Advanced Ruthenium-Catalyzed Synthesis of Chiral Lactams for Commercial API Production
Advanced Ruthenium-Catalyzed Synthesis of Chiral Lactams for Commercial API Production
The pharmaceutical industry continuously seeks more efficient pathways to construct chiral scaffolds, particularly lactams, which serve as critical backbone structures in numerous bioactive molecules ranging from receptor antagonists to opioid modulators. A significant technological breakthrough in this domain is detailed in patent CN112300047A, which discloses a highly efficient method for synthesizing chiral lactams via a tandem reductive amination and cyclization process. This innovation addresses long-standing challenges in asymmetric catalysis by utilizing a specialized ruthenium complex to facilitate the direct transformation of aromatic keto acids and ketoesters into valuable cyclic amine compounds. For R&D directors and procurement strategists, this represents a pivotal shift from multi-step, low-yield protocols to a streamlined, one-pot methodology that promises substantial improvements in both atom economy and stereochemical control.
The core value proposition of this technology lies in its ability to utilize simple, commercially available starting materials—specifically aromatic ketone derivatives and ammonium salts—to generate complex chiral architectures with exceptional precision. Unlike traditional approaches that often struggle with racemization or require harsh conditions, this ruthenium-catalyzed system operates under relatively mild hydrogenation conditions while delivering enantiomeric excess values reaching up to 99%. By integrating the reductive amination and ring-closing steps into a single catalytic cycle, the process not only simplifies the synthetic route but also minimizes the generation of hazardous waste, aligning perfectly with modern green chemistry principles and the stringent environmental compliance standards required by global supply chain heads.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of chiral lactams has been fraught with inefficiencies, primarily relying on stepwise synthetic strategies that are both time-consuming and resource-intensive. As illustrated in prior art methodologies, the conventional route typically involves the initial formation of an imine intermediate followed by a separate reduction step, and finally, a deprotection and cyclization sequence to close the ring. 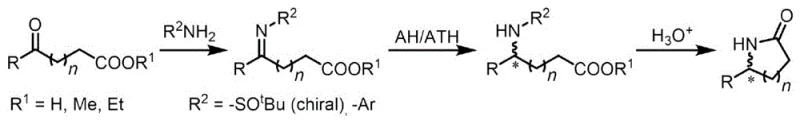 This fragmented approach necessitates the isolation and purification of unstable intermediates at each stage, leading to cumulative yield losses and increased exposure of sensitive chiral centers to conditions that might induce racemization. Furthermore, alternative cascade methods attempted in the past have often suffered from poor catalytic efficiency and inadequate stereocontrol, resulting in mixtures of diastereomers that are difficult and costly to separate on a commercial scale.
This fragmented approach necessitates the isolation and purification of unstable intermediates at each stage, leading to cumulative yield losses and increased exposure of sensitive chiral centers to conditions that might induce racemization. Furthermore, alternative cascade methods attempted in the past have often suffered from poor catalytic efficiency and inadequate stereocontrol, resulting in mixtures of diastereomers that are difficult and costly to separate on a commercial scale.
The reliance on complex amine sources or pre-functionalized substrates in these older methods further exacerbates the cost burden and supply chain vulnerability. Procurement managers often face difficulties sourcing specialized chiral amines in bulk quantities, and the additional reagents required for protection and deprotection groups contribute significantly to the overall material cost and E-factor of the process. Consequently, the manufacturing of high-purity chiral lactams via these traditional routes has remained a bottleneck for the rapid development of new drug candidates, driving the urgent need for a more direct and robust catalytic solution that can bypass these inherent structural limitations.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the technology described in patent CN112300047A introduces a groundbreaking tandem reductive amination and cyclization strategy that consolidates the entire synthesis into a single operational step. 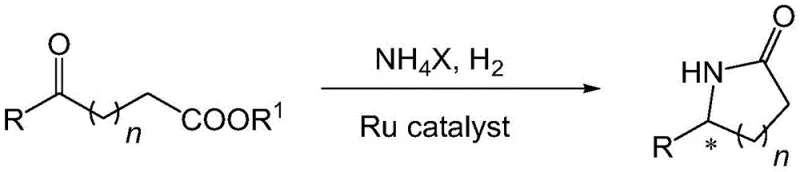 This novel approach leverages the unique reactivity of a chiral ruthenium catalyst to simultaneously activate the ketone substrate and the simple ammonium salt source, facilitating an immediate cascade that forms the C-N bond and closes the lactam ring in situ. By eliminating the need for intermediate isolation and protecting group manipulation, this method drastically reduces the number of unit operations required, thereby enhancing the overall throughput and reliability of the manufacturing process.
