Advanced Synthesis of 4-Methyl-5-(β-Hydroxyethyl)-Thiazole for Industrial Scale-Up
Advanced Synthesis of 4-Methyl-5-(β-Hydroxyethyl)-Thiazole for Industrial Scale-Up
The global demand for high-purity heterocyclic intermediates, particularly those serving as foundational blocks for vitamins and fine flavors, necessitates continuous innovation in synthetic methodology. Patent CN115819370A introduces a transformative five-step synthesis for 4-methyl-5-(β-hydroxyethyl)-thiazole, also known as thiothiazole, a critical precursor for Vitamin B1 and a valued flavor compound. This novel approach fundamentally shifts the raw material paradigm from expensive halogenated esters to readily available α-acetyl-γ-butyrolactone. By optimizing reaction conditions across chlorination, hydrolysis, and cyclization stages, the process achieves exceptional yields exceeding 96% while significantly simplifying purification protocols. For R&D directors and procurement strategists, this represents a pivotal opportunity to enhance supply chain resilience and reduce the total cost of ownership for thiazole-based manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 4-methyl-5-(β-hydroxyethyl)-thiazole has been plagued by economic and technical inefficiencies inherent to older pathways, such as the method disclosed in CN111635375A. Traditional routes typically rely on methyl acetoacetate and 3-bromo-3-chloropropyl acetate as starting materials, which are not only costly but also introduce significant handling hazards due to their halogen content. As illustrated in the reaction scheme below, these conventional methods often require a cumbersome two-step hydrolysis process to unmask the necessary functional groups, leading to a proliferation of side reactions that complicate downstream purification. The accumulation of by-products not only depresses overall yield but also creates substantial waste streams, posing environmental compliance challenges and inflating operational expenditures for waste treatment facilities.
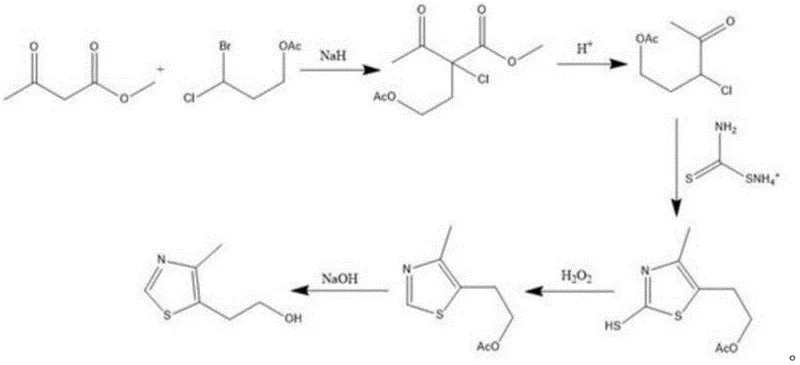
The Novel Approach
In stark contrast, the methodology detailed in CN115819370A leverages α-acetyl-γ-butyrolactone as a superior, cost-effective feedstock that streamlines the entire synthetic sequence. This innovative route initiates with a highly selective chlorination followed by a direct ring-opening hydrolysis, effectively bypassing the multi-step degradation required in legacy processes. The strategic use of ammonium dithiocarbamate, generated in situ from carbon disulfide and ammonia, facilitates a clean condensation reaction that constructs the thiazole core with remarkable precision. By eliminating the need for expensive brominated precursors and reducing the number of unit operations, this novel approach offers a robust pathway for cost reduction in vitamin B1 precursor manufacturing, ensuring a more stable and predictable supply of this essential intermediate for global markets.
