Advanced Halogenation Oxidation Strategy for Scalable Production of High-Purity Heterocyclic Intermediates
Introduction to Efficient Heterocyclic Synthesis
The pharmaceutical and fine chemical industries constantly seek robust methodologies for constructing complex heterocyclic scaffolds, particularly those serving as critical building blocks for active pharmaceutical ingredients (APIs). Patent CN106928121B, published on May 12, 2020, introduces a groundbreaking approach for the efficient preparation of 3-substituted-benzo five-membered heterocyclic-2-carbonyl compounds. This technology addresses the longstanding challenges associated with synthesizing halogenated indole and benzofuran derivatives, which are ubiquitous motifs in medicinal chemistry. By leveraging a novel halogenation oxidation strategy, this method transforms readily available alcohol precursors into valuable carbonyl intermediates with exceptional selectivity. The significance of this innovation lies not only in its chemical elegance but also in its potential to streamline supply chains for global manufacturers seeking reliable sources of high-purity pharmaceutical intermediates.
The core of this invention involves the use of N-E amides, where E represents chlorine or bromine, as dual-function reagents that facilitate both oxidation and halogenation in a single operational step. This contrasts sharply with legacy processes that often require multiple discrete steps involving harsh conditions. For procurement and supply chain leaders, the implication is a reduction in process complexity and a mitigation of risks associated with handling dangerous reagents. As we delve deeper into the technical specifics, it becomes evident that this patent offers a viable pathway for the commercial scale-up of complex heterocyclic intermediates, ensuring consistent quality and availability for downstream drug synthesis applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-halogenated benzo five-membered heterocycle-2-carbonyl compounds has been fraught with significant technical and safety hurdles. Traditional routes frequently rely on cumbersome sequences involving separate reduction, oxidation, and halogenation steps, each requiring distinct reaction conditions and workup procedures. More critically, these conventional methods often necessitate the use of highly toxic and hazardous chemicals such as butyllithium, phosphorus oxychloride, and hydrogen bromide. The handling of such reagents demands specialized infrastructure, rigorous safety protocols, and expensive waste treatment systems, all of which drive up the overall cost of manufacturing. Furthermore, the harsh conditions employed in these legacy processes can lead to poor selectivity, resulting in complex impurity profiles that are difficult and costly to remove during purification. These factors collectively create bottlenecks in production capacity and pose substantial risks to supply chain continuity.
The Novel Approach
In stark contrast, the methodology disclosed in CN106928121B offers a streamlined, one-pot solution that dramatically simplifies the synthetic landscape. By utilizing N-E amides as the halogenating and oxidizing agents, the process achieves the transformation of 3-substituted-benzo five-membered heterocycle-2-alcohols directly into the corresponding carbonyl compounds under remarkably mild conditions.  . This reaction typically proceeds at ambient temperatures, around 25°C, eliminating the need for energy-intensive heating or cryogenic cooling. The versatility of the method is further enhanced by its compatibility with a wide range of solvents, including ethyl acetate, dichloromethane, and acetonitrile, allowing manufacturers to optimize for cost and environmental impact. This novel approach not only improves yield and selectivity but also aligns with modern green chemistry principles by reducing the reliance on hazardous reagents.
. This reaction typically proceeds at ambient temperatures, around 25°C, eliminating the need for energy-intensive heating or cryogenic cooling. The versatility of the method is further enhanced by its compatibility with a wide range of solvents, including ethyl acetate, dichloromethane, and acetonitrile, allowing manufacturers to optimize for cost and environmental impact. This novel approach not only improves yield and selectivity but also aligns with modern green chemistry principles by reducing the reliance on hazardous reagents.
Mechanistic Insights into N-E Amide Mediated Halogenation Oxidation
The mechanistic underpinning of this transformation relies on the unique reactivity of N-halo amides, which serve as electrophilic sources of halogen atoms while simultaneously facilitating oxidation. 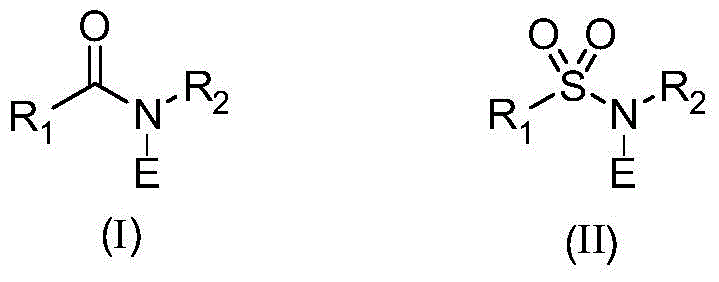 . In the proposed pathway, the N-E amide interacts with the electron-rich heterocyclic system, likely initiating an electrophilic attack at the 3-position of the indole or benzofuran ring. Concurrently, the alcohol moiety at the 2-position undergoes oxidation to the corresponding aldehyde or ketone. This tandem process is highly efficient because it avoids the isolation of unstable intermediate species that might degrade under harsher conditions. The choice of solvent plays a crucial role in stabilizing the transition states and ensuring high conversion rates. The ability to tune the reaction by selecting specific N-E amides, such as N-chlorosuccinimide (NCS) or N-bromosuccinimide (NBS), provides precise control over the final halogen substitution, enabling the synthesis of diverse libraries of chlorinated or brominated intermediates tailored for specific drug discovery programs.
