Advanced Synthesis of 2-Thiopheneethanol: A Cost-Effective Heck Coupling Strategy for Cardiovascular Intermediates
The pharmaceutical industry's relentless pursuit of efficient synthetic routes for cardiovascular drug intermediates has found a significant breakthrough in the methodology detailed in Chinese Patent CN102964334B. This pivotal intellectual property introduces a robust, two-step synthesis for 2-thiopheneethanol and its derivatives, fundamentally shifting the paradigm from hazardous organometallic chemistry to elegant palladium-catalyzed cross-coupling. For R&D directors and procurement strategists alike, this patent represents more than just a chemical curiosity; it outlines a pathway to cost reduction in pharmaceutical intermediate manufacturing by replacing expensive, dangerous reagents with stable, commercially available starting materials. The core innovation lies in utilizing 2-bromothiophene as a foundational building block, subjecting it to a Heck coupling reaction to form a key vinyl intermediate, which is subsequently converted into the target alcohol or amine precursors through selective hydrogenation. This approach not only simplifies the operational workflow but also addresses critical environmental and safety concerns that have long plagued the production of thiophene-based scaffolds used in major antiplatelet agents like clopidogrel.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 2-thiopheneethanol has been dominated by methodologies that impose severe operational burdens and safety risks, primarily relying on aggressive metallization strategies. As illustrated in the legacy butyllithium technique shown below, the process necessitates the use of cryogenic conditions to stabilize highly reactive organolithium species, which are not only prohibitively expensive but also pose significant fire and explosion hazards during large-scale handling.
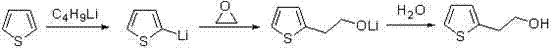
Furthermore, alternative historical approaches such as the sodium reagent technique or the Grignard reagent method suffer from equally daunting drawbacks, including the requirement for strictly anhydrous solvents and the generation of substantial amounts of hazardous waste. The Grignard route, while common, involves the use of volatile bromine and corrosive magnesium, leading to severe equipment degradation and complex purification challenges that drive up the total cost of ownership. Additionally, the ester reduction pathway, though avoiding some metallization issues, involves a multi-step sequence with low atom economy, requiring tedious operations like Willgerodt rearrangement and hydrolysis that result in poor overall yields and significant environmental pollution. These conventional methods collectively create a bottleneck for commercial scale-up of complex pharmaceutical intermediates, as they demand specialized infrastructure and rigorous safety protocols that erode profit margins.
The Novel Approach
In stark contrast to these archaic techniques, the novel process disclosed in the patent leverages the power of transition metal catalysis to achieve a streamlined, high-yielding synthesis under remarkably mild conditions. The strategy employs a palladium-catalyzed Heck coupling reaction between 2-bromothiophene and a vinyl tosylate derivative, effectively constructing the carbon-carbon bond necessary for the side chain without the need for cryogenic temperatures or stoichiometric metals. As depicted in the reaction scheme below, this intermediate is then subjected to a selective hydrogenation step where the final product profile—whether the alcohol or the tosylate precursor for amines—is dictated simply by adjusting the hydrogen pressure.
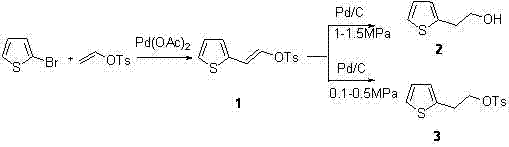
This bifurcation capability is a game-changer for supply chain flexibility, allowing a single production line to service multiple downstream drug synthesis needs, such as the production of prasugrel or ticlopidine intermediates. The reaction conditions are notably benign, operating at moderate temperatures between 120-140°C for the coupling step and ambient temperatures for the reduction, thereby eliminating the energy-intensive cooling requirements of the butyllithium route. Moreover, the use of catalytic palladium, which can be efficiently recovered from the reaction mixture, ensures that the process remains economically viable and environmentally sustainable, aligning perfectly with modern green chemistry principles and the demands of a reliable pharmaceutical intermediate supplier.
Mechanistic Insights into Palladium-Catalyzed Heck Coupling and Selective Hydrogenation
The mechanistic elegance of this synthesis lies in the precise orchestration of the palladium catalytic cycle during the Heck coupling phase, which facilitates the formation of the C-C bond with high regioselectivity. The cycle initiates with the oxidative addition of the palladium(0) species into the carbon-bromine bond of the 2-bromothiophene, generating a reactive organopalladium(II) intermediate that is stabilized by the electron-rich thiophene ring. This is followed by the coordination and migratory insertion of the vinyl tosylate olefin, a step that is critically influenced by the choice of ligand and solvent, with polar aprotic solvents like N-Methyl pyrrolidone (NMP) proving optimal for stabilizing the transition state. The subsequent beta-hydride elimination releases the coupled vinyl thiophene product and regenerates the active palladium catalyst, ready to enter another turnover, thus ensuring high catalytic efficiency and minimizing the loading of this precious metal.