Mechanistic Insights into Chlorination and Cyclization
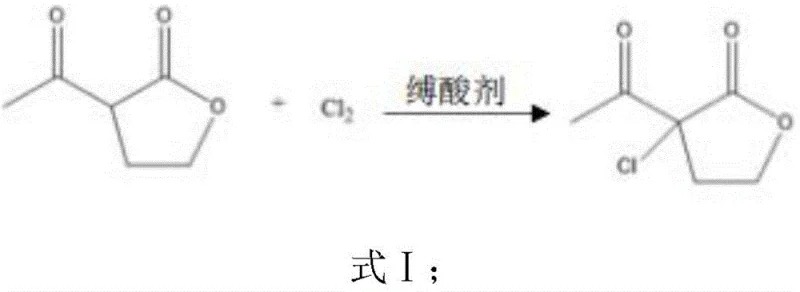
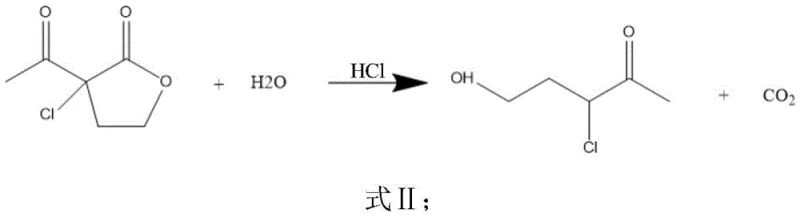
The cornerstone of this improved synthesis lies in the precise control of the initial chlorination and subsequent ring-opening steps, which dictate the purity of the final product. In the first stage, α-acetyl-γ-butyrolactone undergoes electrophilic substitution with chlorine gas in the presence of an acid-binding agent such as sodium bicarbonate or sodium acetate. This reaction is meticulously maintained at low temperatures between 10-20°C to prevent over-chlorination and the formation of polychlorinated impurities. The resulting α-acetyl-α-chloro-γ-butyrolactone is then subjected to acid-catalyzed hydrolysis using hydrochloric acid solution. As shown in the reaction equations below, heating the mixture to 70-90°C induces a clean ring-opening of the lactone moiety, yielding 3-chloro-4-oxo-1-pentanol with high fidelity. This intermediate serves as the perfect electrophile for the subsequent cyclization, ensuring that the carbon backbone remains intact throughout the transformation.
Following the preparation of the chloro-ketone intermediate, the synthesis proceeds through a sophisticated condensation mechanism involving ammonium dithiocarbamate. This nucleophile, prepared separately via the reaction of carbon disulfide and ammonia in dimethyl carbonate, attacks the carbonyl center of the chloro-ketone under acidic conditions. The reaction temperature is carefully ramped from 40°C to 70°C to drive the cyclization forward while minimizing the risk of thermal decomposition. The final step involves an oxidative desulfurization using hydrogen peroxide and concentrated sulfuric acid, which converts the mercapto-thiazole intermediate into the target 4-methyl-5-(β-hydroxyethyl)-thiazole. This oxidative step is crucial for impurity control, as it effectively removes sulfur-containing by-products that could otherwise compromise the organoleptic properties of the final flavor compound or the safety profile of the pharmaceutical intermediate.
How to Synthesize 4-Methyl-5-(β-Hydroxyethyl)-Thiazole Efficiently
Implementing this synthesis requires strict adherence to the optimized parameters defined in the patent to ensure reproducibility and safety at scale. The process begins with the controlled introduction of chlorine gas into an aqueous slurry of the lactone and acid binder, followed by a distinct phase separation to isolate the chlorinated intermediate. Subsequent hydrolysis and neutralization steps must be monitored closely to maintain the integrity of the hydroxyethyl side chain. The condensation reaction demands precise pH control during the addition of hydrochloric acid to facilitate ring closure without degrading the sensitive thiazole nucleus. Finally, the oxidative workup utilizes dichloromethane extraction to separate the organic product from aqueous salts before final rectification.
- Chlorinate α-acetyl-γ-butyrolactone with chlorine gas and an acid-binding agent at 10-20°C.
- Perform ring-opening hydrolysis using hydrochloric acid at 70-90°C to form 3-chloro-4-oxo-1-pentanol.