. In the proposed pathway, the N-E amide interacts with the electron-rich heterocyclic system, likely initiating an electrophilic attack at the 3-position of the indole or benzofuran ring. Concurrently, the alcohol moiety at the 2-position undergoes oxidation to the corresponding aldehyde or ketone. This tandem process is highly efficient because it avoids the isolation of unstable intermediate species that might degrade under harsher conditions. The choice of solvent plays a crucial role in stabilizing the transition states and ensuring high conversion rates. The ability to tune the reaction by selecting specific N-E amides, such as N-chlorosuccinimide (NCS) or N-bromosuccinimide (NBS), provides precise control over the final halogen substitution, enabling the synthesis of diverse libraries of chlorinated or brominated intermediates tailored for specific drug discovery programs.
From an impurity control perspective, the mildness of the reaction conditions is a decisive factor in achieving high purity specifications. Harsh oxidants often lead to over-oxidation or ring degradation, generating byproducts that are structurally similar to the target molecule and thus difficult to separate. However, the selective nature of the N-E amide reagents minimizes these side reactions. The patent data indicates that the reaction exhibits high chemoselectivity, tolerating various functional groups on the aromatic ring and the nitrogen atom without interference. This selectivity translates directly into simplified downstream processing, as fewer impurities mean less rigorous chromatography or recrystallization steps are required. For R&D directors, this mechanistic clarity offers confidence in the robustness of the process when transferring from laboratory scale to pilot plant operations.
How to Synthesize 3-Chloro-1-hydro-indole-2-carbaldehyde Efficiently
To implement this technology effectively, manufacturers must adhere to optimized parameters regarding reagent stoichiometry and reaction time. The patent outlines a straightforward protocol where the substrate and the N-E amide are mixed in a suitable solvent, such as ethyl acetate, and stirred at room temperature. Detailed standard operating procedures for scaling this reaction, including specific quenching and purification techniques, are essential for maintaining batch-to-batch consistency.
- Prepare the reaction mixture by charging a reactor with the 3-substituted-benzo five-membered heterocycle-2-alcohol substrate and a suitable solvent such as ethyl acetate or dichloromethane.
- Add the halogenating reagent, specifically an N-E amide like N-chlorosuccinimide or N-bromosuccinimide, maintaining a molar ratio between 1: 0.1 and 1:10 relative to the substrate.
- Stir the mixture at a mild temperature ranging from -80°C to 200°C, preferably at 25°C, for 1 to 360 minutes until conversion is complete, followed by solvent removal and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis method presents compelling economic and logistical benefits. The shift away from hazardous reagents like butyllithium and phosphorus oxychloride fundamentally alters the risk profile of the manufacturing process. By eliminating these dangerous materials, companies can significantly reduce the costs associated with specialized storage, handling equipment, and hazardous waste disposal. This reduction in operational overhead contributes directly to a more competitive cost structure for the final intermediate. Moreover, the use of common, commercially available solvents and reagents ensures that the supply chain remains resilient against market fluctuations, as there is no dependency on exotic or tightly controlled chemicals that might face export restrictions or shortages.
- Cost Reduction in Manufacturing: The streamlined one-pot nature of the reaction eliminates the need for multiple isolation and purification steps that are typical in traditional multi-step syntheses. This consolidation of unit operations leads to substantial savings in labor, energy, and solvent consumption. Additionally, the high yields reported in the patent examples mean that less raw material is wasted, further driving down the cost per kilogram of the produced intermediate. The avoidance of expensive catalysts or specialized equipment also lowers the capital expenditure required for setting up production lines, making this technology accessible for both large-scale and niche manufacturing facilities.
- Enhanced Supply Chain Reliability: The raw materials required for this process, specifically the benzo five-membered heterocycle-2-alcohols and N-halo amides, are widely available from multiple global suppliers. This abundance mitigates the risk of supply disruptions that can occur with proprietary or single-source reagents. Furthermore, the mild reaction conditions allow for flexible manufacturing schedules, as the process does not require extreme temperatures or pressures that might strain utility systems. This flexibility enables producers to respond more agilely to changes in demand, ensuring timely delivery of critical intermediates to pharmaceutical clients who operate on tight development timelines.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges regarding heat transfer and mixing, but the exothermic nature of this reaction is manageable due to the mild conditions employed. The process generates minimal hazardous waste, aligning with increasingly stringent environmental regulations worldwide. By adopting this greener synthesis route, manufacturers can enhance their sustainability credentials, which is becoming a key differentiator in supplier selection criteria for major multinational corporations. The ease of scale-up from gram to kilogram quantities ensures that the technology can support projects from early-stage clinical trials through to commercial production without the need for process re-engineering.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis method is crucial for stakeholders evaluating its integration into their supply chains. The following questions address common inquiries regarding the practical application and benefits of the technology described in CN106928121B. These answers are derived directly from the experimental data and technical disclosures within the patent, providing a factual basis for decision-making.
Q: What are the primary advantages of using N-E amides for this oxidation compared to traditional reagents?
A: The use of N-E amides eliminates the need for highly toxic and hazardous chemicals such as butyllithium, phosphorus oxychloride, or hydrogen bromide, significantly improving operational safety and reducing environmental waste treatment costs.
Q: Can this method be applied to substrates with sensitive functional groups?
A: Yes, the reaction conditions are remarkably mild, typically proceeding at 25°C, which preserves sensitive functional groups like esters, cyano groups, and halogens on the aromatic ring without causing degradation.
Q: What is the typical yield range for these halogenation oxidation reactions?
A: According to the experimental data in the patent, the method consistently achieves high reaction selectivity and yields, often exceeding 70% and reaching up to 95% for various indole and benzofuran derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Halo-Indole Carbonyl Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of new therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volume requirements of even the most demanding projects. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our expertise in heterocyclic chemistry allows us to navigate the complexities of reactions like the halogenation oxidation described in CN106928121B, guaranteeing consistent quality and reliability for our partners.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can accelerate your development timeline and optimize your manufacturing costs. Let us be your trusted partner in bringing innovative medicines to market faster and more efficiently.
