Advanced Pterene-Modified Platinum Complexes for Next-Generation OLED Manufacturing
Advanced Pterene-Modified Platinum Complexes for Next-Generation OLED Manufacturing
The rapid evolution of Organic Light Emitting Diodes (OLEDs) has positioned them as the dominant technology for next-generation displays and lighting solutions. However, the industry continues to face significant challenges regarding the stability and efficiency of blue phosphorescent emitters, which are critical for achieving full-color displays with long operational lifetimes. Patent CN110862350B introduces a groundbreaking class of pterene pyridazine tetradentate platinum complex phosphorescent materials designed to overcome these limitations. By integrating a rigid, non-conjugated stereo pterene structure that wraps around the platinum core, this innovation effectively suppresses the concentration quenching effect typically caused by intermolecular stacking. This structural modification not only maximizes the luminous efficiency and color purity of the platinum complex but also ensures high internal and external quantum yields, making it a highly attractive candidate for reliable OLED material supplier networks seeking high-performance emitters.
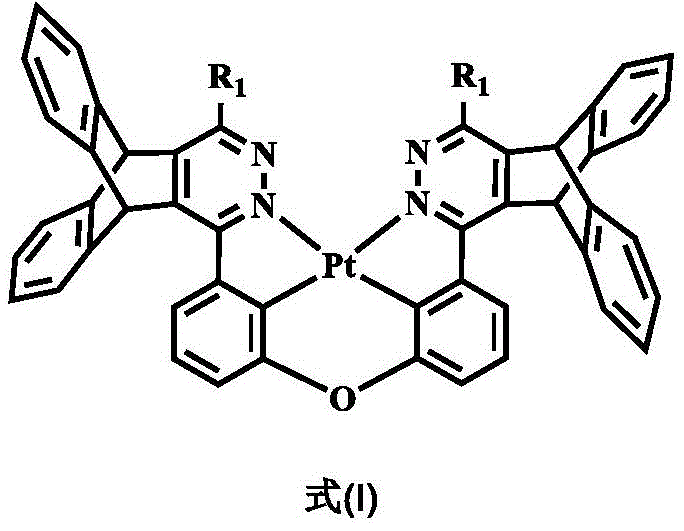
The technical breakthrough lies in the specific molecular architecture where the bulky pterene moiety acts as a steric shield. In traditional planar platinum complexes, the tendency for molecules to aggregate in the solid state often leads to the formation of excimers or aggregates that quench luminescence, particularly at high concentrations required for efficient device operation. The invention described in CN110862350B mitigates this by enforcing spatial separation between the emissive centers without disrupting the conjugation necessary for charge transport. This balance allows for the fabrication of electroluminescent devices with superior brightness and stability, addressing a critical pain point for manufacturers aiming to reduce lead time for high-purity display materials while maintaining rigorous quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of phosphorescent emitters for OLEDs has relied heavily on iridium (III) and platinum (II) complexes with bidentate or tridentate ligands. While these materials have shown promise, they often suffer from inherent structural flexibility that allows for vibrational and rotational relaxation around the metal center, leading to significant non-radiative energy loss. Furthermore, the square planar geometry of Pt(II) complexes makes them particularly prone to intermolecular interactions, resulting in severe concentration quenching at the doping levels necessary for practical device fabrication. Conventional synthesis routes for these complexes often involve harsh conditions or expensive precursors that complicate the supply chain and increase the overall cost reduction in electronic chemical manufacturing efforts. The lack of sufficient steric bulk in traditional ligands means that achieving high efficiency often requires very low doping concentrations, which can compromise charge balance and device uniformity.
The Novel Approach
The novel approach detailed in the patent leverages the unique properties of triptycene-derived pterene structures to create a massive steric hindrance space around the active platinum site. By synthesizing a symmetrical tetradentate ligand system based on pyridazine and modifying it with this three-dimensional pterene framework, the inventors have created a complex that maintains high rigidity. This rigidity is crucial as it locks the molecular conformation, minimizing energy loss through molecular motion. The synthesis strategy involves a modular approach where various functional groups can be introduced to fine-tune the electronic properties without sacrificing the steric protection. This flexibility allows for the customization of emission colors and energy levels, providing a versatile platform for developing red, green, and potentially stable blue emitters. The result is a material that offers high luminous brightness and efficiency while being chemically stable and easier to process via spin-coating methods.
