Advanced Copper-Catalyzed Synthesis of N-Methyl-3-Methylmercapto-4-Amino Maleimide Derivatives for Commercial Scale
Advanced Copper-Catalyzed Synthesis of N-Methyl-3-Methylmercapto-4-Amino Maleimide Derivatives for Commercial Scale
The pharmaceutical industry continuously seeks robust synthetic methodologies for constructing complex heterocyclic scaffolds, particularly maleimide derivatives which serve as critical pharmacophores in kinase inhibitors and anticancer agents. Patent CN111440109A introduces a groundbreaking preparation method for N-methyl-3-methylmercapto-4-amino maleimide compounds, utilizing a transition metal copper-catalyzed tandem reaction under oxygen conditions. This innovation replaces traditional, hazardous thiolation reagents with stable, odorless methyl Bunte salts, marking a significant leap forward in green chemistry and process safety. The technology enables the direct functionalization of N-methylmaleimide with secondary amines and sulfur sources in a single pot, achieving exceptional yields and purity profiles that are essential for downstream drug development. By leveraging this advanced catalytic system, manufacturers can access high-value intermediates with reduced environmental impact and operational complexity.
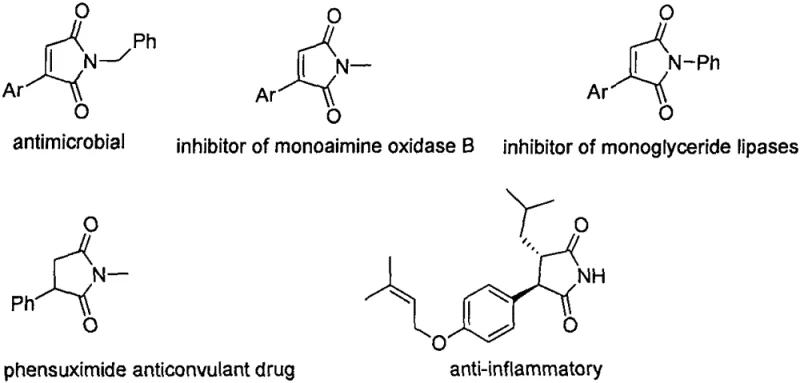
Maleimide scaffolds are ubiquitous in medicinal chemistry, appearing in natural products and clinical drugs due to their ability to act as Michael acceptors and protein binders. As illustrated in the structural diversity of bioactive maleimides, the capacity to introduce specific substituents at the 3 and 4 positions is paramount for tuning biological activity. However, conventional synthetic routes often suffer from harsh conditions or limited substrate scope. The present invention addresses these challenges by providing a versatile platform for synthesizing 3,4-heteroatom-functionalized maleimides. This capability is crucial for R&D teams aiming to explore structure-activity relationships (SAR) efficiently. The method's compatibility with various secondary amines allows for the rapid generation of diverse libraries, accelerating the discovery of novel therapeutic candidates while maintaining high standards of chemical integrity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
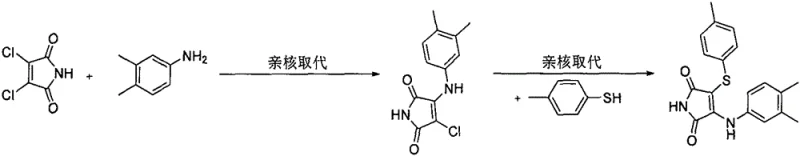
Historically, the synthesis of 3-amino-4-mercapto maleimide derivatives has relied on nucleophilic substitution reactions using 3,4-dichloromaleimide as a starting material. As depicted in earlier literature from 2002, this approach requires the use of expensive and less accessible dichlorinated precursors, which significantly drives up raw material costs. Furthermore, the introduction of the sulfur moiety typically involves thiophenols or aliphatic thiols, which are notorious for their intense, unpleasant odors and potential toxicity, posing severe health and safety risks in a manufacturing environment. These volatile sulfur compounds are also prone to oxidation, leading to disulfide impurities that complicate purification and reduce overall yield. Additionally, the requirement for strong bases and reflux conditions can lead to side reactions and degradation of sensitive functional groups, limiting the scope of compatible amines.
The Novel Approach
In stark contrast to legacy methods, the patented process employs a copper-catalyzed three-component coupling reaction that directly utilizes N-methylmaleimide, a secondary amine, and a methyl Bunte salt. This strategy eliminates the need for pre-functionalized dichloromaleimides, thereby reducing the step count and associated costs. The use of methyl Bunte salt (sodium S-methyl thiosulfate) as the sulfur source is a game-changer; it is a stable, odorless solid that is easy to weigh and handle, effectively removing the safety hazards associated with liquid thiols. The reaction proceeds smoothly under an oxygen atmosphere, which acts as a green oxidant to drive the catalytic cycle without generating stoichiometric toxic waste. This streamlined approach not only simplifies the workflow but also enhances the atom economy of the process, making it an ideal candidate for sustainable industrial application.
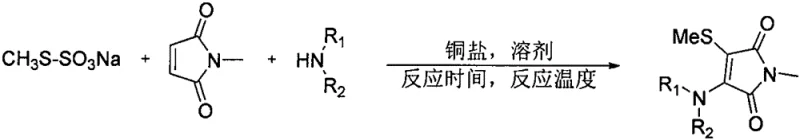
Mechanistic Insights into Copper-Catalyzed C-H Thioamination
The core of this transformation lies in the efficient activation of the C(sp2)-H bond on the maleimide ring by the copper catalyst. Mechanistically, the cuprous iodide likely coordinates with the nitrogen of the maleimide or the amine, facilitating the deprotonation and subsequent formation of a copper-amido or copper-enamide intermediate. The presence of molecular oxygen is critical, as it re-oxidizes the copper species back to its active state after the reductive elimination step, ensuring the catalytic turnover continues without the need for stoichiometric oxidants. The Bunte salt serves as a masked thiol equivalent, undergoing cleavage under the reaction conditions to release the reactive sulfur species in situ, which then attacks the activated maleimide core. This concerted mechanism ensures high regioselectivity, favoring the formation of the 3-mercapto-4-amino substitution pattern observed in the products.
