Advancing Cycloalkane Oxidation Efficiency with Novel Bimetallic Porphyrin Catalysts
The chemical industry continuously seeks advancements in oxidation technologies to enhance efficiency and sustainability, particularly in the production of key intermediates like cycloalkanols and cycloalkanones. Patent CN112090449B introduces a groundbreaking bimetallic central metalloporphyrin catalyst designed to address the longstanding challenges associated with cycloalkane catalytic oxidation. This innovation leverages a unique dual-metal center architecture to precisely control the reaction pathway, ensuring high selectivity for target alcohol and ketone products while minimizing the formation of unwanted aliphatic diacids. For R&D directors and process engineers, this represents a significant shift from traditional homogeneous salt catalysts, offering a more controllable and safer oxidation environment that aligns with modern green chemistry principles and stringent purity requirements for downstream pharmaceutical and polymer applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional industrial processes for cycloalkane oxidation predominantly rely on homogeneous cobalt or manganese salts operating under harsh conditions, typically requiring temperatures between 130°C and 160°C and pressures up to 3.0 MPa. These severe reaction parameters often lead to the uncontrollable thermal decomposition of cycloalkyl hydroperoxide intermediates, resulting in deep oxidation products such as adipic acid and other aliphatic diacids. The accumulation of these diacids not only reduces the yield of valuable KA oil (ketone-alcohol oil) but also causes significant operational issues, including pipeline blockage and equipment corrosion, which hinder continuous production capabilities. Furthermore, the high concentration of unstable hydroperoxides poses substantial safety risks, necessitating complex safety protocols and limiting the scalability of these conventional methods in large-scale chemical manufacturing facilities.
The Novel Approach
In contrast, the novel bimetallic metalloporphyrin catalyst system described in the patent operates effectively at lower temperatures ranging from 100°C to 150°C and moderate oxygen pressures, significantly mitigating the risks associated with high-energy oxidation processes. By utilizing a biomimetic approach inspired by cytochrome P-450 enzymes, this catalyst facilitates a more selective activation of molecular oxygen, directing the reaction pathway towards the desired cycloalkanol and cycloalkanone products with exceptional precision. The dual-metal center design allows for synergistic interactions that stabilize reaction intermediates, effectively suppressing the formation of aliphatic diacids and ensuring a cleaner product profile that requires less intensive downstream purification. This methodological shift not only enhances process safety by maintaining low hydroperoxide levels but also improves overall economic viability through reduced energy consumption and extended equipment lifespan.
Mechanistic Insights into Bimetallic Porphyrin Catalytic Oxidation
The core innovation lies in the sophisticated molecular architecture of the catalyst, which features two distinct metal centers linked by an alkyl chain, creating a cooperative catalytic environment that single-metal systems cannot achieve. As illustrated in the structural diagram below, the specific arrangement of metal ions such as Cobalt, Manganese, or Iron paired with Copper, Zinc, or Nickel within the porphyrin macrocycles enables precise modulation of the electronic properties at the active sites. This configuration is critical for managing the radical chain reactions inherent in autoxidation processes, allowing the catalyst to intervene specifically at the hydroperoxide decomposition stage to favor the formation of alcohols and ketones over carboxylic acids.
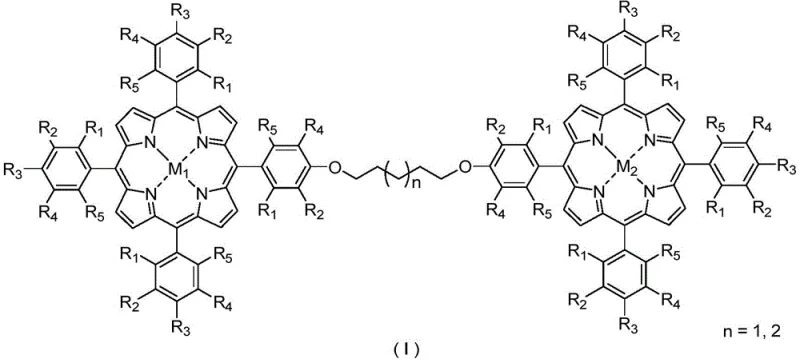
Furthermore, the mechanism involves the effective regulation of cycloalkyl hydroperoxide conversion, preventing its无序 thermal decomposition which is the primary source of selectivity loss in traditional systems. The planar structure of the metalloporphyrin ensures maximum exposure of the catalytic metal centers to the substrate, enabling high turnover frequencies even at extremely low catalyst loadings relative to the substrate mass. This high efficiency translates to reduced metal contamination in the final product, a crucial factor for pharmaceutical intermediate synthesis where heavy metal residues are strictly regulated. The ability to fine-tune the substituents on the porphyrin ring further allows chemists to optimize steric and electronic effects for specific cycloalkane substrates, providing a versatile platform for customizing oxidation processes across different chemical value chains.
