Advanced Room-Temperature Synthesis of Liquid Tungsten ALD Precursors for Semiconductor Manufacturing
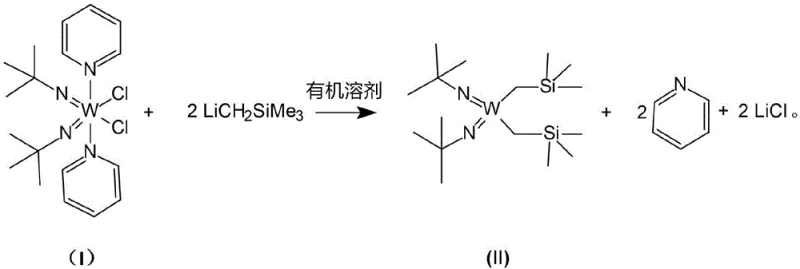
The semiconductor industry continuously demands advanced materials that enable precise thin-film deposition with minimal thermal budget and maximum conformity. Patent CN115651026A introduces a groundbreaking preparation method for a liquid tungsten complex, specifically W(NtBu)2(CH2TMS)2, which serves as a critical Atomic Layer Deposition (ALD) precursor. This innovation addresses the longstanding challenges associated with synthesizing high-purity organometallic tungsten species by replacing expensive, cryogenically dependent protocols with a robust, ambient-temperature ligand exchange strategy. By utilizing the mature and cost-effective W-0.5 intermediate (W(NtBu)2Cl2Py2) as the starting substrate, the process significantly lowers the barrier to entry for producing electronic-grade precursors. The resulting material exhibits exceptional metal purity, reaching levels up to 5N (99.9998%), which is paramount for preventing contamination in sensitive semiconductor device fabrication. This technical leap not only optimizes the chemical profile of the precursor but also fundamentally reshapes the economic and operational landscape for suppliers of electronic chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for tungsten-carbon precursors, such as the production of W(NtBu)2(CH2TMS)2, have historically relied on the use of Bis(tert-butylamido)bis(dimethylamido)tungsten(VI) (BTBMW) as the starting material. This conventional pathway necessitates the use of Grignard reagents, specifically TMS-methylmagnesium chloride, which must be added slowly to a reaction mixture cooled to extreme cryogenic temperatures of -78°C using acetone and dry ice baths. Maintaining such low temperatures over extended reaction periods, often spanning 16 hours, imposes a substantial energy burden and requires specialized, expensive equipment capable of sustaining deep freeze conditions. Furthermore, the handling of Grignard reagents introduces significant safety risks due to their pyrophoric nature and the potential for violent exothermic reactions if the addition rate is not meticulously controlled. The complexity of the workup procedure, involving filtration through celite and multiple concentration steps, often leads to lower overall yields and complicates the purification process, making it less ideal for the rigorous demands of high-volume semiconductor manufacturing.
The Novel Approach
In stark contrast, the methodology disclosed in the patent leverages a direct displacement reaction between the W-0.5 intermediate and trimethylsilylmethyllithium (LiCH2SiMe3) under remarkably mild conditions. This innovative route operates effectively at temperatures ranging from 25°C to 60°C, completely eliminating the need for energy-intensive cryogenic cooling systems. The reaction proceeds smoothly in common organic solvents such as toluene or hexane, where the byproduct, lithium chloride, precipitates out of the solution, driving the equilibrium towards the desired product and simplifying the isolation process. The operational simplicity allows for straightforward feeding without the need for slow, dropwise addition protocols, thereby reducing the total process time and minimizing the risk of thermal runaway. This shift from a hazardous, low-temperature Grignard process to a safe, ambient-temperature lithiation process represents a paradigm shift in precursor manufacturing, offering a scalable and economically superior alternative for producing high-performance ALD materials.
Mechanistic Insights into Ligand Exchange and Purification
The core chemical transformation involves a nucleophilic substitution where the trimethylsilylmethyl anion acts as a potent nucleophile, attacking the tungsten center to displace the chloride ligands present in the W-0.5 starting material. Unlike the bulky dimethylamido groups in the conventional BTBMW route, the chloride leaving groups in the W-0.5 complex are more readily displaced by the organolithium reagent, facilitating a cleaner and more efficient conversion. The reaction mechanism benefits from the hard-soft acid-base principles, where the lithium cation strongly coordinates with the chloride anions to form insoluble lithium chloride salts. This precipitation event is crucial as it removes the byproduct from the reaction equilibrium, preventing reverse reactions and ensuring high conversion rates even without excessive reagent loading. The use of an inert nitrogen atmosphere throughout the process protects the sensitive organometallic intermediates from oxidation and hydrolysis, preserving the integrity of the tungsten-carbon bonds which are essential for the precursor's volatility and thermal stability during the subsequent ALD cycle.
Purity control is intrinsically built into this synthetic design through the physical properties of the reaction components. The formation of solid lithium chloride allows for its removal via simple filtration or decantation after solvent evaporation and alkane extraction, effectively separating the inorganic salt from the organic tungsten complex. Following this initial cleanup, the liquid nature of the crude product enables purification via vacuum distillation, a technique highly effective for removing volatile organic impurities and residual solvents. The patent data indicates that this combination of precipitation and distillation yields a product with metal purity as high as 99.9998%, a specification that is critical for preventing metallic contamination in logic and memory devices. The absence of magnesium salts, which are typical byproducts of Grignard reactions and can be difficult to remove completely, further enhances the final purity profile, ensuring that the precursor meets the stringent requirements for depositing uniform tungsten oxide films with excellent electrical properties.
