Revolutionizing Corticosteroid Production: A Deep Dive into High-Yield Triamcinolone Acetonide Synthesis
The pharmaceutical industry is constantly seeking more efficient pathways for synthesizing high-value corticosteroids, and the recent disclosure in patent CN114478681A offers a transformative solution for the production of Triamcinolone Acetonide. This high-efficiency fluorine-containing adrenocortical hormone is critical for treating a wide spectrum of conditions ranging from neurodermatitis and eczema to severe arthralgia and bronchial asthma. The patent details a sophisticated preparation method that fundamentally restructures the synthetic route starting from tetraene acetate. Unlike traditional linear sequences that suffer from yield erosion at every isolation step, this novel methodology employs a strategic combination of oxidation, one-pot cyclization, and fluoroketalization. For R&D directors and process chemists, the significance lies in the drastic reduction of production periods and the simultaneous elevation of product quality. By addressing the historical pain points of partial hydrolysis and excessive wastewater generation, this technology represents a pivotal shift towards greener, more cost-effective pharmaceutical intermediates manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
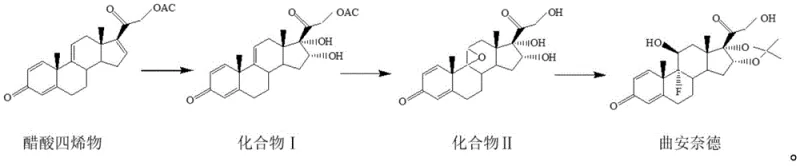
Historically, the industrial synthesis of Triamcinolone Acetonide has been plagued by inefficiencies inherent in multi-step batch processing. As highlighted in the background art, specifically referencing prior patents like CN102863505A, the conventional route involves distinct, isolated stages for oxidation, condensation, hydroxyl bromide epoxy reaction, and fluorination. A critical failure point in these legacy processes occurs during the epoxy reaction at the 9(11) position, where the acetate group at the 21-position is prone to partial hydrolysis. This side reaction not only complicates the purification landscape but also severely impacts the overall mass balance, capping the total yield at approximately 70%. Furthermore, the reliance on repeated solvent exchanges and intermediate isolations generates a substantial volume of chemical wastewater, imposing heavy burdens on environmental treatment facilities and inflating operational expenditures related to waste management and solvent recovery.
The Novel Approach
The breakthrough presented in the current patent data lies in the ingenious consolidation of reaction steps, particularly the transformation of Compound I into Compound II. Instead of separating the bromohydrin formation, epoxidation, and hydrolysis into discrete unit operations, the inventors have developed a one-pot combined feeding process. This approach utilizes perchloric acid and dibromohydantoin in a tetrahydrofuran medium to drive the cascade reaction seamlessly. By maintaining precise temperature controls between 5°C and 52°C and employing a specific mixed alkali solution of potassium hydroxide and sodium sulfite, the process effectively suppresses the unwanted hydrolysis side reactions that plagued previous methods. This integration not only simplifies the operational workflow but also drastically shortens the production cycle. Moreover, the strategic replacement of acetone with tetrahydrofuran in this critical step mitigates the formation of toxic by-products, aligning the synthesis with modern green chemistry principles and offering a robust pathway for cost reduction in pharmaceutical manufacturing.
Mechanistic Insights into the One-Pot Cyclization and Fluorination
To fully appreciate the technical superiority of this route, one must examine the mechanistic nuances of the oxidation and cyclization steps. In Step S1, the oxidation of tetraene acetate is meticulously controlled using potassium permanganate in an acetone-formic acid system at sub-zero temperatures (-5°C to 0°C). This low-temperature regime is crucial for preventing over-oxidation and ensuring the selective formation of the diol functionality in Compound I with high chromatographic purity (reported >97.9%). The subsequent addition of sodium sulfite acts as a quenching agent to reduce excess permanganate, preventing oxidative degradation of the sensitive steroid backbone. Moving to Step S2, the use of dibromohydantoin as a brominating agent in the presence of perchloric acid facilitates the formation of the bromohydrin intermediate, which spontaneously undergoes intramolecular nucleophilic substitution to form the epoxide. The immediate follow-up with alkaline hydrolysis in the same vessel ensures that the 21-acetate is cleaved under controlled conditions, avoiding the random hydrolysis observed in batch-wise additions. This precise orchestration of reagents is the key to achieving the reported total yield of over 84%.
Impurity control is another cornerstone of this advanced synthesis, particularly vital for meeting the stringent specifications required for a reliable pharmaceutical intermediates supplier. The final fluoroketal reaction (Step S3) utilizes hydrofluoric acid in an acetone mixture at extremely low temperatures (-40°C to -30°C). This cryogenic environment is essential for the regioselective introduction of the fluorine atom at the 9-alpha position while simultaneously forming the acetonide protecting group at the 16,17-positions. Any deviation in temperature could lead to defluorination or rearrangement impurities. The patent specifies a rigorous workup involving neutralization with potassium carbonate to pH 7.0-7.5, which effectively removes acidic residues without degrading the acid-sensitive acetonide moiety. The final recrystallization using a dichloromethane-methanol-water system further polishes the crude product, removing trace organic impurities and ensuring the final HPLC content exceeds 99.7%, thereby satisfying the most demanding regulatory requirements for API production.
