Advanced One-Pot Synthesis of Bisazo Bisphenol Compounds for Industrial Dye Applications
Advanced One-Pot Synthesis of Bisazo Bisphenol Compounds for Industrial Dye Applications
The landscape of organic synthesis for functional dyes and photoconductive materials is undergoing a significant transformation driven by the need for higher efficiency and lower environmental impact. A pivotal development in this sector is detailed in patent CN113548979A, which discloses a novel preparation method for bisazo bisphenol compounds. This technology addresses long-standing inefficiencies in azo chemistry by streamlining the coupling process into a highly effective one-step reaction sequence following diazotization. For R&D directors and procurement specialists seeking reliable dye intermediate suppliers, this patent represents a critical pathway to optimizing production costs while maintaining stringent quality standards. The core innovation lies in the precise manipulation of reaction conditions—specifically temperature control at 0-5°C during diazotization and pH regulation during coupling—to maximize yield and minimize byproduct formation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of disazo compounds has been plagued by cumbersome multi-step procedures that inherently limit production throughput and economic viability. Traditional routes often rely on the Mills reaction or sequential oxidation and reduction steps, such as reducing nitrobenzene derivatives or dehydrogenating arylhydrazines. These methods frequently suffer from low atom economy and require harsh reaction conditions that can degrade sensitive functional groups. Furthermore, achieving the specific bis-substitution pattern required for high-performance photoconductive materials often necessitates protecting group strategies or difficult separation processes to isolate the desired disazo product from monoazo impurities. The cumulative effect of these inefficiencies is a substantial increase in manufacturing costs and extended lead times, creating bottlenecks for supply chain heads managing inventory for downstream applications in fiber coloring and electronic materials.
The Novel Approach
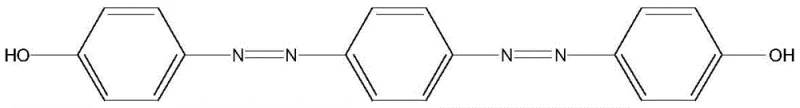
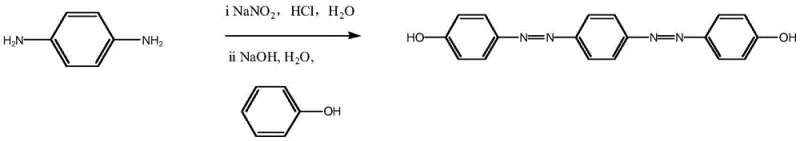
In stark contrast, the methodology outlined in CN113548979A introduces a streamlined protocol that bypasses these historical hurdles through a direct coupling strategy. By utilizing p-phenylenediamine as the starting diamine and reacting it directly with phenol derivatives under controlled alkaline conditions, the process achieves the formation of the bisazo structure in a single operational sequence after the initial diazotization. This approach eliminates the need for intermediate isolation of monoazo species, thereby drastically reducing solvent consumption and processing time. The ability to generate the target bisazo bisphenol compound directly from commodity chemicals like phenol and p-phenylenediamine ensures a robust supply chain foundation. This novelty not only enhances production efficiency but also aligns with modern green chemistry principles by minimizing waste generation and energy consumption associated with multi-step purification.
Mechanistic Insights into Diazotization and Electrophilic Aromatic Substitution
The success of this synthesis hinges on a deep understanding of the electronic activation provided by the phenolate anion. In the first stage, p-phenylenediamine is converted into a bis-diazonium salt using sodium nitrite in a strong acid medium at low temperatures (0-5°C). This低温 condition is critical to prevent the decomposition of the unstable diazonium species. Once formed, this electrophilic diazonium salt is introduced into a solution containing the phenolate anion, which is generated by dissolving phenol in a strong alkali such as sodium hydroxide. The deprotonation of the phenolic hydroxyl group significantly increases the electron density at the ortho and para positions of the benzene ring, making it a potent nucleophile. Since the ortho positions are sterically accessible but the para position is electronically favored for attack by the bulky diazonium ion, the reaction proceeds selectively to form the azo linkage at the para position relative to the hydroxyl group.
Furthermore, the mechanism ensures high purity through stoichiometric control and pH management. The patent specifies a molar ratio of phenol to diazonium salt of 2:1, which thermodynamically drives the reaction towards the double substitution required for the bisazo structure, effectively suppressing the accumulation of mono-coupled intermediates. During the addition of the diazonium salt, the pH is carefully maintained at a neutral level. This is a crucial mechanistic nuance; if the solution is too acidic, the concentration of the reactive phenolate anion decreases, slowing the coupling rate. Conversely, if too alkaline, the diazonium salt may convert into unreactive diazohydroxides. By balancing these factors, the process achieves a clean conversion profile, resulting in a crude product that requires minimal purification, typically just recrystallization, to meet high-purity specifications demanded by the electronics and specialty dye industries.
