Advanced One-Pot Synthesis of Bis(triisopropylcyclopentadienyl) Strontium for High-Volume ALD Applications
The semiconductor industry continuously demands higher performance from dielectric materials, driving the need for precise atomic layer deposition (ALD) precursors like bis(triisopropylcyclopentadienyl) strontium. Recent technical advancements documented in patent CN113563390A highlight a breakthrough one-pot synthesis method that addresses long-standing challenges in producing high-purity strontium sources. This innovation is critical for manufacturing nanometer-thickness SrO thin films, which are essential for next-generation capacitors and memory devices. By leveraging strontium hydride and hexamethyldisilazane, the process eliminates the need for complex multi-step purifications that traditionally introduce solvent impurities. For R&D directors and procurement specialists, this represents a pivotal shift towards more reliable electronic chemical supplier networks capable of delivering consistent 5N purity materials. The implications for supply chain stability are profound, as simplified chemistry translates directly to reduced production risks and enhanced commercial scalability for complex organometallic compounds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of strontium dicyclopentadienyl derivatives has been plagued by inefficient methodologies that hinder cost reduction in semiconductor manufacturing. Traditional routes often rely on reacting strontium iodide with sodium salts of cyclopentadiene derivatives in solvents like tetrahydrofuran, which inevitably leads to solvent molecule coordination within the crystal lattice. These solvent adducts are notoriously difficult to remove, requiring multiple vacuum sublimation steps that drastically reduce overall yield and increase energy consumption. Furthermore, alternative methods involving metallic strontium in liquid ammonia present significant safety hazards and operational complexities that are unsuitable for large-scale industrial environments. The resulting products frequently suffer from low purity levels, often containing residual metal impurities that degrade the electrical properties of the deposited films. Consequently, manufacturers face substantial challenges in reducing lead time for high-purity electronic materials when relying on these outdated synthetic pathways.
The Novel Approach
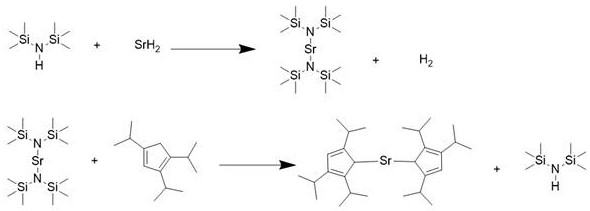
The novel approach detailed in the patent data introduces a streamlined one-pot strategy that fundamentally redefines the production efficiency of bis(triisopropylcyclopentadienyl) strontium. By utilizing strontium hydride dissolved directly in hexamethyldisilazane, the reaction avoids the introduction of extraneous metal ions and prevents the formation of stable solvent adducts that plague conventional methods. This methodology allows for a controlled reflux process where the solvent itself participates in the reaction mechanism, effectively displacing unstable intermediates and driving the equilibrium towards the desired product. The operational simplicity of this route means that reaction conditions can be maintained at moderate temperatures, significantly lowering the energy footprint associated with production. For supply chain heads, this translates to a more robust manufacturing process that is less susceptible to batch-to-batch variability and easier to scale from laboratory to commercial volumes. The elimination of hazardous reagents like liquid ammonia further enhances the safety profile, making it a viable option for continuous production facilities.
Mechanistic Insights into SrH2-Mediated Cyclopentadienyl Coordination
The core chemical innovation lies in the dual role of hexamethyldisilazane, which functions not merely as an inert medium but as an active participant in the coordination chemistry. When strontium hydride is introduced, it forms a reactive intermediate that is stabilized by the silazane ligands, preventing the premature decomposition of the sensitive cyclopentadienyl rings. As 1,2,4-triisopropylcyclopentadiene is added dropwise, a ligand exchange occurs where the bulky cyclopentadienyl groups displace the silazane moieties due to thermodynamic favorability under reflux conditions. This mechanism ensures that the steric hindrance provided by the isopropyl groups is maintained without compromising the binding strength to the strontium center, which is crucial for the volatility required in ALD applications. The careful control of addition rates and temperature prevents exothermic runaway reactions, ensuring the structural integrity of the ligand is preserved throughout the synthesis. Understanding this mechanistic pathway is vital for R&D teams aiming to replicate the high purity standards necessary for advanced electronic material applications.
Impurity control is achieved through the inherent selectivity of the reaction system, which excludes other metal elements from the reactant stream entirely. Since strontium hydride is the sole source of the metal center, there is no risk of contamination from counter-ions such as iodide or sodium that are common in salt-metathesis routes. The boiling point differential between hexamethyldisilazane and the final product allows for the efficient removal of the solvent during the reflux process, driving the reaction to completion without leaving residual coordination complexes. Post-reaction purification via reduced pressure sublimation further refines the material, removing any unreacted starting materials or minor byproducts to achieve a metal purity of 5N. This level of purity is critical for preventing defect formation in the resulting SrO thin films, which directly impacts the leakage current and dielectric constant of the final semiconductor device. The robustness of this impurity control mechanism ensures that the commercial scale-up of complex organometallic compounds can proceed without compromising on quality specifications.