How to Synthesize Bimetallic Central Metalloporphyrin Efficiently
The preparation of this advanced catalyst involves a straightforward yet precise coupling reaction between functionalized porphyrin monomers and dibromoalkane linkers in a polar aprotic solvent system. The process requires careful control of stoichiometry and reaction conditions to ensure the formation of the desired bimetallic dimer rather than polymeric byproducts, with purification steps designed to isolate the high-purity catalyst necessary for consistent performance. Detailed standardized synthesis steps see the guide below.
- Combine porphyrin M1(II), porphyrin M2(II), potassium carbonate, potassium iodide, and dibromoalkanes in N,N-dimethylformamide under nitrogen atmosphere.
- Stir the reaction mixture at temperatures between 50°C and 200°C for 12 to 120 hours to facilitate the coupling reaction.
- Filter, wash, purify, desolvate under reduced pressure, and vacuum dry the resulting solid to obtain the final bimetallic catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this bimetallic catalyst technology offers substantial strategic benefits by fundamentally altering the cost structure and reliability of cycloalkane oxidation operations. The significant reduction in aliphatic diacid byproducts means that less raw material is wasted on non-valuable outputs, directly improving the mass balance and yield of the primary KA oil products which are critical for nylon and pharmaceutical synthesis. Additionally, the milder reaction conditions reduce the energy burden on production facilities, leading to lower utility costs and a smaller carbon footprint, which is increasingly important for meeting corporate sustainability goals and regulatory compliance in global markets.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal salts and the reduction in downstream purification requirements due to higher selectivity contribute to a leaner manufacturing cost profile. By avoiding the formation of corrosive diacids, companies can extend the maintenance intervals for reactors and piping, reducing downtime and capital expenditure on corrosion-resistant materials. The high catalytic efficiency allows for lower catalyst loading per batch, further decreasing the direct material costs associated with the oxidation process while maintaining high throughput rates.
- Enhanced Supply Chain Reliability: The improved safety profile resulting from lower hydroperoxide accumulation reduces the risk of unplanned shutdowns due to safety incidents, ensuring more consistent delivery schedules for downstream customers. The robustness of the catalyst under solvent-free or low-solvent conditions simplifies logistics by reducing the volume of hazardous solvents that need to be transported, stored, and disposed of, thereby streamlining the supply chain workflow. This stability supports long-term production planning and inventory management, providing a competitive edge in markets where supply continuity is a primary differentiator.
- Scalability and Environmental Compliance: The technology is designed with scalability in mind, demonstrated by successful scale-up experiments that maintain selectivity and conversion rates from laboratory to pilot scales. The reduction in hazardous waste generation, particularly organic peroxides and acidic byproducts, simplifies environmental compliance and waste treatment processes, lowering the overall environmental liability of the manufacturing site. This alignment with green chemistry principles enhances the brand reputation of manufacturers and facilitates easier permitting for capacity expansions in regions with strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of bimetallic metalloporphyrin catalysts in industrial oxidation processes, based on the specific data and findings presented in the patent documentation. These insights are intended to clarify the practical benefits and mechanistic advantages for stakeholders evaluating this technology for adoption in their existing or new production lines.
Q: How does the bimetallic catalyst improve selectivity compared to traditional cobalt salts?
A: The bimetallic structure effectively regulates the catalytic conversion of cycloalkyl hydroperoxide intermediates, preventing their uncontrolled thermal decomposition into aliphatic diacids, thereby significantly increasing the selectivity for cycloalkanols and cycloalkanones.
Q: What are the safety advantages of this new oxidation method?
A: By maintaining low levels of cycloalkyl hydroperoxide accumulation through controlled catalytic conversion, the process reduces the risk of explosive decomposition associated with high concentrations of organic peroxides in traditional high-temperature oxidation systems.
Q: Can this catalyst be used for various cycloalkane substrates?
A: Yes, the patent demonstrates efficacy across a range of substrates including cyclopentane, cyclohexane, cycloheptane, and cyclododecane, offering versatility for producing different fine chemical intermediates and polymer precursors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bimetallic Metalloporphyrin Supplier
At NINGBO INNO PHARMCHEM, we understand the critical importance of deploying advanced catalytic technologies to maintain competitiveness in the fine chemical and pharmaceutical intermediate sectors. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering products with stringent purity specifications and supporting our partners with rigorous QC labs to guarantee that every batch meets the highest standards of quality and consistency required for sensitive oxidation applications.
We invite you to collaborate with us to explore how this bimetallic porphyrin technology can optimize your specific manufacturing processes and drive down operational costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis, along with specific COA data and route feasibility assessments tailored to your production needs. Let us help you unlock the full potential of selective catalytic oxidation for your next project.
