Revolutionizing Pharmaceutical Intermediate Production via Mild Halogenation Oxidation Technology
Introduction to Advanced Heterocyclic Synthesis
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for safer, more efficient, and cost-effective synthetic routes. A pivotal advancement in this domain is detailed in Chinese Patent CN106928121B, which discloses a highly efficient preparation method for 3-substituted-benzo five-membered heterocyclic-2-carbonyl compounds. These structures serve as critical building blocks in the synthesis of complex bioactive molecules, including kinase inhibitors and other therapeutic agents. The patent introduces a novel halogenation oxidation strategy that fundamentally shifts the paradigm from traditional, hazardous multi-step processes to a streamlined, one-pot transformation. By leveraging specific N-E amide reagents, this technology enables the direct conversion of alcohol precursors into valuable carbonyl derivatives with exceptional selectivity and yield. For global procurement and R&D teams, understanding this technological leap is essential for optimizing supply chains and reducing the overall cost of goods sold (COGS) in active pharmaceutical ingredient (API) manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-halogenated indole carbonyl compounds has been fraught with significant operational challenges and safety hazards. Traditional methodologies often rely on cumbersome sequences involving distinct reduction, oxidation, and halogenation steps, each requiring isolation and purification. More critically, these legacy routes frequently necessitate the use of highly toxic and dangerous reagents such as butyllithium, phosphorus oxychloride, and hydrogen bromide. The reliance on cryogenic conditions for organolithium species not only escalates energy consumption but also introduces severe safety risks related to thermal runaway and pyrophoric materials. Furthermore, the use of aggressive halogenating agents like POCl3 generates substantial amounts of acidic waste, complicating environmental compliance and wastewater treatment protocols. These factors collectively contribute to extended lead times, inflated production costs, and a fragile supply chain vulnerable to regulatory scrutiny and raw material volatility.
The Novel Approach
In stark contrast, the methodology described in the patent offers a transformative solution by employing N-E amides as dual-function halogenating and oxidizing agents. This innovative approach allows for the direct conversion of 3-substituted-benzo five-membered heterocycle-2-alcohols into their corresponding carbonyl counterparts in a single operational step. The reaction proceeds under remarkably mild conditions, often at ambient temperature (25°C), thereby eliminating the need for energy-intensive heating or cooling systems. The versatility of this method is underscored by its compatibility with a wide array of solvents, including environmentally friendlier options like ethyl acetate. By consolidating multiple synthetic transformations into a unified process, this technology drastically simplifies the manufacturing workflow, reduces the cumulative yield loss associated with multi-step sequences, and minimizes the generation of hazardous byproducts. This represents a significant stride towards greener chemistry and more robust industrial scalability.

Mechanistic Insights into N-E Amide Mediated Oxidation
The core of this technological breakthrough lies in the unique reactivity profile of N-E amides, where E represents a halogen atom such as chlorine or bromine. These reagents, which include commercially available compounds like 1,3-dichloro-5,5-dimethylhydantoin (DCDMH) and N-bromosuccinimide (NBS), act as electrophilic halogen sources that facilitate the oxidation of the benzylic alcohol moiety. The mechanism likely involves the initial activation of the hydroxyl group by the N-halo species, followed by an elimination or hydride transfer process that establishes the carbonyl functionality while simultaneously introducing the halogen at the 3-position of the heterocyclic ring. This tandem transformation is highly chemoselective, preserving sensitive functional groups elsewhere on the molecule, such as esters, nitriles, or additional halogen substituents on the aromatic ring. The ability to control the stoichiometry and reaction environment ensures that over-oxidation or poly-halogenation is minimized, resulting in a cleaner crude product profile that simplifies downstream purification.
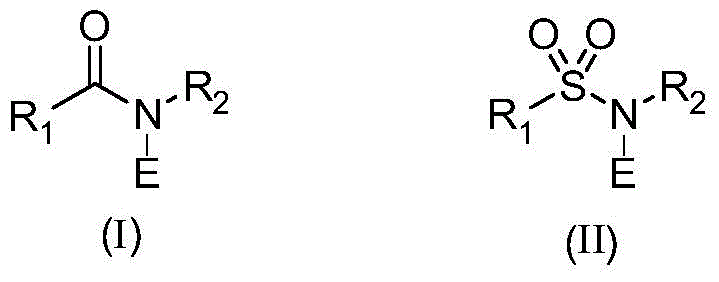
Furthermore, the structural diversity of the N-E amide reagents allows for fine-tuning of the reaction kinetics and thermodynamics. For instance, the use of cyclic imides like succinimide or hydantoin derivatives provides a stable leaving group that drives the reaction forward without generating corrosive mineral acids as primary byproducts. This mechanistic elegance translates directly into practical benefits for process chemistry teams. The reaction tolerance extends to various heterocyclic cores, including indoles, benzofurans, and benzothiophenes, demonstrating the robustness of the catalytic system. By avoiding the formation of reactive intermediates that require strict exclusion of moisture or oxygen, the process becomes inherently more forgiving and easier to manage on a large scale. This level of control over the reaction pathway is crucial for maintaining consistent product quality and meeting the stringent purity specifications required for pharmaceutical applications.
