Revolutionizing Surfactant Production: A Green One-Step Synthesis for Commercial Scale
Revolutionizing Surfactant Production: A Green One-Step Synthesis for Commercial Scale
The global demand for sustainable and high-performance surfactants is driving a paradigm shift in chemical manufacturing, moving away from hazardous multi-step processes toward greener, atom-economical solutions. Patent CN110981758A introduces a groundbreaking synthetic method for fatty acyl amino acid surfactants that fundamentally alters the production landscape for this critical class of personal care and industrial ingredients. By leveraging a direct condensation strategy between fatty acids, liquid ammonia, and isethionates, this technology bypasses the traditional reliance on toxic acyl chlorides, offering a pathway to significantly cleaner manufacturing. For R&D directors and supply chain leaders, this innovation represents not just a chemical curiosity, but a viable route to cost reduction in surfactant manufacturing through the elimination of dangerous reagents and complex purification stages. The following analysis details how this one-step approach delivers superior purity profiles and operational safety.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of N-fatty acyl amino acid surfactants, such as N-acyl taurates, has been dominated by the Schotten-Baumann reaction or similar acyl chloride methodologies. These conventional routes are inherently flawed due to their reliance on aggressive acylating agents like thionyl chloride, phosphorus trichloride, or phosgene to activate the fatty acid. As illustrated in the traditional preparation of fatty acid chlorides, this activation step generates substantial quantities of hazardous byproducts, including phosphorous acids and hydrogen chloride gas, which pose severe safety risks and require specialized containment infrastructure. Furthermore, the subsequent condensation with amino acid salts in an alkaline medium inevitably produces stoichiometric amounts of inorganic salts, such as sodium chloride, as waste. This necessitates energy-intensive downstream processing, including acidification, high-temperature separation, or solvent extraction, to remove salt residues that can comprise 5-8% of the crude product. The resulting discharge of 2-10 tons of salt-containing wastewater per ton of product creates a massive environmental burden and complicates regulatory compliance for modern facilities.
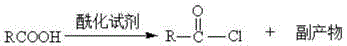
The Novel Approach
In stark contrast, the methodology disclosed in CN110981758A achieves the target surfactant through a direct, one-pot condensation that elegantly sidesteps the formation of acyl chloride intermediates entirely. By reacting fatty acids directly with isethionates in the presence of liquid ammonia under elevated temperature and pressure, the process achieves amidation without introducing chlorine atoms into the reaction matrix. This fundamental change in molecular design means that no inorganic salt byproducts are generated during the bond-forming step, effectively eliminating the need for the cumbersome desalination and washing steps that plague traditional methods. The reaction operates under autocatalytic conditions where liquid ammonia facilitates the nucleophilic attack, allowing for a streamlined workflow that transitions directly from raw materials to a finished product with minimal purification. This simplification not only enhances the safety profile by removing toxic reagents but also drastically improves the overall mass balance of the process, making it an ideal candidate for commercial scale-up of complex surfactants where waste minimization is paramount.
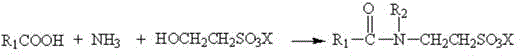
Mechanistic Insights into Autocatalytic Amidation
From a mechanistic perspective, this synthesis relies on the nucleophilic acyl substitution capability of ammonia and amine derivatives under thermal stress. The core transformation involves the attack of the nitrogen nucleophile, generated in situ from the reaction of ammonia with isethionate to form a taurate salt, upon the carbonyl carbon of the fatty acid. Unlike typical esterifications that require strong acid catalysts, this system utilizes the unique properties of liquid ammonia at high pressures (0.1-5.0 MPa) to drive the equilibrium toward amide formation. The reaction mechanism follows a classic addition-elimination pathway where the initial nucleophilic attack forms a tetrahedral intermediate, which subsequently collapses to expel a water molecule, yielding the stable amide bond characteristic of fatty acyl amino acid surfactants. The patent suggests an autocatalytic role for liquid ammonia, implying that the solvent environment itself stabilizes the transition states and facilitates proton transfer, thereby accelerating the reaction rate without the need for exogenous metal catalysts that could contaminate the final product.
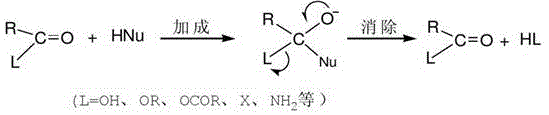
Controlling impurities in this system is intrinsically linked to the absence of halogenated reagents. In traditional acyl chloride routes, residual chloride ions are a persistent impurity that requires rigorous washing to meet cosmetic grade specifications. In this novel direct amidation, the primary potential impurities are unreacted fatty acids or isethionates, which are far easier to manage and often beneficial or neutral in the final surfactant formulation. The reaction conditions, specifically the temperature range of 150-300°C, ensure sufficient energy to overcome the activation barrier for direct amidation while the controlled pressure prevents the loss of volatile ammonia, maintaining a high concentration of the nucleophile. This precise control over reaction parameters ensures a high conversion rate, minimizing the presence of starting materials in the final crude mix and reducing the complexity of the final pH adjustment and granulation steps.
