Scalable Production of High-Purity S-1-Ferrocenylethyldimethylamine via Novel Iridium Catalysis
Scalable Production of High-Purity S-1-Ferrocenylethyldimethylamine via Novel Iridium Catalysis
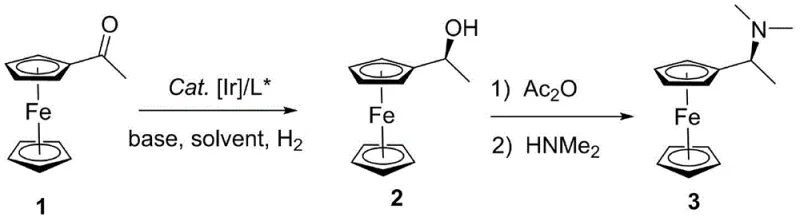
The pharmaceutical and fine chemical industries are constantly seeking more efficient routes to chiral building blocks, particularly those based on the robust ferrocene scaffold. A significant breakthrough in this domain is documented in patent CN108409802B, which discloses a highly efficient preparation technology for (S)-1-ferrocene ethyl dimethylamine. This compound serves as a critical skeleton for chiral catalysts and ligands, widely utilized in asymmetric synthesis such as Morita-Baylis-Hillman reactions. The patented process replaces inefficient traditional resolution methods with a sophisticated asymmetric catalytic hydrogenation strategy. By employing a novel metal Iridium complex paired with a specifically designed chiral ferrocene tridentate ligand, the technology achieves exceptional stereoselectivity and yield. This report analyzes the technical merits of this innovation, highlighting its potential to redefine supply chain standards for high-value chiral intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of optically pure (S)-1-ferrocene ethyl dimethylamine has relied heavily on chiral resolution techniques, typically utilizing chiral tartaric acid to separate racemic mixtures. While chemically straightforward, this approach suffers from inherent thermodynamic limitations, capping the maximum theoretical yield at 50% for a single resolution cycle. To achieve the high enantiomeric purity required for advanced catalytic applications, multiple rounds of recrystallization are often necessary, which drastically reduces overall throughput and increases solvent consumption. Furthermore, the generation of unwanted enantiomers as waste represents a significant loss of raw material value and creates additional burdens for waste treatment facilities. These inefficiencies translate directly into higher manufacturing costs and longer lead times, making conventional resolution less attractive for large-scale commercial procurement where cost-effectiveness and sustainability are paramount.
The Novel Approach
In stark contrast, the methodology outlined in CN108409802B leverages the power of asymmetric catalysis to bypass these yield barriers entirely. The core innovation lies in the direct asymmetric hydrogenation of acetylferrocene, a readily available and inexpensive starting material. By utilizing a highly active Iridium catalyst system, the process converts the prochiral ketone directly into the desired (S)-configured alcohol intermediate with high fidelity. This is subsequently transformed into the target amine through a streamlined acetylation and dimethylamine substitution sequence. The shift from a separation-based paradigm to a synthesis-based paradigm ensures that nearly every molecule of the starting material contributes to the final product, dramatically improving atom economy. This approach not only simplifies the operational workflow but also aligns perfectly with modern green chemistry principles by minimizing waste generation and energy consumption.
Mechanistic Insights into Iridium-Catalyzed Asymmetric Hydrogenation
The success of this synthetic route hinges on the precise design of the chiral ligand environment surrounding the Iridium center. The patent specifies the use of a chiral ferrocene tridentate ligand, denoted as L*, which coordinates with the metal to create a rigid, chiral pocket. As illustrated in the general structure below, the ligand features a ferrocene backbone substituted with a diphenylphosphino group and a chiral amine-sulfonamide side chain derived from cyclohexyl diamine. This unique architecture provides both steric bulk and electronic tuning capabilities essential for discriminating between the enantiotopic faces of the acetylferrocene substrate.
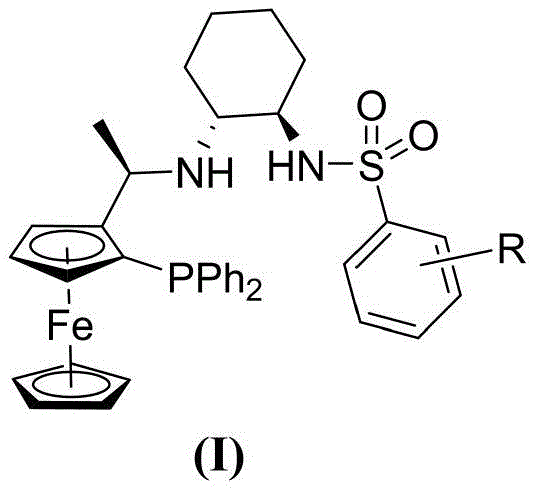
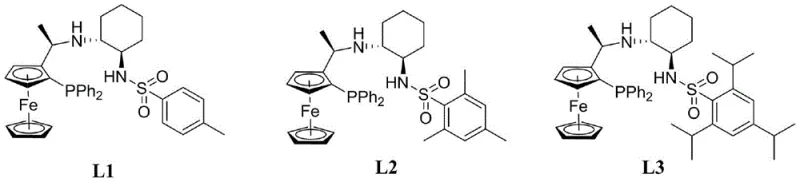
During the catalytic cycle, the Iridium complex activates molecular hydrogen and transfers hydride species to the carbonyl group of the acetylferrocene. The bulky substituents on the ligand, particularly the sulfonyl group and the cyclohexyl ring, exert strong steric control, forcing the substrate to approach the metal center in a specific orientation that favors the formation of the (S)-enantiomer. The patent highlights three specific variations of this ligand (L1, L2, and L3), where the electronic properties of the benzene ring on the sulfonamide are tuned via different substituents (R groups like methyl or isopropyl). This tunability allows chemists to optimize the reaction for specific batches or scales, ensuring consistent high enantiomeric excess (ee) values ranging from 91% to 98%. Such mechanistic robustness is critical for R&D teams aiming to minimize impurity profiles in downstream API synthesis.
