Revolutionizing 2 2 Dithiodibenzothiazole Production via Green Oxygen Oxidation Technology
The global demand for high-performance rubber accelerators continues to drive innovation in the synthesis of key intermediates such as 2 2'-dithiodibenzothiazole commonly known as DM. Patent CN101139338B represents a pivotal technological breakthrough by introducing an oxygen-based oxidation process that fundamentally alters the production landscape for this critical chemical. Unlike traditional methods that rely on hazardous oxidants this new approach utilizes molecular oxygen or air in the presence of a copper catalyst to convert 2-mercaptobenzothiazole ammonium salt directly into the desired disulfide product. This shift not only addresses long-standing environmental concerns regarding wastewater and exhaust emissions but also offers a streamlined pathway that bypasses the need for expensive raw material refinement. For industry leaders seeking a reliable rubber additive supplier understanding the mechanistic and commercial implications of this patent is essential for optimizing supply chains and reducing manufacturing overheads in the competitive specialty chemicals market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically the industrial production of 2 2'-dithiodibenzothiazole has been plagued by significant environmental and safety challenges inherent to older oxidation technologies. The most common conventional method involves the use of sodium nitrite as an oxidant which inevitably leads to the generation of nitrogen oxide gases that are toxic and contribute to atmospheric pollution. Furthermore this process typically requires the raw material 2-mercaptobenzothiazole to be highly refined beforehand using caustic soda and sulfuric acid which produces wastewater with high salt concentrations that poison biological sewage treatment systems. Alternative methods utilizing chlorine oxidation present even greater risks including the potential for explosion when chlorine mixes with air and the handling of highly toxic chlorine gas which demands complex and expensive safety equipment. These legacy processes result in unstable product quality and impose heavy compliance burdens on manufacturers making them increasingly unsustainable in a regulatory environment that prioritizes green chemistry and zero-emission standards.
The Novel Approach
The innovative process detailed in the patent data overcomes these deficiencies by employing a direct oxygen oxidation strategy within an ammoniacal system. By dissolving crude 2-mercaptobenzothiazole directly in an ammonia solution the method eliminates the preliminary acid-base refining step thereby reducing both material consumption and waste generation. The reaction proceeds under controlled temperature and pressure conditions where air or oxygen serves as the clean oxidant producing only water and ammonia as by-products which can be easily recycled back into the system. This closed-loop design ensures that there is no wastewater discharge and no harmful exhaust emissions transforming the production profile from a high-pollution operation to a clean manufacturing process. Additionally the ability to use crude raw materials without prior purification significantly lowers the entry cost for production while maintaining high product purity levels above 90% which meets the stringent requirements of downstream rubber applications.
Mechanistic Insights into Copper-Catalyzed Oxidative Coupling
The core of this technological advancement lies in the formation and subsequent oxidation of the 2-mercaptobenzothiazole ammonium salt intermediate. In the first stage the thiol group of the raw material reacts with ammonia in an aqueous environment to form a soluble ammonium salt which allows for the effective separation of insoluble impurities through simple filtration. This solubilization step is critical as it ensures that only the reactive species enter the oxidation reactor preventing contamination of the final product. Once filtered the solution is subjected to oxidative coupling in the presence of a copper-based catalyst such as copper sulfate or neutralized verdigris. The catalyst facilitates the transfer of electrons from the sulfur atoms to the molecular oxygen enabling the formation of the disulfide bond that characterizes the 2 2'-dithiodibenzothiazole structure. This catalytic cycle is highly efficient and operates under mild thermal conditions typically between 50°C and 80°C which minimizes energy consumption and prevents thermal degradation of the product.
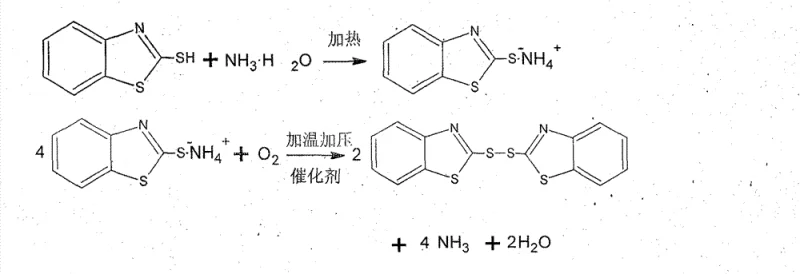
Controlling impurity profiles is another crucial aspect of this mechanism particularly given the use of crude starting materials. The selective solubility of the ammonium salt ensures that non-reactive organic impurities remain in the solid filter cake and are removed before the oxidation step begins. Furthermore the specific choice of copper catalysts and the precise control of reaction parameters such as pressure between 0.4 MPa and 1.0 MPa prevent over-oxidation which can lead to colored by-products and reduced yield. The regeneration of ammonia during the reaction allows it to be recovered and reused creating a sustainable cycle that reduces the need for continuous fresh reagent input. This level of control over the reaction environment ensures consistent batch-to-batch quality which is vital for pharmaceutical and automotive rubber applications where specification adherence is non-negotiable for any credible fine chemical intermediates supplier.
How to Synthesize 2 2-Dithiodibenzothiazole Efficiently
Implementing this synthesis route requires careful attention to the dissolution and oxidation parameters to maximize yield and safety. The process begins with the preparation of the ammonium salt solution followed by catalytic oxidation and final isolation of the solid product. Operators must maintain strict control over the ammonia concentration and temperature during the dissolution phase to ensure complete solubilization of the crude thiol. Following filtration the oxidation reaction is driven by oxygen pressure and catalyst activity requiring robust reactor design capable of handling pressurized gases safely. The detailed standardized synthesis steps see the guide below outline the precise operational sequence required to replicate the high yields reported in the patent data ensuring that production teams can achieve optimal results consistently.