How to Synthesize Bisazo Bisphenol Efficiently
Implementing this synthesis route in a pilot or commercial plant requires strict adherence to the thermal and dosing parameters defined in the patent to ensure reproducibility and safety. The process begins with the careful preparation of the diazonium salt, where temperature control is paramount to prevent exothermic runaway reactions. Following this, the coupling step demands precise metering of reagents to maintain the optimal pH window for electrophilic attack. The simplicity of the workup, involving acidification to precipitate the product followed by filtration, makes this an attractive candidate for scale-up. For technical teams looking to adopt this methodology, the detailed standardized synthesis steps provided below outline the exact operational sequence required to achieve the high yields reported in the intellectual property documentation.
- Dissolve p-phenylenediamine in water, add hydrochloric acid to form amine salt, then add sodium nitrite at 0-5°C to generate the diazonium salt solution.
- Prepare a phenolate solution by dissolving phenol in strong alkali, then slowly add the diazonium salt solution while maintaining neutral pH.
- Acidify the reaction mixture with hydrochloric acid, filter the precipitate, wash thoroughly, and recrystallize to obtain the purified bisazo bisphenol.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers compelling advantages that directly address the pain points of procurement managers and supply chain directors. The shift from complex multi-step syntheses to a direct coupling route fundamentally alters the cost structure of manufacturing bisazo intermediates. By eliminating intermediate isolation steps and reducing the number of unit operations, the process significantly lowers labor costs and utility consumption. Moreover, the reliance on widely available commodity feedstocks like p-phenylenediamine and phenol mitigates the risk of raw material shortages, ensuring a stable and continuous supply for long-term production contracts. This stability is crucial for maintaining consistent delivery schedules to downstream customers in the textile and polymer sectors.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and complex purification columns results in substantial cost savings. Traditional methods often require expensive catalysts that need to be removed to trace levels, adding significant expense to the refining process. In this new route, the reaction is driven by simple acid-base chemistry, removing the need for costly metal scavengers. Additionally, the high yield implies that less raw material is wasted per kilogram of finished product, directly improving the gross margin profile for manufacturers adopting this technology.
- Enhanced Supply Chain Reliability: The use of robust, non-hazardous reagents simplifies logistics and storage requirements. Unlike processes requiring sensitive organometallic reagents that demand inert atmosphere handling, this synthesis can be performed in standard stainless steel reactors with aqueous workups. This reduces the complexity of the manufacturing infrastructure and minimizes the potential for batch failures due to equipment malfunction or operator error. Consequently, suppliers can offer more reliable lead times and greater flexibility in order fulfillment, strengthening the resilience of the global supply chain for azo-based materials.
- Scalability and Environmental Compliance: The aqueous nature of the reaction medium and the straightforward filtration workup make this process inherently scalable from kilogram to tonne quantities without significant re-engineering. From an environmental standpoint, the reduction in organic solvent usage and the absence of heavy metal waste streams simplify wastewater treatment protocols. This alignment with stricter environmental regulations reduces the compliance burden on manufacturing sites, preventing costly shutdowns and ensuring sustainable long-term operation in regulated markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this bisazo synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent literature, providing clarity on how this method outperforms legacy techniques. Understanding these nuances is essential for technical evaluators assessing the feasibility of integrating this route into existing production lines.
Q: What are the primary advantages of this new bisazo synthesis method over traditional Mills reactions?
A: Unlike traditional methods that require multi-step reductions or oxidations with lower yields, this patented process utilizes a direct one-step coupling between a pre-formed diazonium salt and a phenolate anion. This significantly simplifies the workflow, reduces reaction time, and improves overall production efficiency and yield.
Q: How does the process control impurities during the azo coupling stage?
A: The process strictly controls the pH during the addition of the diazonium salt to the phenolate solution, maintaining neutrality to ensure selective para-substitution. Furthermore, the use of specific molar ratios (phenol:diazonium salt at 2:1) minimizes the formation of mono-azo byproducts, ensuring high purity of the final bisazo compound.
Q: Is this synthesis route scalable for industrial production of dye intermediates?
A: Yes, the route is highly scalable as it relies on commodity chemicals like p-phenylenediamine and phenol, and uses standard aqueous acid-base chemistry. The workup involves simple filtration and recrystallization, avoiding complex chromatographic separations, which makes it ideal for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bisazo Bisphenol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthesis route described in CN113548979A for the production of high-performance azo intermediates. As a leading CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in industrial practice. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of bisazo bisphenol meets the exacting standards required for advanced dye and photoconductive applications. We are committed to bridging the gap between innovative academic research and robust commercial manufacturing.
We invite procurement leaders and R&D teams to collaborate with us to optimize their supply chains using this advanced technology. By leveraging our expertise, you can secure a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, ensuring that your next project is built on a foundation of chemical excellence and supply security.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