How to Synthesize 3-Chloroindole-2-Carbaldehyde Efficiently
Implementing this synthesis route requires careful attention to reagent selection and reaction monitoring to maximize efficiency. The process begins with the dissolution of the alcohol substrate in a compatible organic solvent, followed by the controlled addition of the N-halo amide. While the patent outlines a broad range of potential conditions, optimal results are typically achieved by maintaining a slight excess of the halogenating agent to ensure complete conversion of the starting material. The reaction progress can be monitored via standard analytical techniques such as TLC or HPLC, with completion often observed within a short timeframe of 15 to 60 minutes at room temperature. Upon completion, the workup procedure is straightforward, involving solvent removal and standard chromatographic purification to isolate the high-purity carbonyl product. For detailed operational parameters and specific stoichiometric ratios tailored to different substrates, please refer to the standardized synthesis guide below.
- Charge a reaction vessel with the 3-substituted-benzo five-membered heterocycle-2-alcohol substrate and a suitable solvent such as ethyl acetate or dichloromethane.
- Add the halogenating reagent, specifically an N-E amide like 1,3-dichloro-5,5-dimethylhydantoin (DCDMH) or N-bromosuccinimide (NBS), maintaining a molar ratio between 1: 0.1 and 1:10.
- Stir the mixture at mild temperatures ranging from -80°C to 200°C, preferably at ambient temperature (25°C), for 1 to 360 minutes until conversion is complete, followed by standard workup.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this halogenation oxidation technology offers compelling advantages that directly address the pain points of modern chemical procurement and supply chain management. The shift away from hazardous reagents like butyllithium not only enhances workplace safety but also significantly reduces the regulatory burden associated with the storage and transport of dangerous goods. This simplification of the hazard profile allows for more flexible manufacturing site selection and lowers insurance and compliance costs. Moreover, the use of commodity chemicals such as ethyl acetate and DCDMH ensures a stable and reliable supply of raw materials, mitigating the risk of production delays caused by specialty reagent shortages. The overall process intensification leads to a drastic reduction in manufacturing cycle times, enabling faster response to market demands and improved inventory turnover rates for critical intermediates.
- Cost Reduction in Manufacturing: The elimination of cryogenic conditions and expensive, hazardous reagents results in substantial operational expenditure savings. By consolidating multiple synthetic steps into a single pot, the process reduces labor hours, solvent consumption, and waste disposal fees. The high selectivity of the reaction minimizes the formation of difficult-to-remove impurities, thereby increasing the overall yield and reducing the cost per kilogram of the final API intermediate. This economic efficiency is further amplified by the ability to recycle solvents and recover byproducts, aligning with sustainable manufacturing goals.
- Enhanced Supply Chain Reliability: The reliance on widely available, shelf-stable reagents ensures a resilient supply chain that is less susceptible to geopolitical disruptions or vendor-specific bottlenecks. Unlike specialized organometallic reagents that require cold chain logistics, N-E amides can be stored and transported under ambient conditions, simplifying procurement logistics. This reliability is critical for maintaining continuous production schedules and meeting just-in-time delivery commitments to downstream pharmaceutical customers. The robustness of the method also allows for multi-sourcing strategies, further de-risking the supply of key intermediates.
- Scalability and Environmental Compliance: The mild reaction conditions and benign solvent profile make this process ideally suited for scale-up from pilot plant to commercial production volumes. The absence of highly toxic byproducts simplifies wastewater treatment and废气 (exhaust gas) management, ensuring compliance with increasingly stringent environmental regulations. This 'green' credential is becoming a key differentiator in supplier selection, as pharmaceutical companies prioritize partners who can demonstrate a commitment to sustainability. The scalability ensures that the technology can support the growing demand for complex heterocyclic intermediates without compromising on quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and embodiments disclosed in the patent literature, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this method into their existing manufacturing portfolios. For more specific technical data or custom synthesis requests, direct engagement with our technical team is recommended to explore tailored solutions.
Q: What are the primary advantages of using N-E amides over traditional lithiation methods?
A: The use of N-E amides eliminates the need for hazardous reagents like butyllithium and phosphorus oxychloride, allowing reactions to proceed under mild, ambient conditions with significantly improved safety profiles and reduced waste generation.
Q: Can this method be scaled for commercial production of API intermediates?
A: Yes, the process utilizes common industrial solvents like ethyl acetate and operates at ambient temperatures, making it highly scalable from kilogram to multi-ton quantities without requiring specialized cryogenic equipment.
Q: What is the substrate scope for this halogenation oxidation reaction?
A: The method demonstrates broad applicability across various 3-substituted-benzo five-membered heterocycles, including indoles, benzofurans, and benzothiophenes, tolerating diverse functional groups such as halogens, alkyls, and aryls.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Chloroindole-2-Carbaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient intermediate synthesis in the broader context of drug development and commercialization. Our expertise in process chemistry allows us to leverage advanced methodologies like the one described in CN106928121B to deliver high-quality intermediates with unmatched consistency. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients have access to reliable supply volumes regardless of their project stage. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch meets the exacting standards required for GMP manufacturing. By partnering with us, you gain access to a supply chain that is both technically sophisticated and commercially robust.
We invite you to collaborate with us to optimize your sourcing strategy for complex heterocyclic intermediates. Our team is ready to provide a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this novel synthetic route for your specific projects. We encourage you to contact our technical procurement team to request specific COA data, route feasibility assessments, and sample availability. Let us help you accelerate your timeline to market while reducing your overall production costs through innovation and operational excellence.