- React carbon disulfide with ammonia in dimethyl carbonate to generate ammonium dithiocarbamate.
- Condense the chloro-ketone with ammonium dithiocarbamate under acidic conditions to form the thiazole ring.
- Oxidize the mercapto-intermediate with hydrogen peroxide and sulfuric acid, followed by neutralization and rectification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers compelling strategic advantages that extend beyond simple yield improvements. The substitution of high-cost brominated starting materials with commodity chemicals like α-acetyl-γ-butyrolactone and chlorine gas fundamentally alters the cost structure of production. This shift not only lowers the direct material costs but also mitigates the supply risk associated with specialized halogenated reagents that are subject to volatile market pricing and regulatory scrutiny. Furthermore, the simplified workflow reduces the requirement for complex reactor configurations, allowing for faster batch turnover and enhanced asset utilization within existing manufacturing facilities.
- Cost Reduction in Manufacturing: The elimination of expensive 3-bromo-3-chloropropyl acetate and the reduction of hydrolysis steps lead to substantial cost savings in raw material procurement and energy consumption. By avoiding the use of transition metal catalysts or exotic reagents, the process minimizes the need for expensive purification technologies such as scavenger resins or heavy metal removal units. The high overall yield exceeding 96% ensures that less raw material is wasted per kilogram of finished product, directly improving the gross margin profile for manufacturers of Vitamin B1 and related thiazole derivatives.
- Enhanced Supply Chain Reliability: The reliance on bulk commodity chemicals such as chlorine, ammonia, and sulfuric acid ensures a stable and continuous supply of inputs, reducing the likelihood of production stoppages due to raw material shortages. The mild reaction conditions, operating primarily between 10°C and 90°C, place less stress on equipment and reduce maintenance downtime compared to high-pressure or cryogenic alternatives. This operational robustness allows for more accurate forecasting of lead times for high-purity pharmaceutical intermediates, enabling downstream customers to optimize their own inventory levels and production schedules with greater confidence.
- Scalability and Environmental Compliance: The process generates significantly less wastewater and solid waste compared to traditional methods, simplifying effluent treatment and reducing the environmental footprint of the manufacturing site. The use of standard unit operations like liquid-liquid extraction and distillation facilitates seamless scale-up from pilot plant to commercial tonnage without the need for specialized equipment. This scalability ensures that suppliers can rapidly respond to surges in market demand for flavor and fragrance intermediates while maintaining strict adherence to increasingly rigorous environmental regulations regarding halogenated waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and process descriptions provided in the patent documentation, offering clarity on feasibility and performance metrics. Understanding these details is essential for technical teams evaluating the integration of this route into their existing production portfolios.
Q: What are the key advantages of this new synthesis route over conventional methods?
A: The new route replaces expensive 3-bromo-3-chloropropyl acetate with cost-effective α-acetyl-γ-butyrolactone. It eliminates complex multi-step hydrolysis, reduces side reactions, and achieves yields exceeding 96%.
Q: Is this process suitable for large-scale Vitamin B1 production?
A: Yes, the process uses mild reaction conditions (10-90°C) and common reagents (Cl2, HCl, H2O2), making it highly scalable and safe for producing Vitamin B1 intermediates like thiothiazole.
Q: How does the process handle impurity control?
A: Impurities are minimized through controlled low-temperature chlorination and specific oxidative desulfurization. The final rectification step ensures high purity suitable for pharmaceutical and flavor applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Methyl-5-(β-Hydroxyethyl)-Thiazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthesis plays in the competitiveness of the global fine chemicals market. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN115819370A are fully realized in practical manufacturing environments. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 4-methyl-5-(β-hydroxyethyl)-thiazole meets the exacting standards required for pharmaceutical and food grade applications.
We invite you to collaborate with us to leverage this advanced technology for your supply chain needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing capabilities can drive value and efficiency for your organization.
