with modern green chemistry principles. The absence of toxic heavy metals simplifies waste stream treatment, reducing the environmental footprint of the manufacturing process. Scalability is further supported by the exothermic nature of the amination reaction, which can be safely managed in large-scale reactors with appropriate cooling capacity. This ease of scale-up from laboratory to commercial production ensures that demand surges can be met without compromising quality or safety, a critical factor for maintaining regulatory compliance and market availability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these bicyclic amino-substituted quinolones. The answers are derived directly from the mechanistic data and experimental examples provided in the patent literature, ensuring accuracy and relevance for technical decision-makers. Understanding these details helps in assessing the feasibility of integrating this technology into existing development pipelines.
Q: What is the primary structural advantage of the 3-azabicyclo[3.2.0]heptane substituent?
A: The rigid bicyclic structure enhances metabolic stability and improves in vivo antibacterial activity compared to flexible acyclic or monocyclic amine substituents found in earlier generation quinolones.
Q: How does the patented amination method improve process efficiency?
A: The method utilizes direct nucleophilic substitution under mild conditions with common organic bases, avoiding complex catalytic systems and enabling easier purification through crystallization.
Q: Are these intermediates suitable for large-scale commercial production?
A: Yes, the synthesis relies on robust chemical transformations and readily available starting materials like cyclobutene derivatives, making it highly amenable to kilogram and ton-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyridonecarboxylic Acid Derivative Supplier
The development of advanced antibacterial agents requires a partner who understands both the molecular complexity and the commercial realities of pharmaceutical manufacturing. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from benchtop discovery to full-scale commercialization. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of characterizing complex bicyclic intermediates and ensuring they meet the highest international standards for API production. We are committed to delivering high-purity pharmaceutical intermediates that empower your R&D teams to advance their most promising candidates.
We invite you to engage with our technical procurement team to discuss how our capabilities align with your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized synthetic routes can reduce your overall development costs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your target molecules, ensuring a successful and efficient path to market.
