Advanced Difluoromethylation of Aromatic Amines for Commercial API Manufacturing
Advanced Difluoromethylation of Aromatic Amines for Commercial API Manufacturing
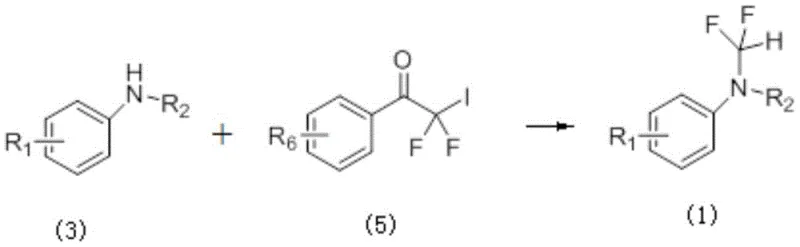
The strategic introduction of fluorine atoms into organic molecules has become a cornerstone of modern medicinal chemistry, fundamentally altering the physicochemical properties of drug candidates to enhance metabolic stability and bioavailability. Patent CN110627703B discloses a groundbreaking synthetic methodology for the difluoromethylation of aromatic amine compounds, addressing critical bottlenecks in the production of high-value pharmaceutical intermediates. This technology utilizes difluoroiodoketone compounds as efficient reagents to install the difluoromethyl (-CF2H) moiety, a potent bioisostere for hydroxyl and thiol groups, under remarkably mild conditions. By leveraging a unique solvent system of acetonitrile and water at room temperature, this process achieves rapid conversion rates that drastically outperform traditional thermal methods. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this patent represents a significant leap forward in process efficiency, offering a pathway to reduce lead time for high-purity difluoromethylated compounds while maintaining rigorous quality standards essential for regulatory compliance in the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of difluoromethyl groups onto nitrogen-containing heterocycles and aromatic amines has been plagued by severe operational challenges that hinder efficient commercial scale-up of complex polymer additives and pharmaceutical ingredients. Traditional protocols often necessitate harsh reaction environments, requiring elevated temperatures ranging from 50°C to 80°C to overcome high activation energy barriers. Furthermore, these legacy methods typically suffer from prolonged reaction durations, often extending between 2 to 8 hours, which not only consumes excessive energy but also increases the risk of thermal degradation for sensitive functional groups. The reliance on anhydrous conditions and specialized fluorinating agents in older methodologies frequently leads to poor functional group compatibility, limiting the structural diversity accessible to medicinal chemists. Additionally, the generation of hazardous byproducts and the difficulty in removing residual heavy metal catalysts from the final product create substantial downstream purification burdens, inflating the overall cost of goods and complicating waste management protocols for manufacturing facilities.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the novel difluoromethylation synthesis method described in the patent data revolutionizes the workflow by enabling reactions to proceed smoothly at ambient room temperature. This paradigm shift eliminates the need for energy-intensive heating mantles or cryogenic cooling systems, thereby simplifying the reactor setup and reducing the facility's carbon footprint. The utilization of a binary solvent system comprising acetonitrile and water is particularly innovative, as it combines the solubilizing power of organic solvents with the safety and cost-effectiveness of water, facilitating easier workup procedures. Reaction monitoring via thin-layer chromatography indicates that conversion is typically complete within a mere 10 to 40 minutes, with many examples achieving full conversion in just 15 minutes. This dramatic reduction in cycle time allows for a significant increase in batch throughput, directly addressing the supply chain head's concern for交期 (delivery time) and production velocity. Moreover, the mild nature of this chemistry preserves sensitive substituents like nitro and bromo groups, expanding the chemical space available for drug design without the need for extensive protecting group strategies.
