Advanced Purification Technology for Critical Antibiotic Intermediates: Scaling High-Purity Production
Advanced Purification Technology for Critical Antibiotic Intermediates: Scaling High-Purity Production
The global demand for broad-spectrum antibiotics such as thiamphenicol and florfenicol necessitates a robust and reliable supply chain for their key precursors. Patent CN110229087B introduces a transformative purification methodology specifically designed for (1R,2R)-1-[(4-methylsulfonyl)phenyl]-2-amino-1,3-propanediol, a critical chiral building block in veterinary and human medicine. This technology addresses the persistent challenge of removing structurally similar impurities that traditional refining methods fail to eliminate. By leveraging the reversible chemistry of Schiff base formation, the process ensures that the final intermediate meets the rigorous purity specifications required for downstream synthesis, thereby safeguarding the quality and safety of the finished pharmaceutical products.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
In the established manufacturing landscape for chloramphenicol-family antibiotics, the production of the core amino-diol intermediate typically relies on the reduction of D-p-methylsulfonylphenylserine ethyl ester. However, the subsequent purification of the resulting crude amine has long been a bottleneck for quality control. Conventional strategies predominantly utilize simple recrystallization from organic solvents, which often proves inadequate when impurities share nearly identical solubility profiles and chiral configurations with the target molecule. Furthermore, while techniques like column chromatography could theoretically separate these close analogs, they are economically and operationally prohibitive for large-scale industrial production due to high solvent consumption, low throughput, and significant waste generation. Consequently, manufacturers frequently struggle with residual impurities that persist through to the final drug substance, posing risks to regulatory compliance and patient safety.
The Novel Approach
The innovative strategy outlined in the patent circumvents these limitations by introducing a chemical derivatization step prior to the final isolation. Instead of attempting to purify the free amine directly, the process converts the crude (1R,2R)-1-[(4-methylsulfonyl)phenyl]-2-amino-1,3-propanediol into a Schiff base through condensation with a ketone. This transformation fundamentally alters the physicochemical properties of the molecule, creating a distinct crystalline lattice that allows for the effective exclusion of impurities which do not undergo the same structural conversion or crystallization behavior. Following the isolation of the purified Schiff base, a mild hydrolysis step regenerates the free amine. This "protect-purify-deprotect" logic effectively breaks the symmetry of solubility that plagues direct recrystallization, enabling the production of ultra-high purity material without the need for complex chromatographic separation.
Mechanistic Insights into Schiff Base Derivatization Purification
The core of this purification technology lies in the reversible nucleophilic addition of the primary amine group to the carbonyl carbon of a ketone, forming an imine or Schiff base linkage. As illustrated in the reaction scheme below, the crude amine reacts with ketones such as acetone or butanone under mild thermal conditions. This reaction is highly selective for the primary amine functionality, and the resulting Schiff base exhibits significantly different polarity and crystal packing energy compared to the starting amine and its associated impurities. The steric bulk introduced by the alkyl groups of the ketone (R1 and R2) enhances the crystallinity of the derivative, facilitating a sharp precipitation from the reaction mixture upon cooling. This phase transition acts as a powerful filter, leaving non-reactive or poorly crystallizing impurities in the mother liquor.
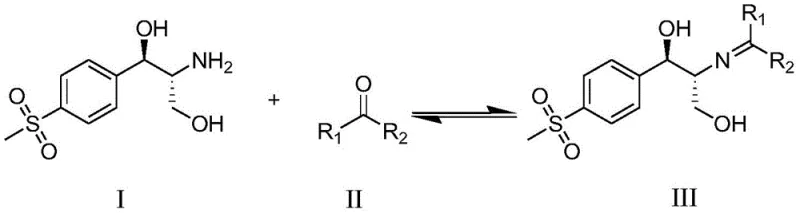
Following the isolation of the crystalline Schiff base, the process employs a hydrolysis mechanism to restore the original amine functionality. By heating the Schiff base in an aqueous environment, the equilibrium shifts back towards the reactants, cleaving the carbon-nitrogen double bond to release the ketone and regenerate the free amine. This step is crucial because it allows the purification to occur on the derivative while delivering the exact chemical entity required for the next synthetic step. The hydrolysis conditions are carefully controlled to prevent racemization or degradation of the sensitive diol structure. The ability to toggle between the free amine and the protected imine form provides a versatile handle for process chemists to optimize yield and purity independently, ensuring that the final product possesses the necessary stereochemical integrity for biological activity.