How to Synthesize W(NtBu)2(CH2TMS)2 Efficiently
The synthesis of this advanced tungsten precursor is designed to be operationally simple while maintaining rigorous control over reaction parameters to ensure reproducibility and safety. The process begins with the careful preparation of anhydrous and oxygen-free solvents, as moisture can degrade the organolithium reagent and compromise the quality of the final product. The W-0.5 intermediate is dissolved in a suitable organic solvent like toluene within a Schlenk flask under a positive pressure of nitrogen gas. Subsequently, the trimethylsilylmethyllithium solution is introduced to the reaction vessel, where the mixture is allowed to stir at ambient or slightly elevated temperatures for an extended period to ensure complete conversion. Detailed standardized synthesis steps, including specific molar ratios, solvent volumes, and purification parameters, are outlined in the technical guide below to assist process engineers in replicating this high-efficiency route.
- Dissolve the W-0.5 intermediate (W(NtBu)2Cl2Py2) in a dried organic solvent such as toluene under an inert nitrogen atmosphere.
- Add trimethylsilylmethyllithium solution dropwise to the reaction mixture and stir at room temperature (25-60°C) for 12 to 24 hours.
- Remove the solvent under reduced pressure, extract the residue with alkane, filter off the lithium chloride precipitate, and purify the liquid product via vacuum distillation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthesis route offers compelling strategic benefits that extend beyond mere chemical efficiency. The elimination of cryogenic cooling requirements translates directly into significant reductions in utility costs and capital expenditure on specialized low-temperature reactors, making the manufacturing process more economically viable at scale. By replacing the expensive BTBMW starting material with the more accessible and cheaper W-0.5 intermediate, the raw material cost base is substantially lowered, allowing for more competitive pricing structures in the volatile electronic chemicals market. Furthermore, the simplified operational protocol, which does not require slow, monitored addition of reagents or maintenance of sub-zero temperatures, reduces the labor intensity and batch cycle time, thereby increasing the overall throughput of the production facility. These factors collectively contribute to a more resilient supply chain capable of responding rapidly to the fluctuating demands of the semiconductor industry.
- Cost Reduction in Manufacturing: The abandonment of energy-intensive dry ice and acetone baths removes a major variable cost associated with traditional precursor synthesis, while the use of commodity-grade solvents and cheaper starting materials further drives down the cost of goods sold. The simplified workup procedure, which relies on filtration and distillation rather than complex chromatographic separations, minimizes solvent consumption and waste disposal costs. Additionally, the higher stability of the reaction reduces the risk of batch failures due to thermal excursions, ensuring consistent yield and reducing the financial impact of lost production time. This holistic approach to cost optimization makes the new method highly attractive for manufacturers seeking to improve margins without compromising on product quality.
- Enhanced Supply Chain Reliability: Sourcing the W-0.5 intermediate is generally more reliable than procuring specialized amido-tungsten complexes, as the former is a well-established material with multiple potential supply sources. The robustness of the room-temperature reaction means that production is less susceptible to disruptions caused by equipment failure in cooling systems or shortages of dry ice, which can be logistical bottlenecks in certain regions. The ability to operate under standard inert atmosphere conditions using widely available nitrogen supplies ensures that the manufacturing process can be easily replicated across different geographic locations, diversifying the supply base and mitigating regional risks. This reliability is crucial for long-term contracts with semiconductor fabs that require guaranteed continuity of supply for their critical deposition processes.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of extreme thermal gradients and the manageable exothermicity of the reaction. The generation of solid lithium chloride as the primary byproduct simplifies waste management compared to the complex aqueous workups often required for magnesium salts, aligning better with green chemistry principles and environmental regulations. The liquid nature of the final product facilitates easy handling, storage, and transportation in standard containers, reducing the logistical complexities associated with shipping solid or highly air-sensitive materials. These attributes make the technology ideally suited for rapid capacity expansion to meet the growing global demand for high-k dielectric and conductive films in advanced node semiconductor manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this tungsten precursor technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on performance metrics and operational feasibility. Understanding these details is essential for technical teams evaluating the integration of this new material into their existing ALD process flows.
Q: What are the primary advantages of this new tungsten precursor synthesis method?
A: The novel method utilizes the cost-effective W-0.5 intermediate instead of expensive BTBMW and operates at room temperature, eliminating the need for energy-intensive cryogenic cooling (-78°C) required by conventional Grignard-based routes.
Q: How is high metal purity achieved in this process?
A: High purity (up to 5N) is achieved through the formation of insoluble lithium chloride byproducts which are easily removed via filtration, followed by vacuum distillation of the liquid tungsten complex.
Q: Is this precursor suitable for large-scale ALD applications?
A: Yes, the mild reaction conditions, simple feeding operations, and liquid state of the final product make it highly suitable for commercial scale-up and consistent atomic layer deposition of tungsten oxide films.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable W(NtBu)2(CH2TMS)2 Supplier
As the semiconductor industry pushes towards smaller nodes and more complex 3D architectures, the demand for high-purity, liquid ALD precursors like W(NtBu)2(CH2TMS)2 is accelerating rapidly. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver this critical electronic chemical. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced purification units capable of meeting stringent purity specifications, ensuring that every batch of tungsten complex we supply performs consistently in your deposition tools. We understand that in the world of electronic materials, trace impurities can dictate device yield, which is why our commitment to quality control is unwavering and deeply integrated into our manufacturing culture.
We invite you to collaborate with us to optimize your supply chain for tungsten oxide film deposition. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how switching to this novel precursor can reduce your total cost of ownership. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with NINGBO INNO PHARMCHEM, you secure not just a supplier, but a strategic ally dedicated to supporting your innovation in semiconductor manufacturing with reliable, high-performance materials.
