Advanced Synthesis of Cannabinoid Receptor Modulators for Commercial Pharmaceutical Production
The pharmaceutical industry is currently witnessing a paradigm shift in the development of cannabinoid receptor modulators, driven by the urgent need for safer, more selective therapeutic agents that avoid the psychotropic side effects associated with traditional cannabis derivatives. Patent CN109206381B, published in late 2021, introduces a groundbreaking preparation method for a compound intermediate capable of regulating the activity of the cannabinoid receptor CB2, offering a distinct advantage over legacy synthesis routes. This technology addresses the critical bottleneck in medicinal chemistry where the complexity of heterocyclic synthesis often clashes with the economic and environmental demands of modern Good Manufacturing Practice (GMP) production. By leveraging a novel four-step sequence that prioritizes mild reaction conditions and high atom economy, this patent provides a robust foundation for the commercial scale-up of complex pharmaceutical intermediates. The strategic value of this intellectual property lies not just in the chemical novelty, but in its inherent design for manufacturability, ensuring that the transition from laboratory bench to multi-ton reactor is seamless and economically viable for global supply chains. 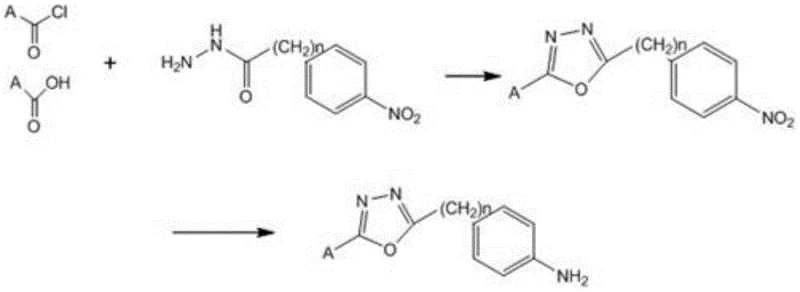
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cannabinoid analogs has been plagued by significant technical hurdles that impede efficient commercial production, as evidenced by prior art such as US20100056507. Traditional routes often rely on harsh reaction conditions that necessitate extreme temperatures or pressures, leading to substantial energy consumption and increased operational risks in a plant setting. Furthermore, these legacy methods frequently utilize toxic reagents and solvents that pose severe challenges for waste management and environmental compliance, driving up the cost of disposal and requiring specialized corrosion-resistant equipment. The formation of impurities is another critical drawback, as side reactions in conventional pathways often generate difficult-to-remove byproducts that compromise the purity profile required for pharmaceutical applications. These factors collectively result in lower overall yields and extended production cycles, creating bottlenecks that affect the reliability of a reliable pharmaceutical intermediate supplier. Consequently, procurement teams face inflated costs and unpredictable lead times, while R&D directors struggle with batch-to-batch variability that threatens regulatory approval.
The Novel Approach
In stark contrast, the methodology disclosed in CN109206381B represents a significant technological iteration that systematically dismantles these barriers through a green chemistry approach. The new route eliminates the use of toxic substances entirely, thereby reducing the hazard profile of the manufacturing process and minimizing the corrosion damage to standard stainless steel reactors. By optimizing the reaction parameters to operate under milder conditions, the process significantly reduces energy input and simplifies the thermal management requirements for large-scale vessels. The design of the synthesis pathway inherently suppresses side reactions, leading to a cleaner crude product profile that simplifies downstream purification and reduces solvent consumption. This operational simplicity translates directly into cost reduction in pharmaceutical intermediate manufacturing, as fewer processing steps and less rigorous waste treatment are required. Ultimately, this novel approach ensures a high yield of the obtained intermediate, providing a stable and efficient supply source for downstream drug development programs.
Mechanistic Insights into Oxadiazole Cyclization and Catalytic Reduction
The core of this synthetic innovation lies in the efficient construction of the oxadiazole heterocyclic core, a structural motif essential for the biological activity of the target cannabinoid modulator. The cyclization reaction (Step S3) involves the condensation of the imidate salt (Formula IV) with the hydrazide intermediate (Formula III) in an ethanol solvent system. This transformation is kinetically favored at reflux temperatures between 80-100°C, where the thermal energy facilitates the nucleophilic attack and subsequent ring closure without degrading the sensitive functional groups present on the tetrahydronaphthalene scaffold. The choice of ethanol as the sole solvent for this step is particularly advantageous, as it acts as both a reaction medium and a proton source, streamlining the workup procedure and avoiding the need for complex solvent swaps that often introduce impurities. 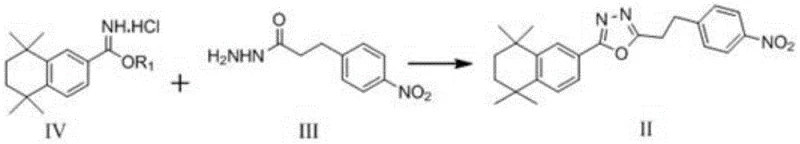
Following the ring formation, the final step involves the selective reduction of the nitro group to a primary amine, a transformation that is critical for the biological function of the CB2 modulator. This reduction (Step S4) utilizes hydrazine hydrate in the presence of graphite, a heterogeneous catalyst system that offers superior selectivity compared to traditional metal-catalyzed hydrogenation. The use of graphite eliminates the risk of heavy metal contamination, a common concern in pharmaceutical synthesis that requires expensive scavenging steps to meet strict residual metal specifications. The reaction proceeds smoothly at temperatures between 60-100°C over a period of 70-80 hours, ensuring complete conversion while maintaining the integrity of the oxadiazole ring. 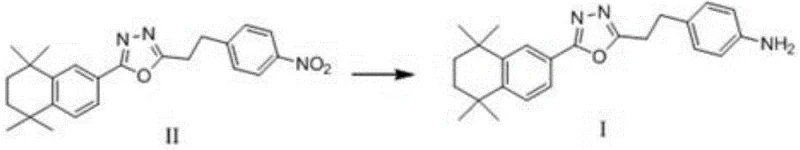
How to Synthesize Cannabinoid Receptor Intermediate Efficiently
The implementation of this synthesis route requires precise control over reaction parameters to maximize yield and purity, particularly during the acyl chlorination and amination stages which set the foundation for the subsequent cyclization. The process begins with the conversion of the nitrile precursor to an imidate salt using acetyl chloride in alcohol solvents at controlled low temperatures, followed by the formation of the hydrazide via reflux with hydrazine hydrate. These initial steps must be monitored closely via TLC to ensure complete conversion before proceeding to the critical ring-closing step. The detailed standardized synthesis steps, including specific molar ratios, addition rates, and workup procedures, are outlined in the technical guide below to ensure reproducibility across different manufacturing sites.