- Dissolve crude 2-mercaptobenzothiazole in aqueous ammonia solution at 40-70°C to form the ammonium salt.
- Filter the solution to remove insoluble impurities and add a copper-based catalyst such as copper sulfate or neutralized verdigris.
- Introduce air or oxygen at 0.4-1.0 MPa pressure and maintain temperature between 50-80°C to complete the oxidative coupling reaction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors the adoption of this oxygen-based process translates into tangible strategic advantages that extend beyond simple unit price considerations. The elimination of the refining step for raw materials means that manufacturers can source lower grade and therefore less expensive crude 2-mercaptobenzothiazole without compromising the quality of the final output. This flexibility in raw material sourcing enhances supply chain resilience by broadening the pool of eligible vendors and reducing dependency on high-purity feedstocks that may be subject to market volatility. Moreover the removal of hazardous reagents like sodium nitrite and chlorine simplifies logistics and storage requirements lowering the overall operational risk profile of the facility. These factors combined create a more robust and cost-effective supply model that aligns with modern corporate sustainability goals and long-term cost reduction in rubber additives manufacturing strategies.
- Cost Reduction in Manufacturing: The most significant economic benefit arises from the ability to bypass the traditional acid-base refining process which typically consumes large quantities of caustic soda and sulfuric acid. By utilizing crude raw materials directly the process saves on both reagent costs and the associated waste disposal fees for high-salt wastewater. Additionally the recycling of ammonia and water within the closed system drastically reduces utility consumption and fresh water intake leading to substantial cost savings over the lifecycle of the plant. The use of minimal amounts of inexpensive copper catalysts further contributes to a leaner cost structure allowing manufacturers to offer more competitive pricing to their clients while maintaining healthy margins.
- Enhanced Supply Chain Reliability: Safety is a critical component of supply chain continuity and this process significantly mitigates the risks associated with storing and handling dangerous chemicals like chlorine gas or sodium nitrite. The use of air or oxygen as the oxidant removes the threat of toxic gas leaks and explosive mixtures ensuring uninterrupted production schedules. Furthermore the simplified workflow with fewer unit operations reduces the likelihood of mechanical failures or process bottlenecks that could delay shipments. This reliability is essential for reducing lead time for high-purity rubber additives and ensures that downstream customers receive their orders on time without the disruption often caused by environmental shutdowns or safety incidents.
- Scalability and Environmental Compliance: As environmental regulations become increasingly stringent globally the ability to operate with zero wastewater and exhaust emissions provides a distinct competitive advantage. This process is inherently scalable because it does not rely on complex waste treatment infrastructure that often limits plant expansion. The absence of toxic by-products means that facilities can operate in regions with strict ecological protections without fear of regulatory penalties. This future-proofs the investment and ensures long-term commercial scale-up of complex rubber additives is viable. Companies adopting this technology demonstrate a commitment to sustainability which is increasingly becoming a prerequisite for partnerships with major multinational corporations in the automotive and industrial sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this oxygen oxidation technology. These answers are derived directly from the experimental data and process descriptions found in the patent documentation to provide accurate guidance for technical teams. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into their existing production lines or sourcing strategies for improved efficiency and compliance.
Q: How does the oxygen oxidation method improve environmental compliance compared to traditional sodium nitrite processes?
A: The traditional sodium nitrite method generates toxic nitrogen oxide gases and high-salt wastewater containing sulfates which are difficult to treat biologically. In contrast the oxygen oxidation process described in patent CN101139338B operates in a closed loop where ammonia is recycled and no wastewater or exhaust gas is emitted significantly reducing the environmental burden and regulatory risks for manufacturers.
Q: Can this process utilize crude raw materials without prior refinement?
A: Yes one of the primary economic advantages of this technology is its ability to dissolve crude 2-mercaptobenzothiazole directly in ammonia solution. The filtration step effectively removes impurities before the oxidation reaction eliminates the need for a separate and costly acid-base refining step required by conventional methods thereby simplifying the workflow and lowering raw material costs.
Q: What catalysts are most effective for this oxidative coupling reaction?
A: Experimental data indicates that copper-series catalysts provide superior performance in this system. Specifically copper sulfate manganese acetate and neutralized verdigris have been tested with neutralized verdigris showing particularly high yields and melting points. The catalyst loading is minimal typically ranging from 0.05% to 0.2% of the ammonium salt solution weight ensuring cost efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2 2-Dithiodibenzothiazole Supplier
At NINGBO INNO PHARMCHEM we recognize that the transition to greener and more efficient chemical processes is vital for the future of the industry. As a leading CDMO partner we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production ensuring that innovative laboratory techniques like the oxygen oxidation process can be successfully translated into large-scale manufacturing realities. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications guaranteeing that every batch of 2 2'-dithiodibenzothiazole meets the highest international standards. We are committed to leveraging advanced catalytic technologies to deliver products that not only perform exceptionally but also align with our clients' sustainability and cost-efficiency objectives.
We invite you to collaborate with us to explore how this advanced manufacturing route can benefit your specific application needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements demonstrating exactly how this process can optimize your budget. Please contact us today to request specific COA data and route feasibility assessments that will empower you to make informed decisions for your supply chain. Let us be your partner in driving innovation and efficiency in the global rubber additives market.