How to Synthesize Bis(triisopropylcyclopentadienyl) Strontium Efficiently
Implementing this synthesis route requires strict adherence to inert atmosphere protocols and precise temperature control to maximize the 87% yield reported in the patent data. The process begins with the dissolution of strontium hydride powder in hexamethyldisilazane, followed by the controlled addition of the cyclopentadiene ligand under reflux conditions ranging from 125°C to 200°C. Detailed standard operating procedures regarding stirring rates, addition times, and sublimation parameters are essential for reproducing the high-purity results consistently across different production batches. Operators must ensure that the protective nitrogen atmosphere is maintained throughout to prevent oxidation of the sensitive hydride and organometallic intermediates. The following guide outlines the critical operational steps required to transition this laboratory-scale innovation into a reliable manufacturing process.
- Dissolve strontium hydride in hexamethyldisilazane under a protective inert atmosphere to form a stable mixed solution.
- Dropwise add 1,2,4-triisopropylcyclopentadiene to the mixture and maintain heating reflux conditions to drive the coordination reaction.
- Collect the crude product and purify via reduced pressure sublimation to achieve 5N metal purity suitable for atomic layer deposition.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthesis method offers substantial cost savings by eliminating the need for expensive metal salts and complex purification solvents. The use of strontium hydride and hexamethyldisilazane, which are readily available industrial chemicals, significantly reduces the raw material cost base compared to traditional precursors that require specialized organometallic reagents. Furthermore, the one-pot nature of the reaction reduces the number of unit operations required, leading to lower labor costs and decreased equipment utilization time per batch. For procurement managers, this efficiency translates into a more competitive pricing structure for high-purity ALD precursor without sacrificing quality or performance metrics. The simplified workflow also minimizes waste generation, aligning with increasingly stringent environmental compliance standards in the chemical manufacturing sector.
- Cost Reduction in Manufacturing: The elimination of solvent coordination means that extensive downstream purification steps, such as multiple recrystallizations or chromatographic separations, are no longer necessary. This reduction in processing complexity directly lowers the operational expenditure associated with energy consumption and solvent disposal. Additionally, the high yield of 87% ensures that raw material utilization is maximized, reducing the cost per kilogram of the final active pharmaceutical ingredient or electronic chemical. By avoiding the use of precious metal catalysts or exotic reagents, the overall bill of materials is significantly optimized, allowing for better margin management in volatile market conditions.
- Enhanced Supply Chain Reliability: The reliance on common chemical feedstocks like strontium hydride and hexamethyldisilazane mitigates the risk of supply disruptions often associated with specialized intermediates. This availability ensures that production schedules can be maintained consistently, reducing lead time for high-purity electronic materials needed by downstream semiconductor fabs. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, further stabilizing the supply chain. For supply chain heads, this reliability is crucial for maintaining just-in-time inventory levels and meeting the demanding delivery windows of global technology clients.
- Scalability and Environmental Compliance: The mild reaction temperatures and absence of hazardous solvents like liquid ammonia make this process inherently safer and easier to scale to multi-ton production volumes. The reduced generation of hazardous waste streams simplifies environmental permitting and lowers the costs associated with waste treatment and disposal. This environmental advantage is increasingly important for manufacturers seeking to meet corporate sustainability goals and regulatory requirements in key markets. The ability to scale without compromising safety or purity ensures that the technology remains viable as demand for strontium-based dielectric materials continues to grow in the electronics sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of bis(triisopropylcyclopentadienyl) strontium based on the patented methodology. These insights are derived directly from the experimental data and beneficial effects described in the intellectual property documentation. Understanding these details helps stakeholders evaluate the feasibility of integrating this precursor into their existing manufacturing workflows. The answers provided reflect the current state of the art in organometallic synthesis for electronic applications.
Q: What is the primary advantage of using hexamethyldisilazane in this synthesis?
A: Hexamethyldisilazane acts as both a solvent and a reactant, preventing solvent molecule coordination in the final product and ensuring higher metal purity up to 5N without requiring complex purification steps.
Q: How does this method improve yield compared to conventional strontium dicyclopentadienyl synthesis?
A: By avoiding unstable intermediates and utilizing a controlled one-pot reflux process, this method achieves yields as high as 87%, significantly outperforming traditional methods that often struggle to reach 50% yield due to side reactions.
Q: Is this precursor suitable for large-scale semiconductor manufacturing?
A: Yes, the process utilizes mild reaction conditions (130°C reflux) and readily available raw materials, making it highly scalable for industrial production while maintaining the stringent purity required for ALD processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bis(triisopropylcyclopentadienyl) Strontium Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes like this one-pot synthesis are successfully translated into industrial reality. Our technical team is equipped to handle the stringent purity specifications required for ALD precursors, utilizing rigorous QC labs to verify metal content and organic impurity profiles at every stage. We understand that the transition from patent to production requires not just chemical expertise but also robust project management and supply chain coordination. Our commitment to quality ensures that every batch of bis(triisopropylcyclopentadienyl) strontium meets the exacting standards necessary for high-performance semiconductor manufacturing.
We invite potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your volume requirements. By collaborating with us, you can leverage our manufacturing capabilities to achieve a Customized Cost-Saving Analysis that aligns with your budget and timeline goals. Let us help you secure a stable supply of high-quality electronic chemicals that drive your innovation forward.