How to Synthesize (1R,2R)-1-[(4-methylsulfonyl)phenyl]-2-amino-1,3-propanediol Efficiently
The implementation of this purification route requires precise control over reaction temperatures and solvent systems to maximize the efficiency of the crystallization steps. The patent data outlines a streamlined protocol that begins with the condensation of the crude amine with a selected ketone in a lower alcohol solvent, followed by a controlled cooling ramp to induce nucleation. Once the Schiff base is isolated, it undergoes aqueous hydrolysis, after which the ketone is removed via distillation, and the product is crystallized using an anti-solvent strategy. This sequence is designed to be robust and reproducible, minimizing the risk of oiling out or amorphous precipitation which can trap impurities. For detailed operational parameters and specific stoichiometric ratios validated in the patent examples, please refer to the standardized synthesis guide below.
- React crude amine with a ketone (e.g., acetone) in an alcoholic solvent at 10-80°C, then cool to -20°C to crystallize the Schiff base intermediate.
- Hydrolyze the isolated Schiff base with water at 30-105°C to regenerate the amine.
- Concentrate the mixture, add an organic anti-solvent, and cool to -15 to 25°C to crystallize the final high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this purification technology represents a significant strategic advantage in securing high-quality raw materials. The shift from inefficient recrystallization to a derivatization-based purification model resolves long-standing issues regarding batch-to-batch consistency and impurity profiles. By eliminating the reliance on chromatographic purification, the process drastically reduces solvent usage and processing time, leading to substantial cost savings in manufacturing overhead. Furthermore, the use of commodity chemicals such as acetone, methanol, and water as primary reagents ensures that the supply chain remains resilient against fluctuations in the availability of exotic or expensive specialty reagents. This operational simplicity translates directly into improved reliability of supply and reduced lead times for critical antibiotic intermediates.
- Cost Reduction in Manufacturing: The elimination of column chromatography and the use of common, low-cost solvents significantly lower the overall production cost. The process avoids the need for expensive silica gel or resin beds and reduces the volume of hazardous waste generated, simplifying disposal logistics and reducing environmental compliance costs. Additionally, the recovery and recycling of the ketone reagent during the hydrolysis distillation step further enhance the economic efficiency of the process, making it highly competitive for large-volume production.
- Enhanced Supply Chain Reliability: The robustness of the crystallization steps ensures high yields and consistent quality, reducing the risk of batch failures that can disrupt downstream API synthesis. The method's tolerance for variations in crude feedstock quality means that suppliers can maintain steady production schedules even when upstream reduction steps show minor fluctuations. This stability is critical for maintaining continuous inventory levels for major pharmaceutical customers who require Just-In-Time delivery of GMP-grade intermediates.
- Scalability and Environmental Compliance: The process has been successfully demonstrated at the kilogram scale, proving its viability for commercial expansion without the engineering challenges associated with scaling chromatographic columns. The reliance on standard unit operations like filtration, distillation, and crystallization allows for easy integration into existing multipurpose chemical plants. Moreover, the reduced solvent intensity and the ability to recycle reagents align with modern green chemistry principles, helping manufacturers meet increasingly stringent environmental regulations and sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this purification technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the method's capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this process into their current manufacturing workflows.
Q: Why is conventional recrystallization insufficient for this intermediate?
A: Conventional recrystallization often fails because impurities possess similar chiral structures and chemical properties to the target molecule, making separation via simple solubility differences ineffective without derivatization.
Q: What purity levels can be achieved with this Schiff base method?
A: The patented method consistently achieves HPLC purity greater than 99.8%, with any single impurity reduced to less than 0.1%, meeting stringent pharmaceutical standards.
Q: Is this purification process suitable for industrial scale-up?
A: Yes, the process avoids complex column chromatography and utilizes standard unit operations like crystallization and distillation, as demonstrated by successful kilogram-scale examples in the patent data.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (1R,2R)-1-[(4-methylsulfonyl)phenyl]-2-amino-1,3-propanediol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the purity of key intermediates dictates the success of the entire pharmaceutical value chain. Our technical team has extensively analyzed advanced purification protocols like the one described in CN110229087B to ensure our manufacturing processes meet the highest global standards. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, utilizing state-of-the-art facilities equipped with rigorous QC labs to verify every batch. Our commitment to stringent purity specifications ensures that the (1R,2R)-1-[(4-methylsulfonyl)phenyl]-2-amino-1,3-propanediol we supply is perfectly suited for the synthesis of high-quality thiamphenicol and florfenicol.
We invite potential partners to engage with our technical procurement team to discuss how our advanced manufacturing capabilities can support your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized processes can reduce your overall cost of goods. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your production needs, ensuring a seamless and efficient supply partnership.