How to Synthesize (S)-1-Ferrocenylethyldimethylamine Efficiently
The synthesis protocol described in the patent is operationally simple and amenable to standard reactor setups found in most fine chemical manufacturing plants. The process begins with the in-situ generation of the active catalyst by mixing the Iridium precursor with the chiral ligand in a solvent like methanol under an inert argon atmosphere. Once the catalyst is formed, acetylferrocene is introduced, and the system is pressurized with hydrogen. The reaction proceeds under mild thermal conditions, typically between 20°C and 60°C, which reduces the risk of thermal degradation and simplifies temperature control requirements. Following the hydrogenation, the crude chiral alcohol is subjected to a one-pot acetylation and amination sequence. Detailed standardized operating procedures for each step, including precise stoichiometric ratios and workup protocols, are essential for reproducibility.
- Prepare the active Iridium catalyst by reacting an Ir precursor like [Ir(COD)Cl]2 with a chiral ferrocene tridentate ligand (L*) in solvent under inert atmosphere.
- Perform asymmetric hydrogenation of acetylferrocene using the prepared catalyst under hydrogen pressure (2.0-5.0 MPa) and mild temperature (20-60°C) to obtain (S)-1-ferrocenylethanol.
- Convert the chiral alcohol to the target amine via acetylation with acetic anhydride followed by nucleophilic substitution with dimethylamine aqueous solution.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this catalytic technology offers compelling economic and logistical benefits. The primary driver for cost reduction is the dramatic improvement in yield efficiency. By eliminating the 50% yield ceiling associated with chiral resolution, the new process effectively doubles the output per unit of raw material input. Additionally, the patent notes that the catalyst loading can be reduced to extremely low levels, with molar ratios reaching as high as 1:100,000 in optimized scenarios. This means that the consumption of expensive Iridium metal and complex chiral ligands is minimized, leading to substantial cost savings in the bill of materials. The use of common, low-cost solvents like methanol and dichloromethane further enhances the economic viability of the process compared to specialized solvent systems often required for resolution.
- Cost Reduction in Manufacturing: The elimination of resolution agents and the reduction of recycling loops significantly lowers the operational expenditure. Since the reaction proceeds with high conversion rates, the burden on downstream purification units is reduced, saving energy and labor costs associated with distillation and crystallization. The ability to operate at lower pressures (2.0-5.0 MPa) and temperatures also reduces utility costs compared to high-energy thermal processes.
- Enhanced Supply Chain Reliability: Acetylferrocene is a commodity chemical with a stable global supply, unlike specialized chiral resolving agents which may have limited suppliers. By basing the synthesis on widely available feedstocks, the risk of supply disruption is mitigated. Furthermore, the shortened production cycle—removing the time-consuming recrystallization steps—allows for faster turnaround times from order to delivery, enabling manufacturers to respond more agilely to market demand fluctuations.
- Scalability and Environmental Compliance: The process generates significantly less waste, particularly the unwanted enantiomer salts typical of resolution methods. This reduction in solid waste simplifies disposal and lowers environmental compliance costs. The mild reaction conditions and robust catalyst system make the process highly scalable from kilogram laboratory batches to multi-ton commercial production without the need for exotic high-pressure equipment, facilitating a smoother technology transfer to manufacturing sites.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric hydrogenation technology. These answers are derived directly from the experimental data and claims presented in patent CN108409802B, providing a factual basis for evaluating the process feasibility.
Q: What are the advantages of this iridium-catalyzed method over traditional chiral resolution?
A: Unlike traditional resolution which is limited to a maximum theoretical yield of 50% and requires multiple recrystallizations, this asymmetric hydrogenation method offers near-quantitative conversion, significantly higher atom economy, and eliminates the need for resolving agents, thereby reducing waste and production time.
Q: What level of enantioselectivity can be achieved with this process?
A: The patent demonstrates that by optimizing the chiral ligand structure (specifically ligands L1, L2, and L3), the process can achieve excellent enantiomeric excess (ee) values ranging from 91% to 98%, ensuring high optical purity suitable for pharmaceutical applications.
Q: Is this synthesis route suitable for large-scale industrial manufacturing?
A: Yes, the process is designed for industrial scalability. It utilizes mild reaction conditions (20-60°C), common solvents like methanol and dichloromethane, and allows for extremely low catalyst loadings (molar ratios up to 1:100,000), making it economically viable for kilogram to ton-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-1-Ferrocenylethyldimethylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-purity chiral intermediates in the development of next-generation pharmaceuticals and advanced materials. Our technical team has thoroughly analyzed the pathway described in CN108409802B and possesses the expertise to implement this iridium-catalyzed route effectively. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to industrial manufacturing is seamless. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including precise measurement of enantiomeric excess (ee) and trace metal residuals, guaranteeing that every batch meets the exacting standards required by global regulatory bodies.
We invite R&D directors and procurement leaders to collaborate with us to leverage this advanced synthesis technology for their projects. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a stable, cost-effective supply of this critical chiral building block, empowering your innovation pipeline with reliable chemistry.