Mechanistic Insights into Tetradentate Platinum Coordination
The core mechanism driving the superior performance of these materials is the formation of a robust tetradentate coordination environment around the platinum (II) ion. Unlike octahedral Ir(III) complexes, the square planar Pt(II) center in this system is coordinated by nitrogen atoms from the pyridazine rings and carbon atoms from the phenyl bridges, creating a highly stable chelate effect. The introduction of the pterene unit does not participate directly in the pi-conjugation of the emissive core, which preserves the intrinsic electronic transitions responsible for phosphorescence. Instead, it functions purely as a steric modulator. Theoretical calculations and electrochemical studies indicate that the Highest Occupied Molecular Orbital (HOMO) is primarily distributed over the central platinum atom and the connected benzene rings, while the Lowest Unoccupied Molecular Orbital (LUMO) resides mainly on the pyridazine rings. This separation facilitates efficient Metal-to-Ligand Charge Transfer (MLCT), which is the primary pathway for phosphorescent emission in these heavy metal complexes.
Furthermore, the suppression of concentration quenching is mechanistically attributed to the physical barrier created by the pterene wings. In the solid state or in high-concentration films, these bulky groups prevent the planar aromatic surfaces of adjacent molecules from coming into close contact, thereby inhibiting the pi-pi stacking interactions that typically lead to non-radiative decay channels. This mechanism ensures that the triplet excitons generated during electrical excitation remain localized on individual molecules long enough to undergo radiative decay. The patent data demonstrates that complexes like Pt-DPT can achieve current efficiencies as high as 44.66 cd/A at optimal doping concentrations, validating the effectiveness of this steric engineering strategy. For R&D teams, understanding this structure-property relationship is vital for designing future generations of emitters that push the boundaries of efficiency and lifetime.
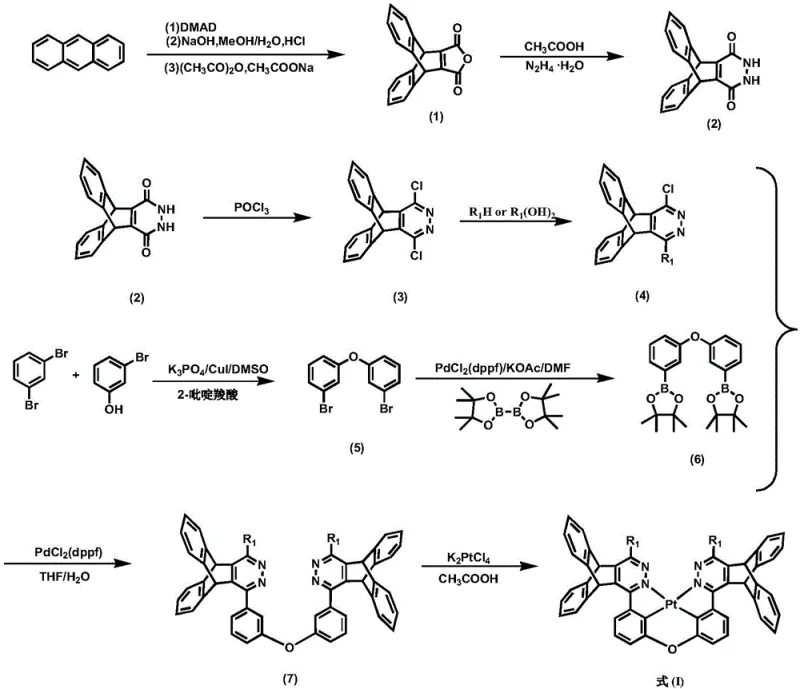
How to Synthesize Pterene-Modified Pyridazine Platinum Complex Efficiently
The synthesis of these advanced materials follows a logical, step-wise progression that begins with readily available commodity chemicals and builds up complexity through well-established organic transformations. The process starts with the Diels-Alder reaction of anthracene with dimethyl butynedioate (DMAD) to form the foundational pterene skeleton, followed by hydrolysis and cyclization to generate the pyridazine core. Subsequent functionalization via palladium-catalyzed cross-coupling reactions allows for the precise installation of the bridging units and substituent groups. The final step involves the coordination of the pre-synthesized tetradentate ligand with potassium tetrachloroplatinate under controlled thermal conditions. This route is designed to be robust and scalable, avoiding the use of exotic reagents that could hinder commercial adoption. For detailed procedural specifics, please refer to the standardized guide below.