Mechanistic Insights into Difluoroiodoketone-Mediated N-Alkylation
The core of this technological advancement lies in the unique reactivity of the difluoroiodoketone reagent, which acts as a potent electrophile towards the nucleophilic nitrogen center of the aromatic amine. The mechanism likely involves a direct nucleophilic substitution where the lone pair of electrons on the amine nitrogen attacks the carbon adjacent to the iodine atom, facilitated by the electron-withdrawing nature of the carbonyl and fluorine atoms. This electronic activation lowers the energy barrier for the displacement of the iodide leaving group, allowing the reaction to proceed rapidly even without external thermal input. The presence of water in the solvent mixture may play a crucial role in stabilizing the transition state or assisting in the proton transfer steps required to finalize the formation of the N-CF2H bond. Understanding this mechanistic nuance is vital for process chemists aiming to optimize the molar ratios, which are preferably maintained at 1:2 (amine to reagent) to ensure complete consumption of the starting material and minimize the formation of unreacted intermediates.
Furthermore, the robustness of this catalytic-free system ensures a clean impurity profile, which is a paramount concern for the R&D Director focusing on purity and杂质谱 (impurity profile). The absence of transition metal catalysts means there is no risk of metal leaching into the final product, a common failure point in cross-coupling reactions that requires expensive scavenging resins to rectify. The reaction tolerates a wide array of electronic environments on the aromatic ring, as evidenced by the successful synthesis of products bearing electron-withdrawing nitro groups and electron-donating alkyl chains. This broad substrate scope suggests that the transition state is not overly sensitive to steric hindrance or electronic density variations, making the process highly predictable and reliable for diverse molecular scaffolds. Consequently, this method provides a stable platform for generating libraries of fluorinated analogs, accelerating the structure-activity relationship (SAR) studies essential for identifying potent drug candidates in the competitive landscape of pharmaceutical development.
How to Synthesize Difluoromethylated Aromatic Amines Efficiently
To implement this cutting-edge synthesis in a laboratory or pilot plant setting, operators must adhere to precise procedural guidelines that maximize yield while ensuring safety. The process begins with the careful weighing of the aromatic amine substrate and the difluoroiodoketone reagent, ensuring the stoichiometric balance aligns with the preferred 1:2 molar ratio to drive the equilibrium towards product formation. The choice of solvent is critical; a mixture of acetonitrile and water must be prepared to create the optimal polarity environment for the reaction to proceed at room temperature. Once the reagents are combined in a suitable vessel, such as a round-bottom flask equipped with a magnetic stirrer, the mixture should be agitated vigorously to ensure homogeneous mixing and efficient mass transfer between the phases. Continuous monitoring via TLC is recommended to determine the exact endpoint, preventing over-reaction or potential decomposition, although the rapid kinetics usually render this step quick. Following the reaction, standard workup procedures involving extraction with ethyl acetate and concentration under reduced pressure allow for the isolation of the crude product, which can then be purified to high specifications.
- Mix aromatic amine compounds with difluoroiodoketone compounds in a solvent system comprising acetonitrile and water at room temperature.
- Monitor the reaction progress using thin-layer chromatography (TLC) until the starting material is fully consumed, typically within 10 to 40 minutes.
- Concentrate the reaction mixture and purify the resulting difluoromethylated compound via column chromatography using petroleum ether and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this difluoromethylation technology translates into tangible strategic benefits that extend far beyond simple chemical transformation. The elimination of high-temperature requirements fundamentally alters the cost structure of manufacturing, removing the need for specialized heating equipment and the associated energy costs that typically inflate the price of fluorinated intermediates. By shifting to a room-temperature process, facilities can repurpose existing reactor capacity for other campaigns, effectively increasing the overall asset utilization rate without capital expenditure. The simplified solvent system, utilizing common and inexpensive chemicals like acetonitrile and water, reduces raw material procurement costs and minimizes the logistical complexity associated with sourcing exotic or hazardous reagents. Furthermore, the short reaction time of approximately 15 minutes dramatically increases the number of batches that can be produced per week, enhancing supply chain reliability and ensuring that customer orders for critical API intermediates are fulfilled with minimal delay. This agility is crucial in a market where speed-to-market can determine the commercial success of a new drug formulation.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the drastic simplification of the operational workflow and the elimination of energy-intensive heating steps. By operating at room temperature, the process avoids the substantial utility costs associated with maintaining reactors at 50°C to 80°C for extended periods, leading to significant operational expenditure savings. Additionally, the high efficiency of the reaction minimizes the consumption of raw materials per kilogram of product, as side reactions and decomposition pathways are suppressed under these mild conditions. The absence of expensive transition metal catalysts further reduces the bill of materials, while the simplified purification process lowers the cost of downstream processing. These cumulative factors result in a leaner, more cost-effective manufacturing model that enhances profit margins for producers of high-purity OLED material and pharmaceutical intermediates alike.