How to Synthesize N-Acyl Taurate Efficiently
The operational protocol for this synthesis is remarkably straightforward, designed to maximize throughput while minimizing operator exposure to hazardous conditions. The process begins by charging a high-pressure reactor with the fatty acid substrate and the isethionate salt, followed by sealing the vessel to create a closed system. The mixture is then heated to the target reaction temperature, typically between 150°C and 300°C, before liquid ammonia is slowly introduced. Maintaining the system pressure between 0.1 MPa and 5.0 MPa during ammonia addition is critical to ensure the reagent remains in the liquid phase and participates effectively in the autocatalytic cycle. Once the ammonia charge is complete, the reaction is held at temperature until the internal pressure naturally drops below 0.1 MPa, signaling the consumption of the gaseous reactant and the completion of the amidation. The detailed standardized synthesis steps see the guide below.
- Load fatty acid and isethionate salts into a high-pressure reactor and seal the system.
- Heat the mixture to 150-300°C and slowly introduce liquid ammonia while maintaining pressure between 0.1-5.0 MPa.
- Maintain temperature until pressure drops below 0.1 MPa, indicating reaction completion, then remove unreacted ammonia and adjust pH.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this one-step synthesis offers profound strategic advantages that extend beyond simple chemistry. By collapsing a multi-stage process involving hazardous intermediate isolation into a single reactor operation, manufacturers can significantly reduce capital expenditure on specialized corrosion-resistant equipment and waste treatment facilities. The elimination of thionyl chloride and phosgene from the supply chain removes a major logistical bottleneck, as these reagents often require special permits for transport and storage, thereby reducing lead time for high-purity surfactants and simplifying inventory management. Furthermore, the drastic reduction in wastewater generation translates directly into lower operational costs associated with effluent treatment and environmental compliance fees, enhancing the overall margin profile of the final product.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the complete removal of expensive and dangerous acylating agents. Traditional methods consume stoichiometric amounts of reagents like thionyl chloride, which are not only costly to purchase but also generate acidic waste that requires neutralization with base, adding further material costs. By switching to a direct fatty acid route, the process eliminates these reagent costs entirely. Additionally, the removal of the desalination step—often requiring large volumes of water and energy for crystallization or extraction—results in substantial utility savings. The simplified workflow also reduces labor hours and reactor occupancy time, allowing for higher throughput in existing facilities without the need for major retrofitting.
- Enhanced Supply Chain Reliability: Sourcing hazardous chemicals like phosgene or phosphorus pentachloride can be subject to strict regulatory quotas and supply disruptions, creating vulnerability in the production schedule. This new method relies on commodity chemicals—fatty acids, ammonia, and isethionates—which are produced on a massive global scale with stable pricing and abundant availability. This shift to widely available raw materials insulates the manufacturing process from the volatility of the specialty reagent market. Moreover, the robustness of the one-step reaction reduces the risk of batch failures associated with complex multi-step sequences, ensuring a more consistent and reliable supply of high-purity N-acyl taurate to downstream customers.
- Scalability and Environmental Compliance: As regulatory pressure mounts regarding industrial discharge, the ability to produce surfactants with minimal salt waste is a decisive competitive advantage. This process generates negligible inorganic salt byproducts, meaning the facility avoids the heavy burden of treating tons of saline wastewater per ton of product. This "green" credential is increasingly valuable for securing contracts with major multinational corporations that have strict sustainability mandates. The scalability is further enhanced by the simplicity of the unit operations; since the reaction is a homogeneous or semi-homogeneous liquid-phase process under pressure, it scales linearly from pilot plant to full commercial production without the mixing and heat transfer issues often seen in heterogeneous slurry reactions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing a clear understanding of the process capabilities and limitations for potential partners.
Q: How does this new synthesis method improve environmental compliance compared to traditional acyl chloride routes?
A: The new method eliminates the use of hazardous acylating agents like thionyl chloride and phosgene, thereby removing the generation of corrosive byproducts and significantly reducing the volume of salt-containing wastewater typically associated with the neutralization and desalination steps of conventional processes.
Q: What are the critical reaction parameters for ensuring high conversion in this one-step process?
A: Critical parameters include maintaining a reaction temperature between 150-300°C and controlling the system pressure within the 0.1-5.0 MPa range during the introduction of liquid ammonia, which acts as both a reactant and an autocatalyst to drive the condensation forward.
Q: Can this process be scaled for industrial production of N-acyl taurates?
A: Yes, the process is designed for industrial feasibility by simplifying the workflow into a single reactor step, removing the need for intermediate isolation of dangerous acyl chlorides, and utilizing readily available raw materials like fatty acids and sodium isethionate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fatty Acyl Amino Acid Surfactant Supplier
The transition to greener, more efficient chemical processes is no longer optional but a necessity for long-term viability in the fine chemical sector. NINGBO INNO PHARMCHEM stands at the forefront of this transition, leveraging advanced technologies like the one described in CN110981758A to deliver superior surfactant solutions. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale innovation to market-ready product is seamless. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying the absence of halogenated impurities, guaranteeing that every batch meets the exacting standards required for personal care and pharmaceutical applications.
We invite you to explore how this innovative synthesis can optimize your supply chain and reduce your environmental footprint. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your growth. Let us help you engineer a more sustainable and profitable future for your surfactant portfolio.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