This novel approach leverages the unique reactivity of a chiral ruthenium catalyst to simultaneously activate the ketone substrate and the simple ammonium salt source, facilitating an immediate cascade that forms the C-N bond and closes the lactam ring in situ. By eliminating the need for intermediate isolation and protecting group manipulation, this method drastically reduces the number of unit operations required, thereby enhancing the overall throughput and reliability of the manufacturing process.
The implementation of this tandem reaction not only streamlines the workflow but also unlocks superior stereoselectivity that was previously unattainable with standard catalysts. The system effectively discriminates between prochiral faces of the ketone intermediate during the hydrogenation phase, ensuring that the final lactam product is obtained with high optical purity directly from the reactor. For supply chain leaders, this translates to a more predictable production timeline and reduced dependency on complex chromatographic separations. The ability to transform simple ketoesters directly into high-value chiral building blocks using inexpensive ammonia sources represents a paradigm shift in cost reduction in pharmaceutical intermediate manufacturing, offering a scalable solution that meets the rigorous demands of modern API production.
Mechanistic Insights into Ru-Catalyzed Asymmetric Reductive Amination
The success of this transformative synthesis hinges on the precise design of the catalytic system, specifically the utilization of a ruthenium complex coordinated with bulky, chiral diphosphine ligands. 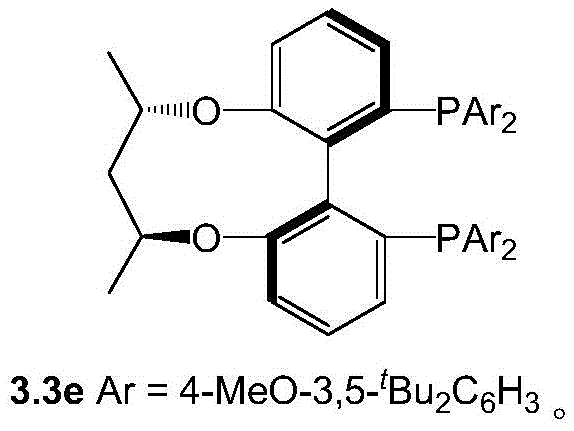 The patent identifies the Ru(3.3e)(OAc)2 complex as the optimal catalyst, featuring a sophisticated ligand framework that creates a highly defined chiral pocket around the metal center. This steric environment is crucial for directing the approach of the substrate and the hydride species, ensuring that the reduction occurs with strict facial selectivity. The mechanism likely proceeds through the initial formation of a hemiaminal or imine species from the ketone and ammonium acetate, which is then rapidly reduced by the ruthenium-hydride species generated under hydrogen pressure, followed by spontaneous intramolecular amidation to form the lactam ring.
The patent identifies the Ru(3.3e)(OAc)2 complex as the optimal catalyst, featuring a sophisticated ligand framework that creates a highly defined chiral pocket around the metal center. This steric environment is crucial for directing the approach of the substrate and the hydride species, ensuring that the reduction occurs with strict facial selectivity. The mechanism likely proceeds through the initial formation of a hemiaminal or imine species from the ketone and ammonium acetate, which is then rapidly reduced by the ruthenium-hydride species generated under hydrogen pressure, followed by spontaneous intramolecular amidation to form the lactam ring.
