Advanced Suzuki Coupling Strategy for High-Purity Cystic Fibrosis Drug Intermediates and Commercial Scale-Up
Introduction to Next-Generation Cystic Fibrosis Intermediate Synthesis
The pharmaceutical landscape for cystic fibrosis treatment has been significantly advanced by the development of CFTR modulators, with Lumacaftor standing out as a critical therapeutic agent. As detailed in patent CN108658851B, the demand for high-quality intermediates such as 2-chloro-3-methyl-6-acylaminopyridine has necessitated a re-evaluation of traditional synthetic methodologies. This patent introduces a robust and innovative approach that leverages modern cross-coupling chemistry to overcome the limitations of earlier processes. By shifting away from hazardous oxidation protocols, this new method provides a safer, more efficient pathway for producing key building blocks required for anti-cystic fibrosis active ingredients. The strategic implementation of Suzuki-Miyaura coupling in this context represents a significant technological leap, ensuring that manufacturers can meet the stringent purity and safety standards required by global regulatory bodies while maintaining economic viability.
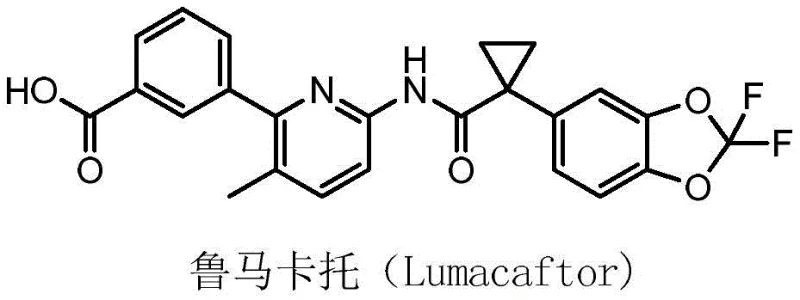
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-chloro-3-methyl-6-aminopyridine derivatives has relied heavily on pathways involving the formation of pyridine N-oxides. As illustrated in prior art literature such as WO2007056341, these conventional routes typically commence with 2-amino-5-picoline, which undergoes pivaloylation followed by oxidation using peroxides. This reliance on peroxides introduces substantial safety hazards, including the risk of thermal runaway and explosion, which are unacceptable in large-scale industrial settings. Furthermore, the subsequent chlorination step requires phosphorus oxychloride, and the final deprotection necessitates strong acidic hydrolysis. These harsh conditions not only pose severe corrosion risks to stainless steel reactor equipment but also generate significant amounts of acidic waste, complicating environmental compliance and increasing the overall cost of waste treatment. Additionally, older methods often suffer from poor regioselectivity, leading to mixtures of isomers that are difficult and costly to separate, thereby reducing the overall yield and purity of the final intermediate.
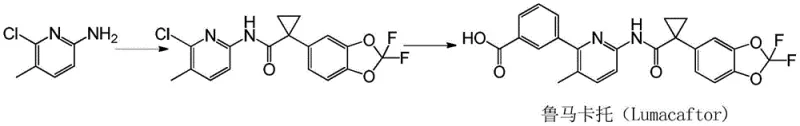
The Novel Approach
In stark contrast to the perilous N-oxide pathways, the methodology disclosed in CN108658851B utilizes a sophisticated Suzuki-Miyaura cross-coupling strategy to install the critical methyl group. This novel approach begins with the readily available 2-chloro-3-bromo-6-aminopyridine, which is first protected via acylation to prevent interference during the coupling step. The core transformation involves reacting this bromo-intermediate with methylboronic acid in the presence of a palladium catalyst. This catalytic cycle operates under significantly milder conditions compared to the extreme temperatures and corrosive acids of the past. By avoiding the use of peroxides entirely, the new process eliminates the associated explosion risks and removes the need for specialized explosion-proof infrastructure. Moreover, the selectivity of the palladium catalyst ensures that the methyl group is installed precisely at the 3-position, minimizing the formation of regioisomeric impurities. This precision translates directly into simplified downstream purification processes, higher overall yields, and a much cleaner impurity profile, making it an ideal candidate for reliable pharmaceutical intermediate supplier operations aiming for GMP compliance.
Mechanistic Insights into Pd-Catalyzed Suzuki Coupling
The heart of this synthetic innovation lies in the mechanistic elegance of the palladium-catalyzed cross-coupling reaction. The process initiates with the oxidative addition of the palladium(0) catalyst into the carbon-bromine bond of the acylated pyridine substrate. This step is facilitated by the electron-deficient nature of the pyridine ring, which activates the C-Br bond towards metal insertion. Following oxidative addition, the methylboronic acid, activated by a base such as sodium bicarbonate or carbonate, undergoes transmetallation with the palladium complex. This transfers the methyl group to the metal center, forming a key organopalladium intermediate. The final step is reductive elimination, where the carbon-carbon bond between the pyridine ring and the methyl group is formed, regenerating the active palladium(0) catalyst to continue the cycle. This catalytic turnover is highly efficient, allowing for the use of low catalyst loadings, which is crucial for minimizing heavy metal residues in the final product. The choice of ligand and solvent system further optimizes this cycle, ensuring that the reaction proceeds to completion with minimal side reactions such as homocoupling or deboronation.