- Perform acyl chlorination on the nitrile precursor (Formula VI) using acetyl chloride in alcohol solvents at 20-30°C to generate the imidate salt (Formula IV).
- Conduct amination reaction on the ketone derivative (Formula V) with hydrazine hydrate under reflux conditions to obtain the hydrazide intermediate (Formula III).
- Execute cyclization between Formula IV and Formula III in ethanol at 80-100°C to form the oxadiazole core structure (Formula II).
- Complete the synthesis via catalytic reduction of the nitro group in Formula II using hydrazine hydrate and graphite to yield the final amine product (Formula I).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patent-protected synthesis route offers substantial strategic advantages that extend beyond simple unit cost metrics. The elimination of toxic reagents and the reduction of corrosive byproducts significantly lower the total cost of ownership for the manufacturing process, as it reduces the need for specialized hazardous waste disposal and expensive alloy-lined equipment. This operational efficiency translates into a more resilient supply chain, as the process is less susceptible to regulatory shutdowns or environmental compliance issues that often plague traditional chemical manufacturing. Furthermore, the high yield and purity profile of the intermediate reduce the volume of raw materials required per kilogram of final product, optimizing inventory turnover and reducing working capital requirements. By partnering with a manufacturer utilizing this technology, companies can secure a more stable supply of high-purity pharmaceutical intermediates with reduced lead time for high-purity pharmaceutical intermediates, ensuring continuity of their own drug development pipelines.
- Cost Reduction in Manufacturing: The process achieves cost optimization primarily through the elimination of expensive transition metal catalysts and the associated purification steps required to remove heavy metal residues. By utilizing graphite as a catalyst for the reduction step, the method avoids the high procurement costs of precious metals like palladium or platinum, while also removing the need for specialized metal scavenger resins. Additionally, the use of common solvents like ethanol and isopropanol throughout the synthesis reduces solvent procurement costs and simplifies solvent recovery systems. The overall simplification of the workflow means less labor hours and lower energy consumption per batch, driving down the variable cost of production significantly without compromising quality.
- Enhanced Supply Chain Reliability: The reliance on easily obtained raw materials ensures that the supply chain is not vulnerable to the geopolitical or logistical bottlenecks often associated with exotic or highly regulated reagents. The robustness of the reaction conditions means that the process can be easily transferred between different manufacturing facilities without significant re-validation, providing flexibility in sourcing strategies. This redundancy is crucial for maintaining supply continuity in the face of global disruptions, allowing procurement teams to diversify their supplier base without risking quality deviations. The reduced generation of hazardous waste also minimizes the risk of logistical delays related to waste transport and disposal permits.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex pharmaceutical intermediates, with reaction conditions that are safe and manageable in large-scale reactors. The absence of toxic substances and the generation of less three wastes align perfectly with increasingly stringent global environmental regulations, future-proofing the manufacturing asset against tighter compliance standards. This environmental stewardship not only reduces regulatory risk but also enhances the corporate sustainability profile of the supply chain, a key metric for modern ESG-focused procurement strategies. The simplicity of the workup procedures further facilitates rapid scale-up, allowing for quicker response times to market demand fluctuations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent specifications and practical manufacturing considerations. These insights are intended to clarify the operational benefits and technical feasibility for stakeholders evaluating this route for their supply chain. Understanding these details is essential for making informed decisions about sourcing strategies and technology transfer.
Q: How does this patent improve upon previous synthesis methods for cannabinoid intermediates?
A: Unlike prior art such as US20100056507 which suffers from harsh conditions and toxic reagents, patent CN109206381B utilizes mild temperatures and avoids toxic substances, significantly reducing equipment corrosion and environmental waste while maintaining high yields.
Q: What are the critical reaction conditions for the cyclization step?
A: The cyclization reaction (Step S3) requires refluxing in ethanol at temperatures between 80-100°C for 3 to 10 hours. This specific thermal profile ensures complete conversion to the oxadiazole ring while minimizing side reactions.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is designed for scalability. It uses easily obtained raw materials, simple operation steps, and generates less three wastes, making it highly suitable for commercial scale-up of complex pharmaceutical intermediates without requiring specialized hazardous handling infrastructure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cannabinoid Receptor Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of novel therapeutic agents depends on a partner who can bridge the gap between innovative patent chemistry and industrial reality. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields demonstrated in patent CN109206381B are realized in full-scale manufacturing. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of cannabinoid receptor intermediate meets the exacting standards required for global pharmaceutical registration. We understand the critical nature of impurity control and are committed to delivering a product profile that supports your regulatory filings without delay.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your supply chain to drive efficiency and value. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make a data-driven decision that secures your supply of this critical pharmaceutical building block for the future.