Beyond the catalyst architecture, the reaction medium plays a pivotal role in modulating the electronic properties of the transition state. Extensive optimization revealed that trifluoroethanol (TFE) is not merely a solvent but a critical promoter of the reaction efficiency and selectivity. The unique hydrogen-bonding capability and polarity of TFE appear to stabilize key ionic intermediates and enhance the proton transfer steps essential for the catalytic cycle. This mechanistic understanding allows for the accommodation of a wide range of substrates, as evidenced by the broad scope of compatible functional groups. 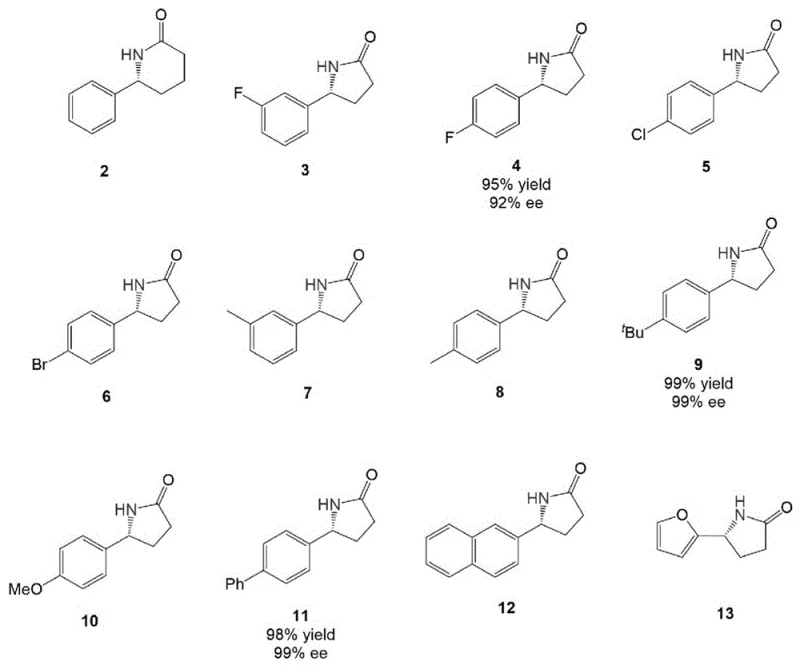 The system tolerates electron-withdrawing halogens, electron-donating alkyl groups, and even fused aromatic systems, consistently delivering high yields and enantiomeric excess. This robustness confirms that the catalytic cycle is resilient against potential poisoning by heteroatoms or steric hindrance, making it a versatile tool for the synthesis of diverse chiral heterocycles required in medicinal chemistry.
The system tolerates electron-withdrawing halogens, electron-donating alkyl groups, and even fused aromatic systems, consistently delivering high yields and enantiomeric excess. This robustness confirms that the catalytic cycle is resilient against potential poisoning by heteroatoms or steric hindrance, making it a versatile tool for the synthesis of diverse chiral heterocycles required in medicinal chemistry.
How to Synthesize Chiral Lactams Efficiently
Implementing this advanced synthesis protocol requires careful attention to reaction parameters to maximize the benefits of the tandem catalytic cycle. The process is designed to be operationally simple, utilizing standard hydrogenation equipment commonly found in pilot and production plants. The key to success lies in maintaining the precise stoichiometry of the ammonium source and ensuring the integrity of the catalyst system throughout the reaction duration. By adhering to the optimized conditions regarding temperature, pressure, and solvent choice, manufacturers can reliably produce target lactams with minimal impurity profiles. For detailed procedural instructions, please refer to the standardized synthesis guide below.
- Prepare the reaction mixture by combining the aromatic ketoester substrate, ammonium acetate as the nitrogen source, and the Ru(3.3e)(OAc)2 catalyst in trifluoroethanol solvent.
- Pressurize the reactor with hydrogen gas to 50 bar and heat the mixture to 90°C to initiate the tandem reductive amination and cyclization sequence.