How to Synthesize Triamcinolone Acetonide Efficiently
Implementing this synthesis route requires strict adherence to the thermal and stoichiometric parameters outlined in the patent examples. The process begins with the preparation of a cooled potassium permanganate solution, which is added dropwise to the tetraene acetate substrate to manage the exotherm effectively. Following the isolation of Compound I, the critical one-pot transformation involves the portion-wise addition of dibromohydantoin to maintain reaction homogeneity and control the rate of bromine release. The subsequent heating phase to 48-52°C drives the cyclization and hydrolysis to completion. Finally, the fluorination step demands specialized equipment capable of handling hydrofluoric acid at cryogenic temperatures, followed by a careful neutralization and a multi-solvent recrystallization protocol.
- Oxidize tetraene acetate using potassium permanganate and formic acid in acetone at -5°C to 0°C to obtain Compound I.
- React Compound I with perchloric acid and dibromohydantoin in THF, followed by alkaline treatment to form Compound II in a single pot.
- Perform fluoroketal reaction on Compound II using hydrofluoric acid and acetone at low temperatures (-40°C to -30°C) to generate the crude product.
- Purify the crude product via dissolution in dichloromethane/methanol, activated carbon decolorization, and water crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible strategic advantages beyond mere technical metrics. The consolidation of multiple reaction steps into fewer unit operations inherently reduces the consumption of utilities such as steam for heating and chilled brine for cooling, leading to significant operational cost savings. By eliminating the need for intermediate isolation and drying between the bromohydrin, epoxy, and hydrolysis stages, the facility can achieve a higher throughput per reactor volume, effectively increasing capacity without capital expansion. This efficiency gain is critical for maintaining competitive pricing in the global market for corticosteroids. Furthermore, the reduction in wastewater volume directly lowers the environmental compliance costs associated with effluent treatment, providing a dual benefit of economic savings and enhanced corporate sustainability profiles.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and the optimization of solvent usage contribute to a leaner cost structure. Specifically, the replacement of large volumes of acetone with tetrahydrofuran in the cyclization step reduces the risk of toxin formation, which in turn lowers the costs associated with hazardous waste disposal and solvent recovery systems. The simplified workflow means fewer man-hours are required for monitoring and handling, allowing labor resources to be allocated more efficiently across the production floor. These cumulative efficiencies result in a substantially lower cost of goods sold (COGS), enabling more aggressive pricing strategies while maintaining healthy margins.
- Enhanced Supply Chain Reliability: Shortening the production period is a decisive factor in improving supply chain resilience. Traditional methods with long cycle times are vulnerable to disruptions; however, this streamlined process ensures faster turnaround from raw material intake to finished goods. The high yield (>84%) means that less raw material (tetraene acetate) is required to produce the same amount of final API, reducing the strain on upstream supply chains. This reliability is paramount for securing long-term contracts with multinational pharmaceutical companies that prioritize consistent delivery schedules and uninterrupted availability of critical dermatological and anti-inflammatory agents.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing common industrial solvents and standard reaction conditions that are easily replicated in large-scale reactors. The significant reduction in chemical wastewater discharge alleviates the pressure on factory treatment plants, ensuring continuous operation even under tightening environmental regulations. This 'green' attribute not only future-proofs the manufacturing site against regulatory changes but also appeals to end-clients who are increasingly mandating sustainable sourcing practices. The ability to scale from pilot batches to 100 MT annual production without process re-engineering demonstrates the robustness of this technology for commercial deployment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. They are derived directly from the comparative data and experimental examples provided in the patent documentation, offering clarity on yield improvements, environmental impact, and scalability. Understanding these aspects is crucial for stakeholders evaluating the feasibility of adopting this technology for their own manufacturing portfolios or sourcing strategies.
Q: How does the new combined feeding process improve yield compared to conventional methods?
A: Conventional methods often suffer from partial hydrolysis at the C21 position during the epoxy formation step, leading to a total yield of only around 70%. The novel approach described in patent CN114478681A integrates the bromohydrin, epoxy, and hydrolysis steps into a single one-pot operation. This minimizes intermediate handling and side reactions, boosting the total yield to over 84% while achieving purity levels exceeding 99.7%.
Q: What are the environmental benefits of switching to this synthesis route?
A: The new process significantly reduces the discharge of chemical wastewater by eliminating multiple isolation and washing steps associated with the traditional multi-step sequence. Furthermore, the substitution of large volumes of acetone with tetrahydrofuran (THF) in the critical cyclization step reduces the load of volatile organic compounds that are prone to forming toxins, thereby lowering the pressure and cost associated with factory wastewater treatment and environmental compliance.
Q: Is this method scalable for commercial production of corticosteroids?
A: Yes, the method is designed for scalability. By simplifying operation steps and shortening the production period through combined feeding, the process enhances throughput. The use of standard solvents like acetone, THF, and hydrofluoric acid, along with robust temperature controls (-40°C to 50°C range), ensures that the reaction can be safely managed and scaled from pilot batches to multi-ton annual commercial production without compromising quality.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triamcinolone Acetonide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity corticosteroids in the global healthcare landscape. Our technical team has thoroughly analyzed the advancements presented in patent CN114478681A and possesses the expertise to implement this state-of-the-art synthesis route. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this one-pot process are fully realized in our manufacturing facilities. Our stringent purity specifications and rigorous QC labs guarantee that every batch of Triamcinolone Acetonide meets or exceeds the 99.7% purity benchmark, providing our partners with the confidence needed for regulatory filings and commercial launch.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis can benefit your specific supply chain needs. By leveraging our capabilities, you can access a Customized Cost-Saving Analysis that quantifies the potential reductions in waste treatment and raw material consumption for your projects. We encourage you to request specific COA data and route feasibility assessments to verify our commitment to quality and efficiency. Together, we can drive the next generation of affordable, high-quality dermatological and anti-inflammatory therapies to the market.