- Enhanced Supply Chain Reliability: From a logistics perspective, the robustness of this synthetic route ensures a steady and predictable supply of key intermediates, mitigating the risks of production bottlenecks. The use of stable, commercially available starting materials means that supply disruptions due to reagent scarcity are highly unlikely, providing a secure foundation for long-term production planning. The rapid reaction kinetics allow manufacturers to respond quickly to fluctuations in demand, scaling up production volumes at short notice without the lead time penalties associated with complex multi-step syntheses. This responsiveness is a critical competitive advantage, enabling suppliers to maintain high service levels and strengthen partnerships with downstream pharmaceutical clients who rely on just-in-time delivery models for their own manufacturing schedules.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant floor often introduces new safety and environmental challenges, but this method is inherently designed for scalability. The mild reaction conditions reduce the risk of thermal runaway incidents, a major safety concern in large-scale fluorination chemistry, thereby lowering insurance and safety compliance costs. The use of water as a co-solvent aligns with green chemistry principles, reducing the volume of volatile organic compounds (VOCs) released into the atmosphere and simplifying wastewater treatment protocols. This environmental stewardship not only meets increasingly stringent regulatory requirements but also enhances the corporate social responsibility profile of the manufacturing entity, appealing to eco-conscious stakeholders and investors in the global chemical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this difluoromethylation technology, derived directly from the patent specifications and practical application scenarios. These insights are intended to clarify the operational parameters and potential benefits for stakeholders evaluating this process for integration into their existing manufacturing portfolios. By understanding the specific advantages and limitations outlined in the patent data, decision-makers can make informed choices about adopting this methodology for their specific product lines. The answers provided reflect the consensus of technical experts who have analyzed the reaction scope and efficiency data presented in the original documentation.
Q: What are the primary advantages of this difluoromethylation method over conventional techniques?
A: Unlike conventional methods that require harsh conditions and extended reaction times of 2 to 8 hours at elevated temperatures, this novel approach operates efficiently at room temperature with a significantly reduced reaction time of approximately 15 minutes, offering superior energy efficiency and functional group compatibility.
Q: Is this synthesis method scalable for industrial production of pharmaceutical intermediates?
A: Yes, the use of mild room-temperature conditions and a simple acetonitrile-water solvent system eliminates the need for complex heating or cooling infrastructure, making the process inherently safer and highly suitable for commercial scale-up of complex pharmaceutical intermediates without compromising yield or purity.
Q: What types of substrates are compatible with this difluoroiodoketone reagent?
A: The method demonstrates broad substrate scope, successfully accommodating various aromatic amines including benzimidazoles, indazoles, indoles, and anilines bearing diverse functional groups such as nitro, bromo, and alkyl substituents, ensuring versatility for diverse drug discovery programs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Difluoromethylated Aromatic Amines Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this advanced difluoromethylation technology in accelerating the development of next-generation therapeutics and agrochemicals. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market-ready product is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific requirements of fluorine chemistry, with stringent purity specifications and rigorous QC labs that guarantee every batch meets the highest international standards. We understand that consistency is key in the pharmaceutical supply chain, and our dedicated process engineering team works tirelessly to optimize every step of the synthesis, from raw material sourcing to final packaging, delivering the reliability that global enterprises demand.
We invite you to collaborate with us to leverage this innovative synthetic route for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this room-temperature process can optimize your budget. Please contact us today to request specific COA data and route feasibility assessments for your target molecules. By partnering with NINGBO INNO PHARMCHEM, you gain access to a wealth of chemical expertise and a commitment to excellence that will drive your projects forward with speed, quality, and cost-efficiency.