- Maintain the reaction for 24 hours to ensure complete conversion, followed by standard workup procedures to isolate the high-purity chiral lactam product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this ruthenium-catalyzed tandem synthesis offers compelling economic and logistical advantages over traditional manufacturing routes. The most significant benefit stems from the drastic simplification of the process flow, which eliminates multiple isolation and purification steps associated with stepwise synthesis. This reduction in unit operations directly correlates to lower labor costs, decreased solvent consumption, and reduced energy usage, collectively driving down the cost of goods sold (COGS). Furthermore, the use of ammonium acetate as a nitrogen source replaces expensive and potentially hazardous chiral amines or protected amino acid derivatives, providing a substantial cost reduction in API manufacturing by leveraging commodity-grade raw materials.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation steps significantly reduces the consumption of auxiliary materials such as silica gel for chromatography and large volumes of extraction solvents. By consolidating the synthesis into a single pot, the process minimizes material loss due to mechanical transfer and purification, leading to higher overall mass balance. Additionally, the catalyst loading is maintained at a low level (1 mol%), which optimizes the usage of the precious metal ruthenium, ensuring that the catalyst cost does not become a prohibitive factor in large-scale production.
- Enhanced Supply Chain Reliability: The reliance on simple aromatic ketoesters and ammonium salts mitigates supply chain risks associated with sourcing complex, custom-synthesized chiral building blocks. These starting materials are widely available from multiple global suppliers, ensuring continuity of supply and reducing the lead time for high-purity intermediates. The robustness of the reaction conditions also means that the process is less susceptible to batch-to-batch variability caused by minor fluctuations in raw material quality, thereby enhancing the predictability of delivery schedules for downstream customers.
- Scalability and Environmental Compliance: The use of trifluoroethanol, while a specialized solvent, allows for high conversion rates that minimize the formation of by-products, simplifying waste treatment protocols. The high atom economy of the tandem reaction ensures that the majority of the starting material ends up in the final product, aligning with green chemistry metrics and reducing the environmental footprint of the manufacturing site. This scalability is further supported by the use of hydrogen gas, a clean reductant that produces no toxic waste, facilitating easier regulatory approval and commercial scale-up of complex heterocycles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on the practical aspects of the technology. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing production portfolios.
Q: What are the primary advantages of this tandem reductive amination method over traditional stepwise synthesis?
A: The primary advantage is the consolidation of multiple synthetic steps into a single operational unit. Traditional methods often require separate reduction, protection, and cyclization steps, which increases material loss and processing time. This patented tandem approach utilizes simple ammonium salts and achieves direct cyclization with high enantioselectivity, significantly reducing waste and operational complexity.
Q: How does the choice of solvent impact the stereoselectivity in this ruthenium-catalyzed process?
A: The patent data explicitly highlights trifluoroethanol (TFE) as the critical solvent for maximizing performance. Comparative studies within the technology show that common solvents like methanol, ethanol, or toluene result in messy reaction profiles or significantly lower conversion rates. TFE uniquely stabilizes the catalytic transition state, enabling enantiomeric excess values up to 99%.
Q: Is this catalytic system suitable for large-scale manufacturing of diverse lactam derivatives?
A: Yes, the system demonstrates broad substrate tolerance, accommodating various electronic and steric environments on the aromatic ring, including halogens, alkyl groups, and heterocycles. The use of a robust ruthenium catalyst at low loading (1 mol%) combined with inexpensive ammonium acetate suggests strong potential for cost-effective commercial scale-up without compromising purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Lactams Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the ruthenium-catalyzed tandem reductive amination technology for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a GMP-compliant manufacturing environment. Our state-of-the-art facilities are equipped with high-pressure hydrogenation reactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of chiral lactam delivered meets the exacting standards required for clinical and commercial applications.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cutting-edge synthesis route for their pipeline candidates. By partnering with our technical procurement team, you can obtain a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating exactly how this streamlined process can optimize your budget. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to delivering high-quality, cost-effective solutions for your complex synthetic needs.
