Revolutionizing Alkynyl Tetrahydroisoquinoline Synthesis via Green Photocatalysis for Commercial Scale
The pharmaceutical industry is constantly seeking more sustainable and efficient pathways for synthesizing complex heterocyclic scaffolds, particularly tetrahydroisoquinoline derivatives which serve as critical backbones for numerous bioactive compounds. Patent CN115894364A introduces a groundbreaking preparation method for alkynyl tetrahydroisoquinoline derivatives that fundamentally shifts the paradigm from traditional metal-catalyzed processes to a green photocatalytic approach. This innovation leverages visible light irradiation to drive cross-dehydrogenative coupling reactions in an aqueous medium, thereby eliminating the reliance on hazardous organic solvents and stoichiometric oxidants. By utilizing an insoluble polyporphyrin as a heterogeneous photosensitizer, the process achieves moderate to good yields ranging from 53% to 79% under mild room temperature conditions. This technological advancement not only addresses the growing regulatory pressure for greener manufacturing but also offers a robust platform for the scalable production of high-purity pharmaceutical intermediates required by modern drug discovery pipelines.
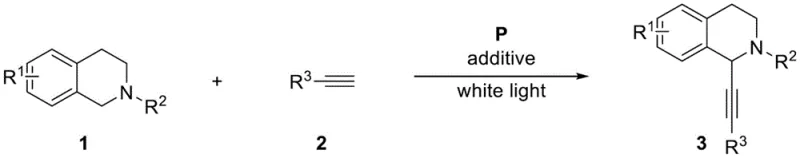
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the alpha-alkynylation of tetrahydroisoquinolines has predominantly relied on transition metal catalysis, often employing copper or other heavy metals in conjunction with stoichiometric oxidants such as tert-butyl hydroperoxide. These conventional methodologies present significant drawbacks for large-scale commercial manufacturing, primarily due to the stringent requirements for removing trace metal residues to meet pharmaceutical purity standards. Furthermore, the use of volatile organic solvents increases the environmental footprint and operational costs associated with solvent recovery and waste disposal. The necessity for chemical oxidants also introduces safety hazards related to exothermic reactions and potential instability during storage and handling. Additionally, homogeneous catalysts used in these traditional routes are difficult to separate from the reaction mixture, leading to product loss and increased downstream processing complexity which ultimately inflates the cost of goods sold for the final active pharmaceutical ingredient.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a heterogeneous insoluble polyporphyrin photosensitizer that operates efficiently in water under 20W white light irradiation. This method circumvents the need for toxic heavy metals and harsh oxidants by harnessing renewable light energy to facilitate the formation of C(sp3)-C(sp) bonds at ambient temperature. The heterogeneous nature of the catalyst allows for straightforward separation via simple centrifugation or filtration, enabling the catalyst to be recycled and reused multiple times without significant degradation in performance. The use of water as the sole reaction medium drastically reduces the volatility and flammability risks associated with organic solvents, enhancing overall plant safety. This shift towards photocatalysis in aqueous media represents a significant leap forward in process chemistry, aligning synthetic efficiency with the principles of green chemistry to deliver a more sustainable and economically viable manufacturing route.
Mechanistic Insights into Polyporphyrin-Catalyzed Cross-Dehydrogenative Coupling
The core of this innovative synthesis lies in the unique electronic properties of the insoluble polyporphyrin photosensitizer, which absorbs visible light to generate excited states capable of initiating single electron transfer processes. Upon irradiation with white light, the photosensitizer promotes the oxidation of the tetrahydroisoquinoline substrate to form an iminium ion intermediate, a critical step that activates the alpha-position for nucleophilic attack. The presence of 4-dimethylaminobenzoic acid as an additive plays a pivotal role in stabilizing this reactive iminium species through ion-pair interactions, thereby preventing side reactions and enhancing the selectivity of the coupling. The alkyne nucleophile then attacks the activated intermediate to forge the new carbon-carbon bond, resulting in the desired alkynyl tetrahydroisoquinoline derivative. This mechanism avoids the high energy barriers typically associated with thermal activation, allowing the reaction to proceed smoothly at room temperature while maintaining high functional group tolerance across a wide range of substrates.
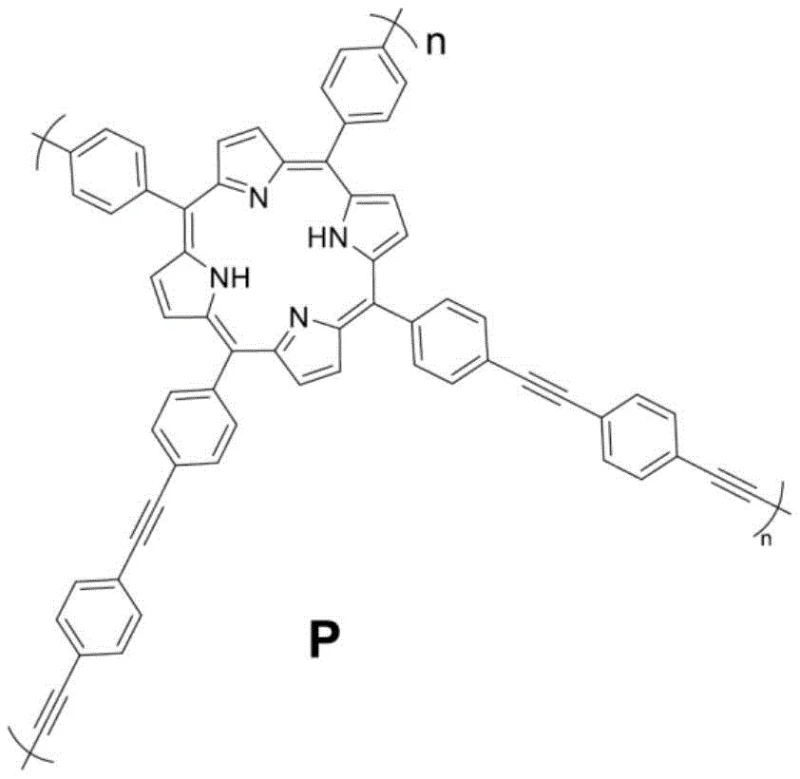
Impurity control in this photocatalytic system is inherently superior due to the mild reaction conditions and the specificity of the light-driven activation. Unlike thermal methods that may promote decomposition or polymerization of sensitive functional groups, the photochemical pathway selectively targets the specific C-H bond adjacent to the nitrogen atom. The insoluble nature of the polyporphyrin catalyst ensures that no metal leaching occurs into the product stream, which is a common source of contamination in homogeneous metal catalysis. This characteristic significantly simplifies the purification process, often requiring only standard extraction and chromatography to achieve high-purity specifications suitable for pharmaceutical applications. Furthermore, the stability of the polymeric catalyst under reaction conditions minimizes the formation of catalyst-derived byproducts, ensuring a cleaner reaction profile that reduces the burden on quality control laboratories during batch release testing.