From an impurity control perspective, the mildness of the reaction conditions plays a vital role in maintaining product quality. Unlike harsh nucleophilic substitutions that can lead to over-substitution or hydrolysis of the imide ring, this catalytic system operates at moderate temperatures (100°C) in a non-protic solvent like 1,2-dichloroethane. This environment minimizes the formation of polymeric byproducts or degradation species. The high specificity of the copper catalyst ensures that the amine and sulfur groups are installed sequentially or concertedly with precision, resulting in a crude reaction mixture that is relatively clean. Consequently, downstream purification via standard column chromatography or crystallization becomes more efficient, yielding products with purity levels that meet the stringent requirements for pharmaceutical intermediates.
How to Synthesize N-Methyl-3-Methylmercapto-4-Amino Maleimide Efficiently
To implement this synthesis effectively, precise control over reaction parameters is essential to maximize yield and reproducibility. The protocol involves mixing the three key components—methyl Bunte salt, the chosen secondary amine, and N-methylmaleimide—in a molar ratio of approximately 3:2:1 to drive the equilibrium towards completion. The choice of solvent is critical, with 1,2-dichloroethane proving superior to polar aprotic solvents like DMF or non-polar solvents like toluene, likely due to its ability to solubilize the ionic Bunte salt while maintaining the stability of the copper catalyst. The detailed standardized synthesis steps for this high-efficiency route are outlined in the guide below.
- Combine methyl Bunte salt, secondary amine, and N-methylmaleimide in 1,2-dichloroethane solvent with cuprous iodide catalyst.
- Purge the reaction vessel with oxygen gas three times to ensure an oxidative atmosphere essential for the catalytic cycle.
- Heat the mixture to 100°C for 24 hours, then cool, dilute with ethyl acetate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers tangible strategic benefits beyond mere chemical novelty. By shifting away from volatile thiols and expensive dichlorinated starting materials, the process inherently reduces the complexity of raw material sourcing and storage. The stability of Bunte salts means they can be purchased in bulk and stored for extended periods without degradation, mitigating the risk of supply disruptions caused by the shelf-life limitations of liquid thiols. Furthermore, the elimination of foul-smelling reagents simplifies facility requirements, potentially lowering the capital expenditure needed for specialized ventilation and scrubbing systems, which translates into significant operational cost savings over the lifecycle of the product.
- Cost Reduction in Manufacturing: The economic advantage of this route is driven primarily by the substitution of high-cost precursors with commodity chemicals. N-methylmaleimide and secondary amines are widely available and inexpensive compared to specialized 3,4-dichloromaleimides. Additionally, the use of a catalytic amount of copper iodide rather than stoichiometric reagents reduces the material cost per kilogram of product. The simplified workup procedure, which avoids complex extraction protocols needed to remove excess thiols, further lowers labor and utility costs. These factors combine to create a leaner manufacturing process that improves overall margin potential for the final API.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of robust, non-hazardous reagents. Methyl Bunte salts are stable solids that do not require cold chain logistics or special hazmat shipping classifications, unlike many liquid thiols which are classified as dangerous goods. This ease of transport expands the pool of potential suppliers and reduces lead times for raw material delivery. Moreover, the reaction's tolerance to oxygen and use of air-stable catalysts means that production is less susceptible to interruptions caused by strict inert atmosphere requirements, ensuring consistent output and reliable delivery schedules to downstream customers.
- Scalability and Environmental Compliance: Scaling this process to commercial volumes is facilitated by its straightforward engineering requirements. The reaction does not generate large quantities of acidic or toxic waste streams associated with thiol disposal, aligning well with modern environmental regulations and ESG goals. The use of oxygen as the oxidant produces water as the primary byproduct, minimizing the burden on wastewater treatment facilities. This green profile not only reduces compliance costs but also future-proofs the manufacturing process against tightening environmental legislation, making it a sustainable choice for long-term production of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this copper-catalyzed synthesis. These answers are derived directly from the experimental data and optimization studies presented in the patent documentation, providing clarity on reaction scope and troubleshooting.
Q: Why are Bunte salts preferred over thiols in this maleimide synthesis?
A: Bunte salts (S-sulfonates) are odorless, stable solids that are easier to handle and store compared to volatile, malodorous, and oxidation-prone thiols, significantly improving workplace safety and process stability.
Q: What is the role of the copper catalyst and oxygen in this reaction?
A: Cuprous iodide acts as the transition metal catalyst facilitating the C-H activation and bond formation, while molecular oxygen serves as the terminal oxidant to regenerate the active catalytic species, making the process greener and more atom-economical.
Q: Can this method be scaled for industrial production of API intermediates?
A: Yes, the method utilizes cheap, commercially available reagents and simple workup procedures like filtration and chromatography, avoiding expensive dichloromaleimide starting materials, which makes it highly suitable for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Methyl-3-Methylmercapto-4-Amino Maleimide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-quality intermediates for drug development pipelines. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to industrial manufacturing is seamless. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of maleimide derivative meets the highest international standards. Our commitment to technical excellence allows us to navigate the complexities of copper-catalyzed reactions, delivering products with consistent quality and reliability.
We invite pharmaceutical partners to collaborate with us to leverage this innovative synthetic route for their specific projects. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to determine how this advanced methodology can optimize your supply chain and accelerate your time to market.