Following the coupling, the selective hydrogenation mechanism offers a sophisticated level of control over the final functional group presentation, governed principally by the partial pressure of hydrogen gas applied to the system. When higher pressures (1.0-1.5 MPa) are applied over a Pd/C catalyst, the system favors the complete reduction of both the alkene double bond and the cleavage of the tosylate ester, yielding the primary alcohol 2-thiopheneethanol directly. Conversely, operating at lower pressures (0.1-0.5 MPa) allows for the selective reduction of only the alkene moiety while preserving the tosylate leaving group, generating a key intermediate for nucleophilic substitution reactions to form thiophene-2-ethylamine. This pressure-dependent selectivity minimizes the formation of unwanted byproducts and simplifies the impurity profile, ensuring that the resulting high-purity pharmaceutical intermediates meet the stringent specifications required for GMP manufacturing of cardiovascular APIs.
How to Synthesize 2-Thiopheneethanol Efficiently
The practical execution of this synthesis protocol is designed for seamless integration into existing multipurpose chemical reactors, requiring no exotic equipment beyond standard hydrogenation vessels. The process begins with the preparation of the vinyl thiophene intermediate via the Heck reaction, followed by a straightforward workup involving aqueous quenching and organic extraction to isolate the crude solid.
- Perform a Heck coupling reaction between 2-bromothiophene and a vinyl tosylate derivative using a palladium catalyst in a non-protonic solvent at elevated temperatures.
- Isolate the intermediate vinyl thiophene compound through extraction, drying, and recrystallization to ensure high purity before reduction.
- Conduct a selective catalytic hydrogenation using Pd/C under controlled pressure (1.0-1.5 MPa for alcohol, 0.1-0.5 MPa for tosylate) to yield the final target molecule.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Heck-based methodology translates into tangible strategic advantages that extend far beyond simple yield improvements. By transitioning away from stoichiometric organometallic reagents like butyllithium or magnesium, the process fundamentally alters the cost structure of production, removing the dependency on volatile raw material markets and reducing the logistical burden of handling hazardous Class 4.2 or 4.3 substances. The elimination of cryogenic cooling requirements further drives down utility costs, while the ability to recover and recycle the palladium catalyst creates a closed-loop system that mitigates the impact of fluctuating precious metal prices on the final unit cost.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven by the replacement of expensive, single-use reagents with catalytic systems and stable feedstocks. Unlike the Grignard process which consumes large quantities of magnesium and requires rigorous solvent drying, this method utilizes commodity chemicals like 2-bromothiophene and vinyl tosylates in a catalytic cycle. The recovery of palladium further enhances the cost-efficiency, ensuring that the overall production expense is significantly lowered compared to traditional metallization routes, providing a competitive edge in pricing for bulk active pharmaceutical ingredients.
- Enhanced Supply Chain Reliability: From a logistics perspective, the stability of the starting materials and the robustness of the reaction conditions greatly enhance supply security. 2-Bromothiophene is a widely available commodity with a mature supply chain, unlike specialized organolithium solutions which often have long lead times and strict storage requirements. The mild reaction conditions also mean that the process is less susceptible to disruptions caused by utility failures or equipment maintenance, ensuring a consistent and reliable flow of critical intermediates to downstream API manufacturers without the risk of batch failures due to moisture ingress or temperature excursions.
- Scalability and Environmental Compliance: The process is inherently designed for scale, utilizing solvents and conditions that are compatible with large-scale stainless steel reactors without the corrosion issues associated with halogenated Grignard reagents. The reduction in hazardous waste generation, particularly the avoidance of heavy metal sludge from magnesium disposal and the minimization of solvent volumes through efficient recycling, aligns with increasingly strict environmental regulations. This 'green' profile not only reduces waste disposal costs but also future-proofs the supply chain against tightening regulatory frameworks regarding chemical manufacturing emissions and effluent treatment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route, derived directly from the experimental data and comparative analysis provided in the patent documentation.
Q: How does the Heck coupling method improve safety compared to Grignard reagents?
A: The novel Heck coupling process eliminates the need for highly reactive magnesium metal and strictly anhydrous conditions required by Grignard reagents. By utilizing stable 2-bromothiophene and catalytic palladium, the reaction operates under milder thermal conditions, significantly reducing the risks associated with exothermic runaway reactions and equipment corrosion common in traditional metallization processes.
Q: Can this process selectively produce different derivatives like thiophene-2-ethylamine precursors?
A: Yes, the process offers exceptional selectivity through pressure control during the hydrogenation step. By adjusting the hydrogen pressure between 0.1-0.5 MPa versus 1.0-1.5 MPa, manufacturers can selectively retain the tosylate group for amine synthesis or fully reduce it to the alcohol, providing a versatile platform for producing multiple downstream cardiovascular drug intermediates from a single precursor.
Q: What are the scalability advantages of using palladium catalysis in this synthesis?
A: The use of catalytic amounts of palladium, which can be recovered and recycled, drastically lowers the material cost per kilogram compared to stoichiometric organometallic reagents. Furthermore, the reaction tolerates standard industrial solvents and does not require cryogenic cooling, making the transition from laboratory scale to multi-ton commercial production significantly more feasible and economically efficient.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Thiopheneethanol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated Heck coupling and selective hydrogenation techniques described in CN102964334B can be executed with precision and consistency. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 2-thiopheneethanol meets the exacting standards required for cardiovascular drug synthesis, providing our partners with absolute confidence in material quality.
We invite global pharmaceutical companies and contract manufacturing organizations to collaborate with us to leverage this cost-effective technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements, along with specific COA data and route feasibility assessments. Let us help you optimize your production of clopidogrel and prasugrel intermediates, securing a sustainable and economical supply of this critical building block for the global market.