- Cyclize anthracene with dimethyl butynedioate (DMAD) followed by hydrolysis and dehydration to form the anhydride derivative.
- React the anhydride with hydrazine hydrate to form the diketone, then chlorinate with phosphorus oxychloride to obtain the dichloropyridazine intermediate.
- Perform Suzuki coupling to attach aryl groups, synthesize the bridging diphenyl ether unit, and finally coordinate with potassium tetrachloroplatinate to form the final complex.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this pterene-modified platinum complex technology offers several strategic benefits that align with the goals of cost optimization and supply security. The synthesis route relies on bulk chemicals like anthracene and standard solvents, which are widely available in the global chemical market, reducing the risk of raw material shortages. The modular nature of the synthesis also means that production can be easily scaled from laboratory grams to multi-tonne annual capacities without requiring fundamentally new equipment or processes. This scalability is a key factor for ensuring supply continuity for large-scale display panel manufacturers who require consistent quality and volume over long product lifecycles.
- Cost Reduction in Manufacturing: The synthetic pathway eliminates the need for extremely expensive or scarce metal precursors beyond standard platinum salts, and the high experimental yields reported in the patent examples suggest a material-efficient process. By avoiding complex purification steps often associated with unstable intermediates, the overall production cost is significantly reduced. The ability to use spin-coating for device fabrication further lowers the capital expenditure required for manufacturing lines compared to vacuum deposition techniques, offering substantial cost savings in the final device assembly.
- Enhanced Supply Chain Reliability: The use of stable intermediates and robust reaction conditions minimizes the risk of batch failures, which is a common disruption point in fine chemical supply chains. The chemical stability of the final platinum complex ensures a long shelf life, allowing for strategic stockpiling and reducing the pressure on just-in-time delivery schedules. This reliability is crucial for maintaining the production timelines of consumer electronics where display components are on the critical path.
- Scalability and Environmental Compliance: The process avoids the use of highly toxic or environmentally hazardous reagents where possible, utilizing standard organic solvents that can be recovered and recycled. The high efficiency of the resulting material means that less emitter is needed per device to achieve the same brightness, reducing the total mass of precious metal required per unit. This aligns with increasingly stringent environmental regulations and corporate sustainability goals, making it a compliant choice for green manufacturing initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers evaluating this material for integration into their product lines.
Q: How does the pterene structure improve OLED performance?
A: The rigid, non-conjugated stereo pterene structure introduces significant steric hindrance around the platinum core. This physically prevents intermolecular pi-pi stacking, effectively suppressing concentration quenching and excited dimer formation, which leads to higher luminous efficiency and color purity.
Q: What are the key advantages of this tetradentate ligand system?
A: Unlike bidentate ligands, the tetradentate design creates a rigid square planar geometry that inhibits vibration and rotation around the metal ion. This reduces non-radiative decay pathways, resulting in improved thermal stability and higher phosphorescence quantum efficiency suitable for commercial display applications.
Q: Is the synthesis scalable for industrial production?
A: Yes, the synthesis utilizes readily available starting materials like anthracene and employs standard organic reactions such as Diels-Alder cyclization, nucleophilic substitution, and palladium-catalyzed coupling. The reported experimental yields are robust, indicating strong potential for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pterene Platinum Complex Supplier
As the demand for high-efficiency OLED materials continues to surge, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM provides a distinct competitive advantage. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from R&D to mass manufacturing is seamless. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required for electronic grade materials, guaranteeing that every batch of pterene platinum complex delivers the consistent performance necessary for high-end display applications.
We invite you to engage with our technical procurement team to discuss how this innovative material can optimize your current product portfolio. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this sterically hindered platinum emitter. We are ready to provide specific COA data and route feasibility assessments tailored to your unique manufacturing requirements, helping you secure a reliable supply of next-generation phosphorescent materials.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