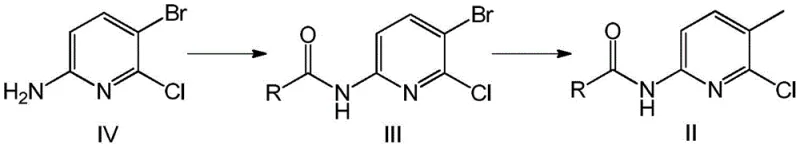
From an impurity control perspective, this mechanism offers distinct advantages over electrophilic substitution or radical-based methods. Traditional methylation strategies often lack specificity, leading to methylation at the nitrogen atom or other carbon positions on the ring. However, the Suzuki coupling is inherently chemoselective for the halogenated position. The acyl protecting group on the amine further enhances this selectivity by preventing the amine from coordinating with the palladium catalyst in a way that might inhibit the reaction or lead to side products. Furthermore, the reaction conditions are sufficiently mild to preserve the integrity of the chloro-substituent at the 2-position, which is vital for subsequent downstream functionalization. The result is a process that consistently delivers high-purity intermediates with a predictable and manageable impurity profile, significantly reducing the burden on analytical quality control teams and ensuring batch-to-batch consistency essential for API manufacturing.
How to Synthesize 2-Chloro-3-Methyl-6-Acylaminopyridine Efficiently
The practical execution of this synthesis involves a straightforward two-step sequence that is highly amenable to standard chemical processing equipment. The first stage involves the protection of the amino group on the starting bromo-pyridine using common acylating agents like pivaloyl chloride or acetyl chloride in the presence of a base. This step is exothermic but easily controlled, yielding the protected intermediate in high purity. The second stage is the critical Suzuki coupling, where the protected intermediate is reacted with methylboronic acid. The reaction is typically conducted in a biphasic solvent system or a mixture of organic solvents and water, heated to reflux under an inert atmosphere. Workup involves simple phase separation and crystallization, avoiding complex chromatographic purifications. For a detailed breakdown of the specific operational parameters, reagent ratios, and workup procedures validated in the patent examples, please refer to the standardized guide below.
- Acylation of 2-chloro-3-bromo-6-aminopyridine with acid chloride or carboxylic acid to form the protected bromo-intermediate.
- Suzuki-Miyaura coupling reaction using methylboronic acid and a palladium catalyst to replace the bromine atom with a methyl group.
- Deprotection of the acyl group under acidic conditions to yield the final 2-chloro-3-methyl-6-aminopyridine intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers compelling strategic advantages that extend beyond mere chemical novelty. The primary benefit lies in the drastic simplification of the safety infrastructure required for production. By eliminating the need for peroxide handling and storage, facilities can avoid the high costs associated with hazard mitigation, specialized containment systems, and rigorous safety monitoring protocols. This reduction in operational complexity directly translates to lower overhead costs and a reduced risk of production stoppages due to safety incidents. Furthermore, the avoidance of strong acids and phosphorus oxychloride mitigates equipment corrosion, extending the lifespan of reactors and piping, and reducing maintenance downtime. These factors collectively contribute to a more resilient and cost-effective supply chain, ensuring consistent delivery of critical intermediates without the volatility associated with hazardous chemical logistics.
- Cost Reduction in Manufacturing: The new process significantly lowers manufacturing costs by removing the need for expensive and hazardous oxidizing agents and corrosive reagents. The high selectivity of the Suzuki coupling reduces the formation of difficult-to-remove impurities, which minimizes the loss of material during purification steps and improves the overall mass balance. Additionally, the ability to use lower catalyst loadings and recover solvents efficiently further drives down the cost of goods sold. This economic efficiency allows for competitive pricing strategies while maintaining healthy margins, making it an attractive option for cost reduction in API manufacturing initiatives.
- Enhanced Supply Chain Reliability: Reliability is bolstered by the use of stable, commercially available starting materials that are not subject to the same supply constraints as specialized peroxides or unstable N-oxide intermediates. The robustness of the reaction conditions means that the process is less sensitive to minor variations in temperature or mixing, leading to higher batch success rates. This consistency ensures that production schedules can be met with greater certainty, reducing lead time for high-purity pharmaceutical intermediates and preventing bottlenecks in the downstream synthesis of the final active pharmaceutical ingredient.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with industrial production in mind. The absence of explosive peroxides and the generation of less hazardous waste streams simplify the environmental permitting process and reduce the cost of waste disposal. The milder reaction conditions also allow for the use of standard glass-lined or stainless steel reactors without the need for exotic alloys to resist corrosion. This ease of scale-up facilitates the commercial scale-up of complex pharmaceutical intermediates, enabling manufacturers to rapidly respond to market demand increases without significant capital investment in new specialized infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is the Suzuki coupling route preferred over traditional N-oxide methods for this intermediate?
A: Traditional methods rely on hazardous peroxides for oxidation and strong acids for hydrolysis, posing safety risks and equipment corrosion issues. The Suzuki route avoids these dangerous reagents, offering milder conditions and higher safety profiles suitable for industrial production.
Q: What represents the key quality advantage of the new synthetic pathway?
A: The new pathway utilizes highly selective palladium catalysis which minimizes side reactions and isomeric impurities often found in non-catalytic halogenation methods, resulting in a cleaner impurity profile and higher purity final product.
Q: Is this method scalable for commercial API manufacturing?
A: Yes, the process uses commercially available starting materials like 2-chloro-3-bromo-6-aminopyridine and avoids unstable intermediates. The reaction conditions are robust and have demonstrated high yields in laboratory examples, indicating strong potential for ton-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Chloro-3-Methyl-6-Acylaminopyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the development of life-saving medications like Lumacaftor. Our team of expert chemists has thoroughly analyzed the methodology presented in CN108658851B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering high-purity 2-chloro-3-methyl-6-acylaminopyridine that meets stringent purity specifications through our rigorous QC labs. Our state-of-the-art facilities are equipped to handle palladium-catalyzed reactions safely and efficiently, ensuring that every batch delivered to our partners adheres to the highest quality standards required for pharmaceutical applications.
We invite you to collaborate with us to optimize your supply chain for cystic fibrosis therapeutics. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can support your long-term production goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