How to Synthesize Alkynyl Tetrahydroisoquinoline Efficiently
The synthesis protocol outlined in the patent provides a clear and reproducible pathway for producing these valuable intermediates with minimal environmental impact. The process begins with the preparation of the insoluble polyporphyrin catalyst, followed by the setup of the photocatalytic reaction in water under white LED illumination. Operators simply need to combine the tetrahydroisoquinoline substrate, the alkyne coupling partner, the additive, and the catalyst in an aqueous suspension and stir at room temperature. Detailed standardized synthesis steps see below guide.
- Prepare the insoluble polyporphyrin photosensitizer via condensation of pyrrole and 4-bromobenzaldehyde followed by metallation and polymerization.
- Mix tetrahydroisoquinoline and alkyne substrates in water with 5mol% photosensitizer and 4-dimethylaminobenzoic acid additive.
- Irradiate the mixture with 20W white light at room temperature for 12 hours, then extract and recover the catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this photocatalytic technology offers substantial strategic benefits that extend beyond mere technical feasibility. The elimination of expensive transition metal catalysts and stoichiometric oxidants directly translates to a reduction in raw material costs and simplifies the sourcing strategy for key inputs. By replacing volatile organic solvents with water, the process significantly lowers the costs associated with solvent purchase, recovery, and hazardous waste disposal, which are major cost drivers in fine chemical manufacturing. The ability to recycle the heterogeneous photosensitizer multiple times further enhances the economic efficiency of the process by reducing the consumption of catalytic materials over time. These factors combined create a more resilient and cost-effective supply chain that is less vulnerable to fluctuations in the prices of precious metals and specialty chemicals.
- Cost Reduction in Manufacturing: The removal of heavy metal catalysts eliminates the need for costly scavenging resins and extensive purification steps required to meet strict residual metal limits in pharmaceutical products. This simplification of the downstream processing workflow reduces both the time and resources needed to bring the product to market. Additionally, the use of water as a solvent removes the capital expenditure associated with explosion-proof equipment and solvent recovery distillation columns. The overall reduction in process complexity leads to significant operational savings and a lower cost of goods sold, making the final intermediate more competitive in the global marketplace.
- Enhanced Supply Chain Reliability: Relying on abundant and non-toxic materials such as water and organic polymers reduces the risk of supply disruptions caused by geopolitical issues or regulatory restrictions on hazardous chemicals. The mild reaction conditions also decrease the likelihood of batch failures due to thermal runaways or equipment malfunctions, ensuring a more consistent and reliable production schedule. This stability is crucial for maintaining long-term contracts with pharmaceutical clients who require guaranteed supply continuity for their drug development programs. The robustness of the process allows for greater flexibility in production planning and inventory management.
- Scalability and Environmental Compliance: The heterogeneous nature of the catalyst and the use of water make this process highly amenable to scale-up from laboratory to commercial production volumes without significant re-engineering. The reduced generation of hazardous waste aligns with increasingly stringent environmental regulations, minimizing the risk of compliance violations and associated fines. This green manufacturing profile enhances the corporate sustainability image and meets the ESG criteria demanded by many multinational corporations. The ease of waste treatment and the lower environmental footprint facilitate smoother permitting processes for new manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic synthesis method. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation to provide clarity on process capabilities. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing supply chains.
Q: Can the polyporphyrin photosensitizer be reused in this synthesis?
A: Yes, the insoluble polyporphyrin catalyst can be recovered from the aqueous phase via centrifugation and reused multiple times without significant loss of activity.
Q: What are the environmental benefits of this water-based protocol?
A: Using water as the solvent eliminates the need for volatile organic compounds (VOCs) and reduces hazardous waste generation compared to traditional organic solvent methods.
Q: Does this method require expensive transition metal catalysts?
A: No, this method utilizes an organic polymeric photosensitizer, avoiding the use of costly and toxic transition metals like copper or palladium often found in conventional coupling reactions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkynyl Tetrahydroisoquinoline Supplier
NINGBO INNO PHARMCHEM stands at the forefront of adopting advanced synthetic technologies to deliver high-quality pharmaceutical intermediates to our global partners. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like this photocatalytic route are successfully translated into robust industrial processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of alkynyl tetrahydroisoquinoline derivatives meets the exacting standards required for drug substance manufacturing. Our commitment to green chemistry aligns with the industry's shift towards sustainable practices, providing our clients with a competitive edge in their own regulatory filings.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your specific project needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how this green photocatalytic method can optimize your supply chain and reduce overall manufacturing costs while ensuring the highest levels of product quality and safety.
